Abstract
Introduction
Renal cell carcinoma (RCC) is the third leading cancer of the urinary system, with a higher incidence in males than females. 1 Patients with RCC have a very high rate of developing metastatic diseases and even metastatic RCC after nephrectomy, accompanied with extremely poor prognosis. 2 There are many treatment options for RCC, and local or early RCC can be either partially or completely cured by surgery.3,4 However, despite the high rate of early diagnosis and cure of RCC, there are still a small number of patients with RCC who cannot avoid distant metastasis.5,6 It has been widely accepted that the invasion and metastasis of tumor cells is one of the main causes of death in patients with malignant tumors. 7 Likewise, the invasion and metastasis of RCC are the major factors that prevent RCC from being cured completely. Previous data showed that more than 90% of deaths of patients with RCC are metastasis-related. 8 Therefore, inhibiting the invasion and metastasis of RCC cells is of great significance for improving the survival of patients with RCC.
Epithelial–mesenchymal transition (EMT) is an important process for tumor cells to acquire the ability to migrate and invade. 9 Pathological EMT results in the loss of the ability of self-regulation, leading to the loss of epithelial cell polarity and intercellular connectivity and subsequently an increase in tumor migration and invasion characteristics. 10 According to a previous report, 11 the expression profile of EMT provides a target for the treatment of RCC and other cancers. In addition, the extracellular matrix (ECM) provides a microenvironment for cells, and the structural destruction of ECM is related to the invasion of cancer cells. 12 Matrix metalloproteinases (MMPs) participate in the degradation of ECM and play a key role in the process of EMT. It was reported that the expression of MMPs is regulated by EMT-related signal transduction pathways. 13 Therefore, inhibiting tumor invasion and metastasis by suppressing the EMT process has also become the focus of recent research.
Finding safe and efficient natural products for cancer treatment has always been a concern of researchers. It is worth mentioning that Traditional Chinese Medicine (TCM) has sound anti-tumor effects because of its multiple targets, satisfying efficacy, and few side effects. 14 Anti-tumor research of TCM is being and will be widely conducted in the future. Previous evidence indicated that TCM possesses solid therapeutic effects on the metastasis of breast cancer, lung cancer, and liver cancer, 15 providing a theoretical basis to study the effects of TCM on the metastasis of RCC.
Poria cocos F. A Wolf {Wolfiporia extensa (Peck) Ginns, family Polyporaceae} has a long medicinal history in China and is used to treat multi-system diseases. P. cocos has the advantages of wide source, low price, low toxicity, and few side effects compared with many synthetic drugs. Therefore, P. cocos has attracted more and more attention. In recent years, it was found that P. cocos has anti-tumor effects,16–18 but its specific anti-tumor constituents are not clear. Pachymic acid (PA), one of the important triterpenoid components of P. cocos, is a lanostene-8-ene triterpenoid (Figure 1A). Since PA is an important effective component of P. cocos, the present study focused on this compound and explored its influence on the adhesion and metastasis of RCC cells and the EMT process.

PA inhibits the proliferation of ACHN cells. (A) Chemical structure of PA. (B) Different concentrations of PA inhibited the growth of ACHN cells for 24 h. (C) Colony formation assay results on ACHN cells. (D) Photos of cell colonies after crystal violet staining. ****P < 0.0001.
Results
Effects of PA on the Viability of ACHN Cells
3-(4,5-Dimethylthiazole-2-yl)-2,5-diphenyltetrazole bromide (MTT) assay was used to detect the effect of PA on cell proliferation. The results showed that the ACHN cells treated with 20, 40, 80, and 160 µM PA for 24 h showed different degrees of growth inhibition, in a concentration-dependent manner (Figure 1B). The IC50 of PA to the ACHN cells at 24 h was 60.4 µM. To avoid the cytotoxicity of PA, only low concentrations (0, 20, 40, and 80 µM) of PA were selected for subsequent experiments. A colony formation experiment further showed that the proliferation of ACHN cells could be inhibited by PA (Figure 1C and D). This result laid a foundation for the study of the anti-RCC effect of PA.
Effects of PA on ACHN Cell Adhesion
Cell adhesion participates in cell proliferation and differentiation and is a regulator of cell movement. Cell adhesion is also a key step in tumor metastasis. The results of the cell adhesion experiment showed that the number of cells that adhered in the PA group was lower than that in the control group (Figure 2). This suggests that PA can effectively inhibit cell adhesion.
