Abstract
Quorum sensing (QS) is an important microbial signaling system that controls the expression of many virulence genes. Combination treatment with QS inhibitors and antibiotics may be the new treatment option for infectious diseases in the future. Therefore, there is an increasing need to discover anti-QS compounds to prevent antimicrobial resistance. We isolated methyl-β-D-glucopyranoside from Scabiosa comosa and found that it exerts an anti-QS activity against Chromobacterium violaceum CV026 without exhibiting any antimicrobial activity.
Introduction
The increase in microbial resistance to antibiotics is a major concern to public health as it reduces the effectiveness of treatments and increases morbidity and mortality. 1 Biofilm formation is one of the mechanisms bacteria use to develop drug resistance. 2 Biofilms are difficult to disrupt, leading to treatment failure and disease recurrence. Although efforts to develop novel antibiotics continue, generating alternative approaches to treat bacterial infections is crucial.
Bacteria carry out many physiological functions, such as antibiotic production, biofilm formation, cell division, sporulation, conjugation, bioluminescence, secretion of virulence factors, primary metabolism, and secretion of specific extracellular signaling molecules called autoinducers. The mechanism of secretion of autoinducers to communicate with each other is called quorum sensing (QS) and allows bacteria to function as multicellular organisms. QS has attracted attention as a promising anti-pathogenic drug target because many pathogenic bacteria employ QS to regulate their pathogenicity. Combination treatment with QS inhibitors and antibiotics may be the new treatment option for infectious diseases in the future. Therefore, there is an increasing need to discover anti-quorum sensing (anti-QS) compounds to prevent antimicrobial resistance.
There are several ways to search for QS inhibitors. In this study, we used Chromobacterium violaceum CV026, which does not produce an acyl homoserine lactone (AHL) autoinducer. Chromobacterium violaceum CV026 lacks the AHL synthase gene and produces the purple pigment violacein, whose production is regulated by QS when AHL is added. 3 Chromobacterium violaceum CV026 was created to search for autoinducers, but it can also be used to search for compounds that inhibit QS by exogenously adding AHL.
Scabiosa comosa Fisch. ex Roem. & Schult. belongs to the Dipsacaceae family and is distributed throughout Khuvsgul, Khentii, Khangai, Mongol Daguur, Khyangan, Dund Khalkh, and Dornod Mongolia. In Mongolian and Tibetan traditional medicine, the inflorescence of S comosa is used to treat inflammatory diseases of the lungs and gastrointestinal tract. Aerial parts are used to treat sepsis. 4 Chemical and pharmacological studies have been conducted on S comosa; however, its chemical properties have not been studied extensively. It was recently reported that chlorogenic acid and 3,5-dicaffeoylquinic acid isolated from the flowers of S comosa had excellent radical scavenging and anti-hepatitis C virus activities. 5 In that study, 3,4-dicaffeoylquinic acid, 3,5-dicaffeoylquinic acid, and 4,5-dicaffeoylquinic acid were identified for the first time from the genus Scabiosa. Moreover, the flower of S comosa is included as a component of Gurigumu-7 medicine to treat liver diseases in Mongolian and Tibetan traditional medicines. It was revealed that the methanol extract of Gurigumu-7 had more hepatoprotective activities than Gurigumu-7. The methanol extract is rich in ellagic acid, luteolin, apigenin glucoside, ursolic acid glycosides, bidenoside, and proanthocyanidins. 6
To the best of our knowledge, our study is the first to report that an active compound isolated from S comosa can inhibit purple pigment production by C violaceum.
Materials and Methods
General Experimental Procedures
Aerial parts of S comosa were collected during flowering in July 2015 from Tuv Province, Mongolia, and identified by Dr Shagdar Dariimaa, Mongolian State University of Education, Ulaanbaatar, Mongolia. The C violaceum CV026 strain was used to evaluate QS inhibition activity. 7 1H-NMR spectra of the isolated compounds in CD3OD-d4 were recorded on a JEOL-500 NMR spectrometer (500 MHz, JEOL) using the solvent as an internal reference. 13C-NMR spectra were recorded at 125 MHz. Optical rotations were measured using a digital polarimeter (P-2200; JASCO).
Reagents
Silica gel (64-200 µm), solvents, methyl-α-D-glucopyranoside, and methyl-β-D-glucopyranoside were purchased from the Wako Pure Chemical Corporation. N-hexanoyl-l-homoserine lactone (N-HHL) was obtained from Santa Cruz Biotechnology Inc. Preparative thin-layer chromatography (TLC) plates (silica gel 60 F254, 20 × 20 cm, 1 mm) were purchased from Merck Co.
