Abstract
Acne vulgaris, typically caused by Cutibacterium acnes (C. acnes) involves chronic inflammation of the sebaceous follicles and is the most common skin disease, afflicting 85% of adolescents. We previously isolated 2 novel phenolic glycosides, 2-caffeoyl-3-hydroxy-3-methylbutyric 4′-β-D-glucopyranosyloxy-3′-hydroxybenzyl ester (citrulluside H [CH]) and 2-caffeoyl-3-hydroxy-3-methylbutyric 4′-β-
Introduction
Acne vulgaris is a rash that appears on the forehead, cheeks, around the mouth, and on the lower jaw, and mainly occurs during adolescence and young adulthood. This is because increased hormone secretion, especially of androgens (such as testosterone) during puberty stimulates the sebaceous glands, resulting in excessive sebum production. Therefore, this condition can be considered a physiological phenomenon. However, these lesions, if left untreated, eventually leave sequelae called acne scars or acne keloids. When sebum accumulates in the pores, closed and open comedones are formed. Acne bacilli interact with this sebum and produce various inflammatory substances that cause inflammation, resulting in a papule.1‐3 It is well known that these symptoms are caused by overgrowth of Cutibacterium acnes (C. acnes) a Gram-positive anaerobic bacterium present within hair follicles and on the skin surface. Thus, compounds that inhibit C.acnes proliferation, promote sebum secretion, and inhibit keratinization of the funnel area have been prescribed as skin care products to control acne. In recent years, compounds extracted from natural products, such as thyme oil, tea tree oil, and natural honey, have been reported to inhibit C acnes proliferation and have been used as antibacterial agents.4‐9 For example, quercetin, a polyphenolic flavonoid compound in numerous plants, induces anti-inflammatory effects by attenuating toll-like receptor (TLR) 2 pathway signaling.10‐12 Several key results regarding the determinants of the severity of inflammatory acne have been obtained in recent years; (1) patients with more severe acne have lower C. acnes strain diversity than healthy individuals and (2) the relative abundance of C. acnes in metagenomic studies is similar in patients with acne and healthy individuals (though there are some reports of slightly higher levels in healthy individuals), indicating that C. acnes overgrowth is not associated with the severity of acne.13‐15 In other words, a loss of diversity in the skin surface microbiome and loss of balance between C. acnes phylotypes are thought to trigger acne. 16 Therefore, it is believed that restoring the diversity of the skin microbiome may effectively ameliorate the severity of acne.
Watermelon (Citrullus lanatus) is an annual herb in the family Cucurbitaceae that grows as a vine. 17 It is believed to have originated from southern Africa, particularly the western Kalahari region of Botswana and Namibia. Watermelon fruits are about 90% water and low in calories; however, they contain several functional components with beneficial physiological effects.18‐21 We previously isolated phytol from watermelon sprouts and the novel phenolic glycosides citrulluside H (CH) and citrulluside T (CT) from young watermelon fruits, which showed anti-tumor and anti-oxidative activities, respectively.22‐24
In this study, we further investigated the function of these recently isolated phenolic glycosides, including their inhibitory effects on the growth of C. acnes and inflammatory effects. C. acnes releases pro-inflammatory cytokines and chemokines via the innate immune receptors TLR2 and TLR4, and the 2 novel phenol glycosides induced anti-inflammatory effects by reducing the activity of nuclear factor κB (NF-κB), which is activated by TLRs.
Results and Discussion
In our previous study, we isolated the 2 novel phenolic glycosides CH and CT (Figure 1) from young watermelon fruits and showed that they suppressed UVB-induced matrix metalloproteinase expression in normal human dermal fibroblasts by scavenging of intracellular reactive oxygen species (ROS). We investigated the potential clinical value of these substances for the treatment of acne.

Appearance of young watermelon (Citrullus lanatus) and chemical structures of citrulluside H (CH) and citrulluside T (CT).