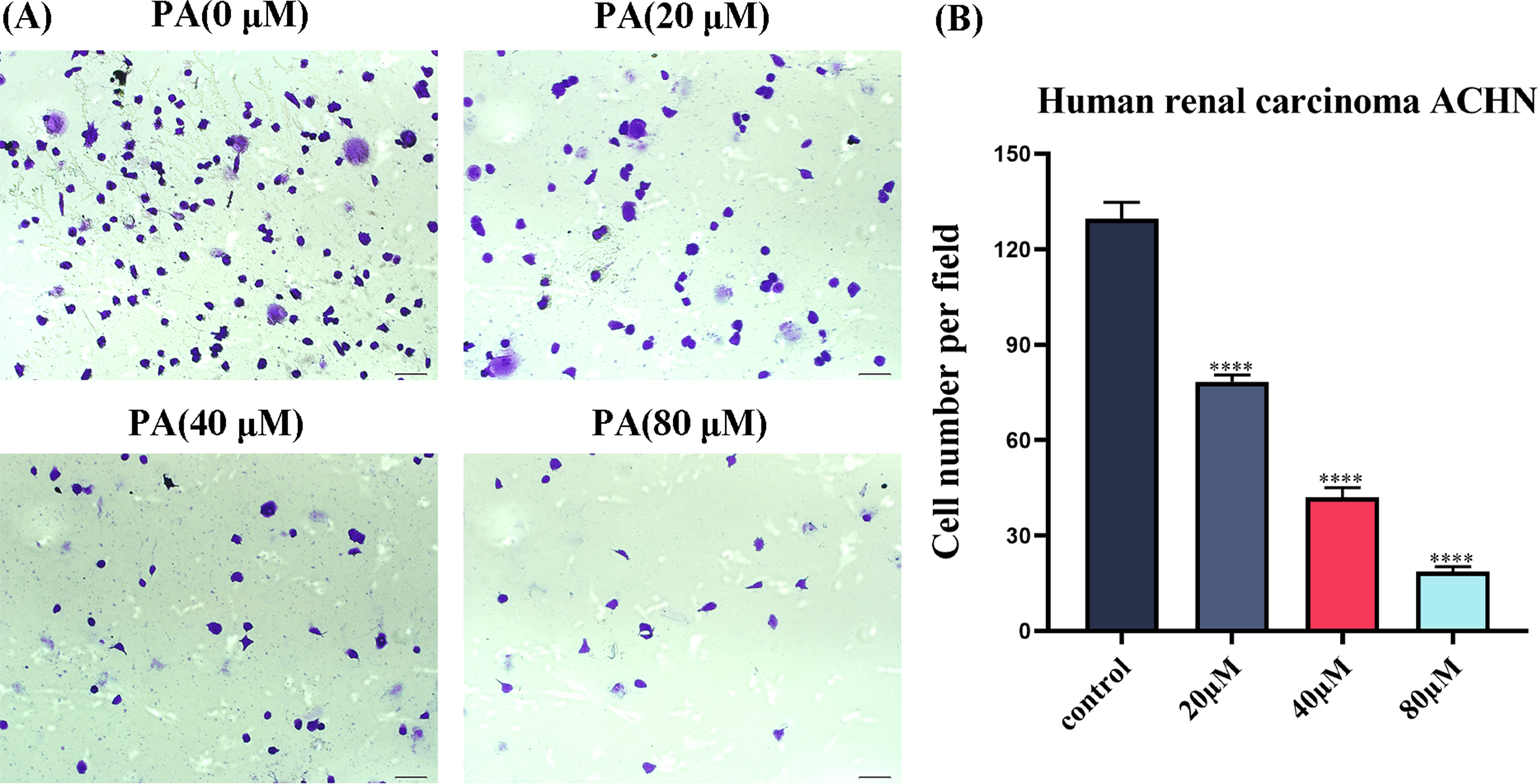
PA inhibits the adhesion ability of ACHN cells. (A) Microscopic image of cells stained with crystal violet. (B) Cell adhesion numbers of ACHN cells. All scales are 200 µm. ****P < 0.0001.
Effects of PA on the Metastasis of ACHN Cells
To explore the effect of PA on cell migration, we preliminarily verified whether PA had an inhibitory effect on cell migration through the wound healing assay. The results showed that when the wound areas of the ACHN cells were treated with PA at concentrations of 20, 40, and 80 µM for 24 h, and the healing ability of these cells decreased (Figure 3B). These results indicated that PA could inhibit the migration of ACHN cells.