Quorum-Sensing Inhibitory Assay and Antimicrobial Activity Test
Chromobacterium violaceum CV026 was cultured in Luria-Bertani (LB) broth (tryptone, 1%; yeast extract, 0.5%; and NaCl, 0.5%, pH7.2) at 27 °C for 20 h. For the QS inhibitory assay, culture broth (100 µL) was mixed with soft-LB agar (LB + 0.8% agar) in a plate (9 cm dish) containing N-HHL (0.1 µM). For the antimicrobial activity test, the culture broth was mixed with soft-LB agar without N-HHL. For the checkerboard assay, 0.03 to 1 µM was added to soft-LB agar. Paper disks (8 mm diameter, Advantec) containing various amounts of sample were placed on the soft-LB agar plate and incubated at 27 °C for 24 h. The anti-QS activity was expressed as the diameter (mm) of the zone of pigment production inhibition on the soft-LB agar plate, and antimicrobial activity was expressed as the diameter of the growth inhibition zone.
Results
Isolation of Quorum-Sensing Inhibitor
The aerial parts of S comosa (700 g) were separated into flowers, leaves, and stems. The parts were then extracted with methanol three times. The extracts were dried in a vacuum and 56.5 g of flower, 64.5 g of leaf, and 50.8 g of stem extracts were obtained. The active compound

Structure of methyl-β-D-glucopyranoside (A) and methyl-α-D-glucopyranoside (B).
Anti-Quorum-Sensing Activity
The anti-QS activity of methyl-β-D-glucopyranoside isolated from S comosa was tested using the paper-disk method. The size of the region where purple pigment production was suppressed increased in a concentration-dependent manner with methyl-β-D-glucopyranoside (3-100 µg/disk) (Figure 2, Figures S3 and S4). Moreover, methyl-β-D-glucopyranoside did not exhibit antimicrobial activity against C violaceum CV026 under the same conditions (Figure S5). On the other hand, the purchased methyl-α-D-glucopyranoside (Figure 1B) showed no anti-QS activity at a concentration of 100 µg/disk (Figure S3).

The anti-QS activity of compound
A checkerboard assay was performed to determine whether methyl-β-D-glucopyranoside competed with N-hexanoyl homoserine lactone (N-HHL). In this test, methyl-β-D-glucopyranoside was used instead of compound
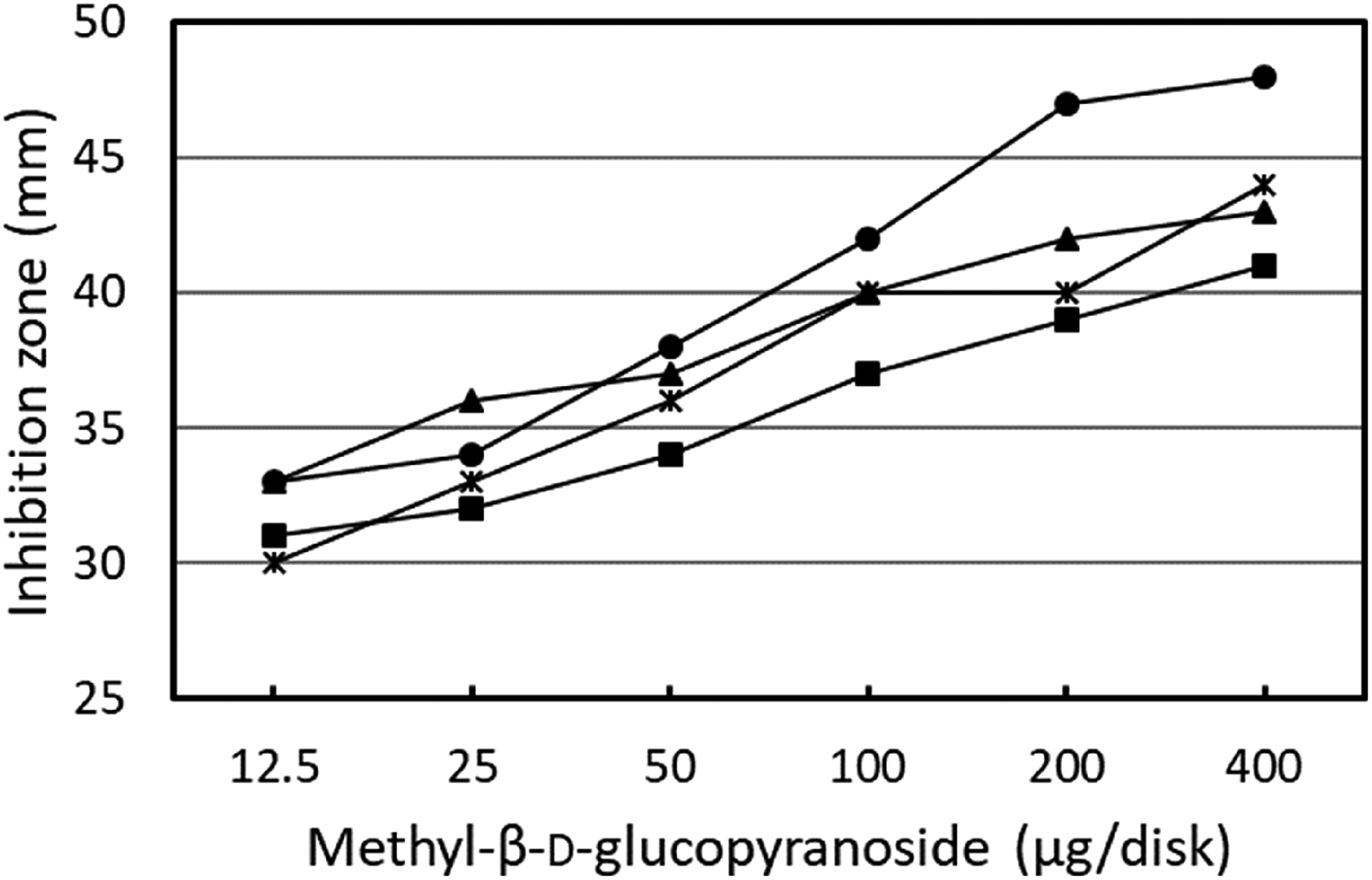
Checkerboard assay for assessing the competition between QS inducer N-HHL and methyl-β-D-glucopyranoside. The diameter of the pigment inhibition zone (mm) was plotted as the concentration of methyl-β-D-glucopyranoside for each N-HHL concentration (circle; 0.03 µM, triangle; 0.1 µM, asterisk; 0.33 µM, and square; 1 µM). Methyl-β-D-glucopyranoside was used as the substrate.
Discussion
In this study, methyl-β-D-glucopyranoside, isolated from S comosa metabolites as a QS inhibitor, exhibited no antimicrobial activity. Methyl-β-D-glucopyranoside was previously isolated from many plants, 10 but not from S comosa.