To examine the inhibitory effects of CH and CT on the growth of C. acnes, disk diffusion and turbidimetry assays were performed. As shown in Figure 2A and 2B, neither compound had an inhibitory effect on the growth of C. acnes. C. acnes overgrowth promotes inflammation of the hair follicle and activates the host immune system by increasing the production of inflammatory cytokines. We further examined the effects of CH and CT on heat-killed C. acnes (hk-C. acnes)-stimulated inflammation in murine macrophage RAW264.7 cells. The amount of nitric oxide (NO) in hk-C. acnes-stimulated RAW264.7 cell culture medium increased by approximately 10-fold compared with levels in untreated cells. However, while there were no significant differences in NO among groups treated with both compounds at up to 100 µM, this increase in NO concentration was strongly inhibited in RAW264.7 cells treated with 200 µM of either CH or CT by 73% and 85%, respectively (IC50 CH; 120 ± 12 µM, IC50 CT; 106 ± 8 µM). Quercetin, found in plants, has been reported to exhibit anti-inflammatory effects via inhibition of NO synthesis by hemeoxygenase-1 (HO-1) induction.25‐27 We assessed the anti-inflammatory effects of quercetin, CH, and CT. In our experimental system, the IC50 value of quercetin for inhibition of NO synthesis was 25 ± 6.3 μM. Both compounds isolated from watermelon required a formulation that was roughly 8 times more active than quercetin. By contrast, the IC50 value of dexamethasone, an anti-inflammatory agent, was 226 ± 4.5 μM, indicating that the concentrations required to achieve the anti-inflammatory effects of both compounds were comparable to the prescription concentration of dexamethasone (Figure 3).

Cutibacterium acnes growth inhibition induced by phenolic glycosides isolated from young watermelon (Citrullus lanatus). (A) Sterilized 6-mm filter paper disks were impregnated with 15 µL of each test compound solution (6, 30, and 60 μg/mL) and placed on the surface of C. acnes inoculated GAM agar plates. The plates were incubated under anaerobic condition at 37 °C for 48 h, and the zones of inhibition were measured in mm. Values are expressed as means ± standard error (SE), (n = 9). (B) Inhibitory effects of CH and CT on C. acnes growth determined by a turbidimetry assay. C. acnes was grown in either the absence or presence of test compounds (25 or 50 µM) at 37 °C for 24, 48, and 72 h under anaerobic conditions. After incubation for the indicated times, 200 µL of the culture media was transferred to a well of a 96-well plate and the absorbance at 600 nm was measured immediately using a microplate reader.
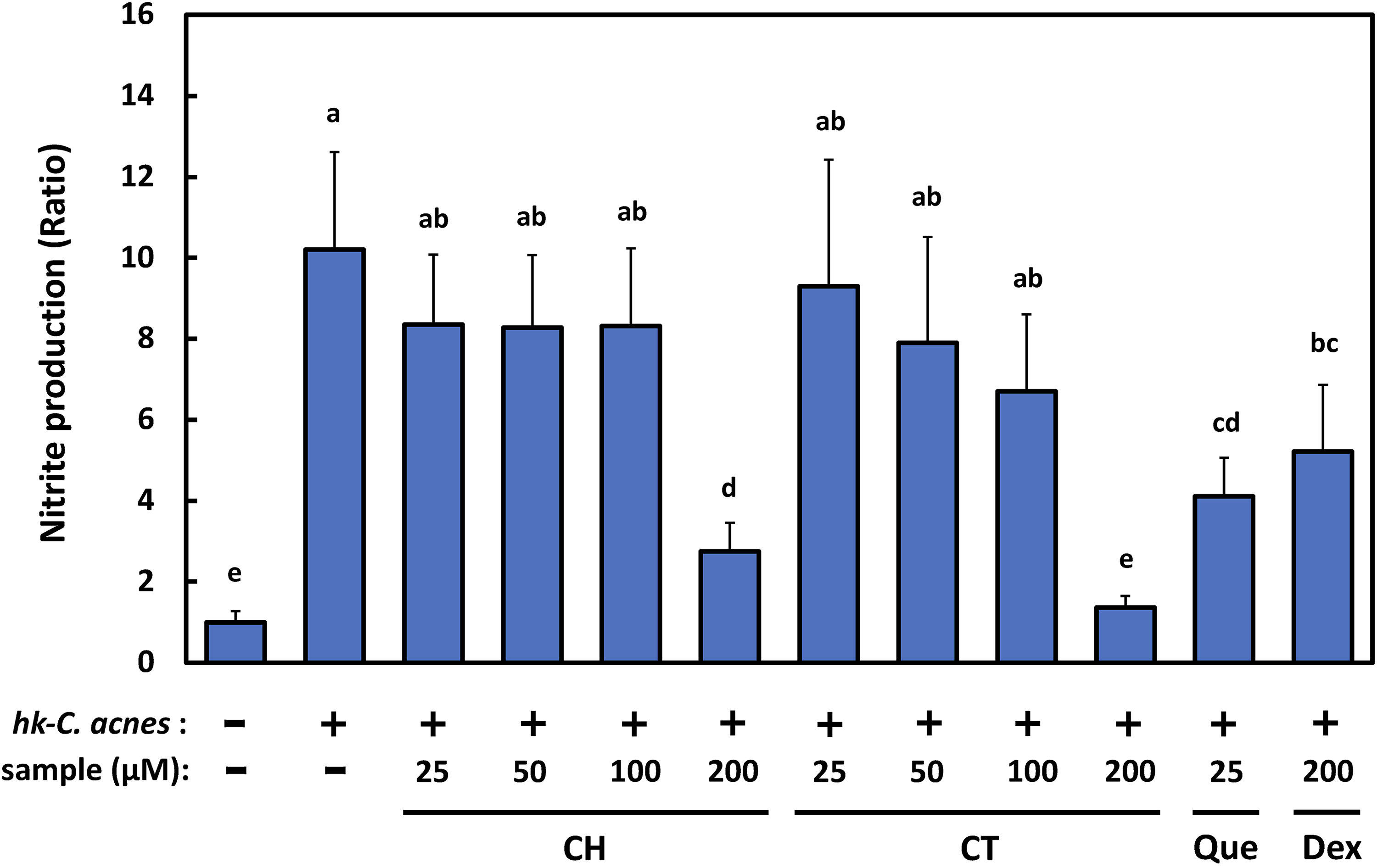
CH and CT inhibit nitric oxide (NO) production. RAW264.7 cells were pretreated with CH or CT (25, 50, 100, and 200 μM) for 2 h. Cells were then stimulated either with or without heat-killed Cutibacterium acnes (hk-C. acnes) (200 μg/mL) for 24 h. Cell culture media were harvested and levels of nitrite, a stable metabolite of NO, measured.
We further examined the molecular mechanisms underlying the inhibitory effects of CH and CT on hk-C. acnes-stimulated NO production in murine macrophage RAW264.7 cells. We initially examined the protein and mRNA expression levels of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2), which function downstream of TLR1/2 or TLR2/6 signaling. As shown in Figure 4A, the expression level of iNOS was approximately 600-fold higher upon stimulation with hk-C. acnes than that in untreated cells, and this level was decreased by approximately 60% to 65% by either CH or CT treatment. The expression of COX-2 was upregulated approximately 140-fold by stimulation with hk-C. acnes compared to the level in untreated cells, and the expression level also decreased by approximately 35% to 40% by either CH or CT treatment (Figure 4A). Since CH and CT decreased iNOS and COX-2 expression levels, we further analyzed protein expression levels by Western blotting. The expression levels of both proteins were significantly lower in RAW264.7 cells stimulated with hk-C acnes after CH and CT treatments than in cells stimulated with hk-C. acnes (Figure 4B ). The expression levels of tumor necrosis factor-α (TNF-α) and interleukin 6 (IL-6), which encode pro-inflammatory cytokines, were also examined. The expression levels of IL-6 in RAW264.7 cells stimulated with hk-C. acnes after CH and CT treatments were approximately 56% and 44% lower, respectively, than those in cells stimulated with hk-C. acnes. The expression levels of TNF-α in the cells treated with CH were not significantly different, although the values were reduced compared with those in control cells (Figure 5). These results suggest that the suppression of the mRNA and protein expression of iNOS and COX-2 and suppression of the mRNA expression of inflammatory cytokines (TNF-α and IL-6) are deeply involved in the anti-inflammatory effects of CH and CT treatments of RAW264.7 cells stimulated with hk-C. acnes. HO-1, also called heat shock protein-32, is a 32-kDa heme degrading enzyme. HO-1 can be induced by various stressors, such as heavy metals and ROS. Heme is enzymatically degraded by HO-1, yielding breakdown products, such as carbon monoxide (CO), ferrous iron, biliverdin, and bilirubin. These by-products could contribute to HO-1's anti-inflammatory effects. 28 HO-1, which is induced by natural products, such as quercetin, suppresses lipopolysaccharide (LPS)-induced inflammation in RAW264.7 cells; accordingly, we next investigated whether CH and CT treatment of RAW264.7 cells induces HO-1 mRNA and protein expression (Figure 6). CH and CT treatment increased HO-1 expression levels slightly but did not affect protein expression levels. These results suggested that the anti-inflammatory effects of both compounds are not mediated by HO-1 expression but by