Methyl-β-D-glucopyranoside inhibited the purple pigment production of C violaceum CV026, but inhibition of pigment production was not observed with methyl-α-D-glucopyranoside. The steric configuration of the anomeric position is, therefore, important for inhibiting pigment production. Among homocysteine thiolactones synthesized as autoinducer analogs, those in L-configuration showed agonistic activity against quorum sensing in Pseudomonas aeruginosa and C violaceum CV026, whereas those in D-configuration showed no effects. 11 Therefore, the steric structure of the QS inhibitor is also an important factor in determining its effectiveness. Since the competition between methyl-β-D-glucopyranoside and N-HHL was very weak, the function of CviR, which is a transcriptional regulator of the QS system in C violaceum, may have been partially inhibited. Other transcriptional regulators, such as LasR and QscR of P aeruginosa and TraR of Agrobacterium tumefaciens, require AHLs with longer acyl-side chains than those required by CviR. 12 The inhibitory activities of methyl-β-D-glucopyranoside will be evaluated in other QS systems to explore the range of its anti-QS activity.
Methyl-β-D-glucopyranoside has been reported as an antitumor agent with low cytotoxicity. 13 Several compounds have been reported to inhibit QS, including patulin from Penicillium 14 and halogenated furanones from the marine alga Delisea pulchra. 15 Some pharmaceutical agents such as azithromycin 16 and chloramphenicol 17 are also known to possess QS inhibitory activity. In addition, many plants have been reported to produce compounds that inhibit microbial virulence factors. 18 However, to date, there have been no reports on the QS inhibitory activities of monosaccharide derivatives. Since there are many natural and synthetic monosaccharides, it should be possible to obtain compounds with high activity and useful anti-QS activity by exploring monosaccharides. Monosaccharides that inhibit the QS system without affecting bacterial growth are expected to facilitate new bacterial infection therapies with conventional antimicrobial agents while suppressing bacterial virulence.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221139972 - Supplemental material for Methyl-β-D-glucopyranoside From Scabiosa comosa as a Quorum-Sensing Inhibitor
Supplemental material, sj-docx-1-npx-10.1177_1934578X221139972 for Methyl-β-D-glucopyranoside From Scabiosa comosa as a Quorum-Sensing Inhibitor by Bekh-Ochir Davaapurev, Atsushi Fukumoto, Byambajav Bolortuya, Delgerbat Boldbaatar, Yoshino Maehata, Yohei Iizaka, Fumio Kato, Javzan Batkhuu and Yojiro Anzai in Natural Product Communications
Footnotes
Acknowledgments
We thank Dr. Tsukasa Ikeda at Utsunomiya University for providing us Chromobacterium violaceum CV026. This work was conducted within the framework of the project Anti-QS Activity of Scabiosa comosa Fisch. ex Roem. & Schult., supported by the Asia Research Center, Mongolia, and the Korea Foundation for Advanced Studies, Korea.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