attenuating intracellular signaling activated by the recognition of hk-C. acnes by TLR 1/2 or TLR2/6 on the macrophage membrane surface. Components of Gram-positive microorganism cell membranes, such as peptidoglycan, lipoproteins, and diacylated lipopeptides stimulate NO production through the canonical NF-κB pathway after being recognized by TLR2 and TLR1 or TLR6 heterodimer receptors, respectively, primarily on the cell surface of macrophages. 29 As shown in Figure 1, CH and CT have a phenylpropanoid portion (caffeic acid portion) in their structure, with well-known inhibitory effects on NF-κB. Therefore, we verified that NF-κB involved in TLR 1/2 and 2/6 signaling, which is activated by hk-C. acnes stimulation, is inhibited by CH and CT. We further examined the transcriptional activity of NF-κB, a downstream molecule of TLR 1/2 and 2/6 signaling using a pGL4.32 [luc2P/NF-κB response element [NF-κB-RE]/Hygro] plasmid vector. As shown in Figure 6A and 6B, NF-κB transactivation was increased 8.7-fold by hk-C acnes stimulation, while both CH and CT reduced hk-C acnes-stimulated NF-κB transactivation by approximately 41% to 56% (Figure 7). Moreover, ROS facilitate NF-κB and mitogen-activated protein kinase (MAPK) activity in LPS-stimulated RAW264.7 cells. Both CH and CT showed anti-oxidative effects in our previous study, and, thus, may contribute to the anti-inflammatory effects.

CH and CT downregulate the expression levels of inflammation-related genes and proteins in RAW264.7 cells induced by heat-killed Cutibacterium acnes (hk-C. acnes). (A) The total RNA of CH- or CT-treated RAW264.7 cells (200 μM) was harvested and subjected to qRT-PCR for inducible NO synthase (iNOS) and cyclooxygenase 2 (COX2) (mean ± standard error [SE], n = 6). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal control. Student's t-test were used for statistical analyses (#P < 0.001, vs untreated cells, *P < 0.05, **P < 0.01, vs the cells treated with hk-C. acnes). (B) Western blot analysis of lysates of cells treated with either CH or CT (200 μM) using antibodies specific for iNOS, COX2, and β-actin (control). A representative blot from 3 independent experiments is shown.

CH and CT downregulate proinflammatory cytokine genes (TNF-α and IL-6) in heat-killed Cutibacterium acnes (hk-C. acnes)-stimulated RAW264.7 cells. The total RNA of CH- or CT-treated RAW264.7 cells (200 μM) was harvested and subjected to qRT-PCR for TNF-α and IL-12. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal control.

CH and CT pretreatment did not induce HO-1 gene and protein expression in RAW264.7 cells. (A) HO-1 gene expression and (B) HO-1 protein expression. After preincubation with 200 μM CH or CT for 2 h, cell lysates were harvested and subjected to qRT-PCR and Western blot analyses to detect HO-1 expression at the mRNA and protein level, respectively. A representative blot from 3 independent experiments is shown.

Effects of treatment with CH and CT on NF-κB transcriptional activity in RAW264.7 cells treated with heat-killed Cutibacterium acnes (hk-C. acnes). RAW264.7 cells transfected with the NF-κB firefly luciferase reporter vector and Renilla luciferase control vector were cultured with either CH or CT (200 μM) for 2 h. NF-κB enhancer activity was evaluated by a luciferase assay. Firefly luciferase activity was normalized to the Renilla luciferase activity. NF-κB enhancer activity is expressed relative to activity in hk-C. acnes-unstimulated RAW264.7 cells and are presented as mean ± standard error (SE) (n = 6). Ammonium pyrrolidinedithiocarbamate (APDC) was used as a positive control.
In general, macrophages are polarized into 2 types (M1 and M2), in response to extracellular microevents.30‐33 M1 macrophages are activated during bacterial, viral and fungal infections and produce various pro-inflammatory cytokines (ie, TNF-α, IL-6, and IL-12), NO, and reactive oxygen intermediates. Simultaneously, they express cluster of differentiation (CD) 86 to eliminate these pathogens. Conversely, certain cytokines, such as IL-4, IL-10, or IL-13, can induce M2 macrophage polarization. M2 macrophages are alternatively activated by exposure to these cytokines and secrete anti-inflammatory factors, such as arginase-1 (Arg-1) and IL-10, accompanied by increased expression of either the scavenger receptor CD163 or mannose receptor CD206, which alleviates the inflammatory events. We finally examined the expression of M1 and M2 macrophage biomarkers in RAW264.7 cells treated with CH or CT, respectively. CD80 and CD86, which are M1 macrophage cell surface markers, were significantly decreased by CH and CT treatment. However, the expression of both CD206, an M2 macrophage cell surface marker, and Arg-1, an M2 macrophage intracellular marker, was not detected (Figure 8). The expression of iNOS, an M1 macrophage intracellular marker, was also decreased, suggesting that CH and CT treatment attenuates M1 macrophage activation.
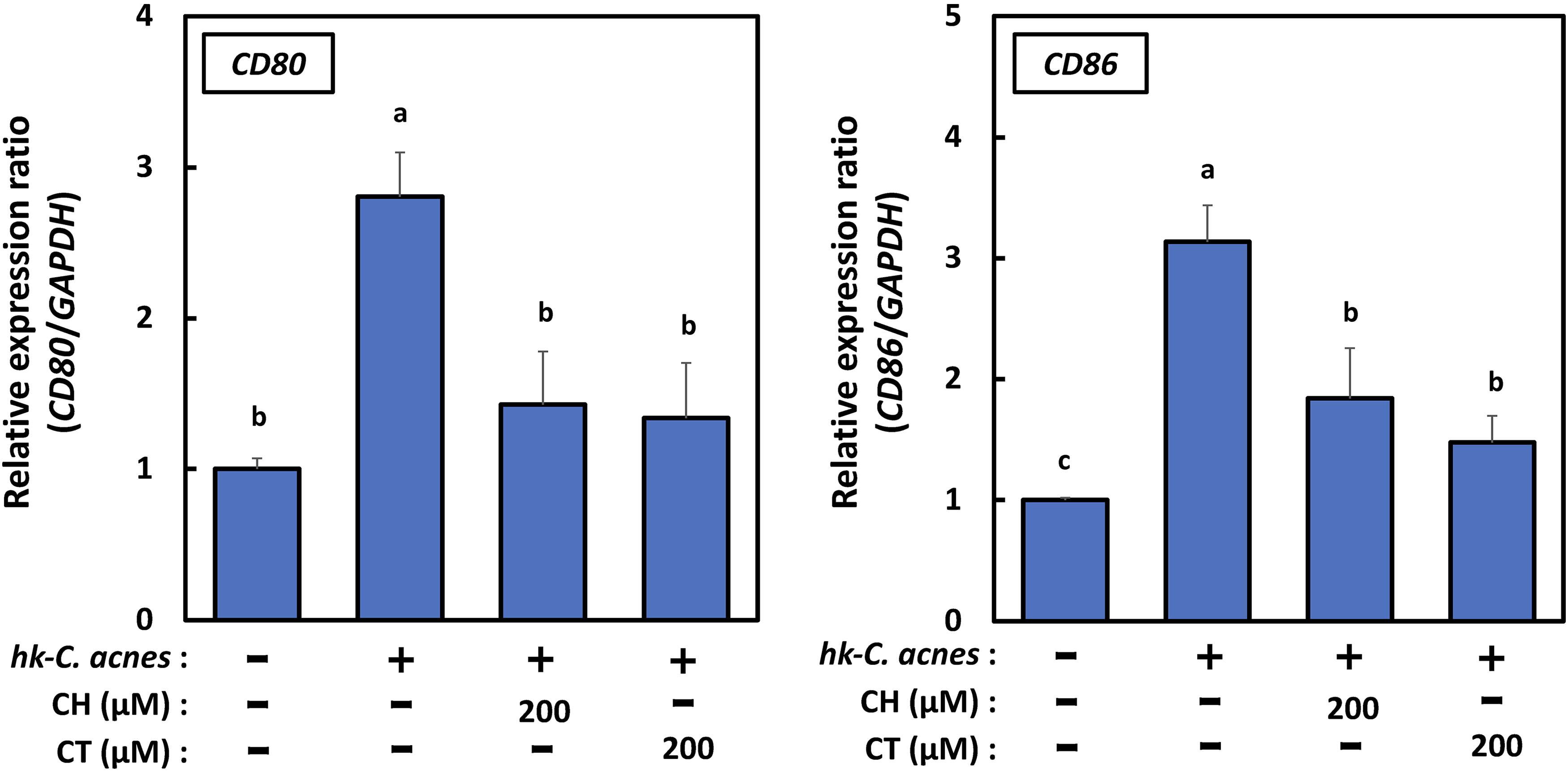
CH and CT downregulate the expression levels of M1 macrophage biomarkers in RAW264.7 cells induced by heat-killed Cutibacterium acnes (hk-C. acnes). The total RNA of CH- or CT-treated RAW264.7 cells (200 μM) was harvested and subjected to qRT-PCR for CD80 and CD86 (mean ± standard error [SE] n = 6). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal control.
In the growth stage of the young watermelon fruits used in our previous study, the fertilized ovule, which is the source of seeds, is enlarged and is soft in the early stage of seed development. As the seed coat is formed, lignin, a constituent of the seed coat, accumulates, and caffeic acid is metabolically consumed to synthesize coniferyl alcohol, which is a constituent of lignin. Zhang et al. isolated lanatusosides from ripening watermelon seeds, in which caffeic acid was removed from citrullusides by hydrolysis, and it is thought that citrullusides are also metabolized to remove caffeic acid, which is reduced in ripening fruits. 34 Therefore, young watermelon fruit with a high content of citrullusides may be useful candidate ingredients for the development of natural skin care products for acne vulgaris.
In conclusion
Materials and Methods
Reagent
Gifu anaerobic medium (GAM) and GAM agar powder were purchased from Nissui Pharmaceutical Co. Ltd, benzal chloride (BZK) from WAKO Pure Chemical Co. Ltd (Osaka, Japan), antibodies to COX-2 (#4842), iNOS, (#2982), and β-actin (#4967) from Cell Signaling Technology, Inc., and fetal bovine serum (FBS) from Life Technologies Corp.
Maintenance of C. acnes
C. acnes (NBRC 107605) was purchased from the Biological Resource Center, National Institute of Technology and Evaluation (NBRC, Japan Kisarazu) and was cultured in GAM under anaerobic conditions using AnaeroPack (Mitsubishi Gas Chemical Co. Inc.) at 37 °C. The culture was incubated until the OD600 reached around 2.0 (logarithmic growth phase) and was then diluted 10 times with GAM (OD600 = 0.2 corresponding to approximately 1 × 106 CFU/mL) for disk diffusion and turbidimetry assays.
Disk Diffusion Assay
The inhibition of C. acnes growth by the test compounds was evaluated using the disk diffusion method. The GAM agar plate was swabbed with a C. acnes suspension (500 µL) and kept at room temperature (22 ± 2 °C) for 30 min under aseptic conditions. Sterilized 6-mm filter paper disks (Advantec Toyo Kaisha, Ltd) were impregnated with 15 µL of test compound solutions (6, 30, and 60 µg/mL) and then placed on the surface of the inoculated GAM agar plates. The plates were incubated under anaerobic conditions at 37 °C for 48 h. BZK (1.25%, 2.5%, 5%, and 10%), a well-known bactericide, was used as a positive control. After incubation for 48 h, microbial zones of inhibition were measured in mm. Each experiment was repeated 5 times.
Turbidimetry Assay
We further examined the inhibitory effects of test compounds using a turbidimetry assay. Each test compound was dissolved in dimethyl sulfoxide (DMSO) and then added to the C acnes suspension (final concentration of DMSO, < 0.3%, which had no significant effect on the growth of C. acnes). C. acnes was grown in either the absence or presence of 25, 50, and 100 µM test samples at 37 °C for 24, 48, and 72 h under anaerobic conditions. After incubation for the indicated time, 200 µL of culture media was transferred to the well of a 96-well plate and absorbance at 600 nm was measured immediately using a microplate reader to estimate C. acnes growth (Varioscan LuxTM, Thermo Fisher Scientific Inc.).
Cell Culture and Treatment
Murine macrophage RAW264.7 cells (RCB 0535) were purchased from the RIKEN Bio Resource Center (Tsukuba, Japan). Cells were cultured in Dulbecco's modified Eagle's medium (DMEM) containing 10% FBS (Life Technologies Corp.), 100 U/mL of penicillin, and 100 µg/mL streptomycin, in a 5% CO2 humidified atmosphere at 37 °C.
Preparation of hk-C acnes Powder and Setting of Concentrations for hk-C acnes-Induced NO Production in Cellular Systems
C. acnes was cultured in GAM at 37 °C for 48 h. C. acnes cells were then harvested by centrifugation at 3000 × g for 20 min and washed twice with sterilized phosphate-buffered saline solution (PBS; 0.85% NaCl, 2.86 mM KCl, 10 mM Na2HPO4, and 1.76 mM KH2PO4 at pH 7.7). After washing, cells were heat-killed at 121 °C for 15 min in an autoclave and then powdered by freeze-drying. The hk-C.acnes powder was suspended in PBS to a concentration of 200 mg/mL. To set the concentration for hk-C. acnes-induced NO production in RAW264 .7 cells, cells (2 × 105 cells/well) were seeded in 24-well multiplates and cultured for 12 h. After incubation for 12 h, the cells were stimulated with the hk-C acnes suspension (200 µg/mL) for 24 h.
Cell Treatment and Measurement of NO Production
RAW264.7 cells (2 × 105 cells/well) were seeded in 24-well multiplates and cultured for 12 h. After incubation for 12 h, the cells were pretreated with either CH or CT (25, 50, 100, or 200 µM) for 2 h. The cells were then stimulated with either vehicle or 200 µg/mL hk C. acnes. Cell culture media were collected and then centrifuged at 1500 × g at 4 °C for 5 min. A Griess Reagent Kit (Promega) was used to measure the amount of nitrite, a stable metabolite of NO, in the supernatants. Briefly, 50 μL of each culture medium was added to 96-well plates in triplicate and the same volume of a sulfanilamide solution was dispensed. After incubation at room temperature for 10 min, 50 μL of an N-1-napthylethylenediamine dihydrochloride solution was added to all wells. After incubation at room temperature for 10 min, the absorbance was measured at 540 nm using a colorimetric microplate reader (Varioscan LuxTM, Thermo Fisher Scientific Inc.).
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
After hk-C. acnes stimulation for 24 h, total RNA was extracted from the cells by TRIzol reagent containing phenol/guanidine isothiocyanate (Thermo Fisher Scientific Inc.) with DNase I treatment. Complementary DNA (cDNA) was then synthesized using reverse transcriptase with 12.5 ng of total RNA. To determine the mRNA levels of Arg-1, CD80, CD86, CD206, COX-2, HO-1, iNOS, IL-6, and TNFα, cDNA was prepared from total RNA samples using a High-Capacity RNA-to-cDNA Kit (Thermo Fisher Scientific Inc.). Subsequently, qRT-PCR was performed using the SYBR™ Green PCR Master Mix Kit (Thermo Fisher Scientific Inc.) and the following primer pairs (Takara Bio Inc.): Arg-1 forward: 5′-AGCTCTGGGAATCTGCATGG-3′, reverse:5′-ATGTACACGATGTCTTTGGCAGATA-3′; CD80 forward: 5′-AAGTTTCCATGTCCAAGGCTCA-3′, reverse:5′-TGTAACGGCAAGGCAGCAATA-3′; CD86 forward: 5′-CCATTTCAGTGCTCTTGGCCTA-3′, reverse:5′-TTTGGGCAATGTCAATTGTGTC-3′; CD206, forward: 5′-GATAGGCATGTTCCGAAATGTTGA-3′, reverse:5′-TATTCCAAAGGCCCGAAGATGA-3′; COX-2, forward: 5′-CTGGAACATGGACTCACTCAGTTTG-3′, reverse:5′-AGGCCTTTGCCACTGCTTGTA-3′; glyceraldehyde-3-phosphate dehydrogenase (GAPDH), forward: 5′-AAATGGTGAAGGTCGGTGTGAAC-3′, reverse:5′-CAACAATCTCCACTTTGCCACTG-3′; HO-1 forward: 5′-TGCAGGTGATGCTGACAGAGC-3′, reverse:5′-GGGATGAGCTAGTGCTGATCTGG-3′; iNOS forward: 5′-TAGGCAGAGATTGGAGGCCTTG-3′, reverse:5′-GGGTTGTTGCTGAATTCCAGTC-3′; IL-6, forward: 5′-CAACCATGATGCACTTGCAGA-3′, reverse:5′-CTCCAGGTAGCTATGGTACTCCAGA-3′; TNFα forward: 5′-AAGCCTGTAGCCCACGTCGTA-3′, reverse:5′-GGCACCACTAGTTGGTTGTCTTTG-3′. The expression level of each gene was determined using the ΔΔCt method and normalized to the level of GAPDH, used as the internal control. PCR consisted of 45 cycles of 95 °C for 15 s and 60 °C for 60 s, after an initial denaturation step (95 °C for 10 min).
Western Blotting
Whole cell extracts were prepared by lysing cells in radioimmunoprecipitation buffer containing complete protease inhibitor and a phosphatase inhibitor cocktail (Roche). Samples were subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis and electroblotted onto polyvinylidene difluoride membranes (GE Healthcare). After blocking nonspecific binding for 1 h with 5% nonfat milk in TBST (tris-buffered saline containing 0.1% Tween 20), the membrane was incubated overnight at 4 °C with the various primary antibodies. The membrane was then washed 3 times in TBST, incubated with a horseradish peroxidase-conjugated secondary antibody at room temperature, and washed again 3 times in TBST. Protein bands were detected using an Enhanced Chemiluminescence Kit (GE Healthcare) and chemiluminescence detector (iBright FL1500 Imaging System, Thermo Fisher Scientific Inc.).
Transfection and Luciferase Assay
The pGL4.32[luc2P/NF-κB-RE/Hygro] vector contained 5 copies of a NF-κB-RE that drives the transcription of the luciferase reporter gene luc2P (Photinus pyralis). The firefly luciferase gene, NF-κB-RE (500 ng, Promega), and pRLSV40 (10 ng, Promega) were co-transfected into cells in 24-well plates using Lipofectamine 2000 (Invitrogen). The luciferase activity of each sample was measured 6 h after treatment with CH or CT at the indicated concentrations using a Dual-Glo Luciferase Assay System (Promega).
Statistical Analyses
All data were analyzed using the Mac statistical analysis software package for Macintosh, version 2.0 (Esumi Co.). All data are expressed as means ± standard error (SE). Comparisons among groups were performed using either the Tukey–Kramer test (for many groups) or Student's t-test (for 2 groups). Values of P < .05 were considered statistically significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Data Availability Statement
The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request.