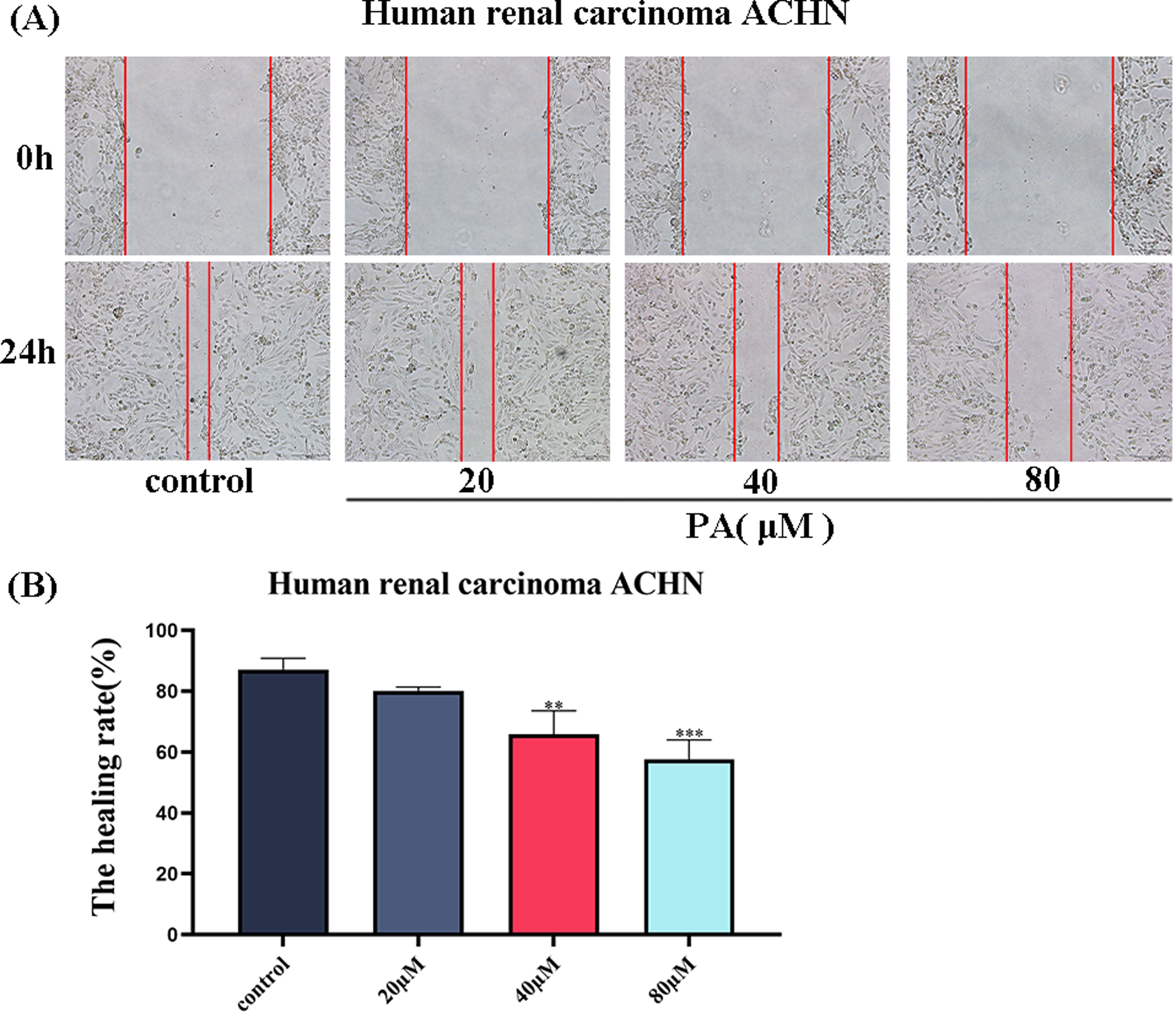
PA inhibits the migration of ACHN cells. (A) Microscopic photos of wound area of ACHN cells treated with PA. (B) Wound healing rate. All scales are 200 µm. **P < 0.01, ***P < 0.001.
Effects of PA on the Migration and Invasion of ACHN Cells
Transwell assay can also directly reflect the invasion ability of tumor cells. Therefore, we further verified the effect of PA on cell invasion ability by Transwell assay. The results showed that different concentrations of PA can effectively reduce the number of cells that migrate (Figure 4A and C). In addition, we employed the Transwell assay with Matrigel in a small chamber to detect the invasion ability of ACHN cells. The results showed that different concentrations of PA can effectively decrease the invasion cell number (Figure 4B and D). The above data together suggest that PA can effectively inhibit the invasion and migration of ACHN cells.

PA inhibits the migration and invasion of ACHN cells. (A) Microscopic photos of migrating cells after crystal violet staining. (B) Microscopic photos of invading cells after crystal violet staining. (C) Different concentrations of PA could inhibit cell migration. (D) Different concentrations of PA inhibited cell invasion. All scales are 200 µm. **P < 0.01, ***P < 0.001, ****P < 0.0001.
Effects of PA on the Dynamic Migration of ACHN Cells
To further examine how PA affects the migration state of ACHN cells, high-content imaging system analysis was performed to visualize cell movement. We plotted the movement trajectories of the cell groups based on the high-content data. The results showed a narrower distribution of trajectories of the ACHN cell populations after PA treatment, compared with that of the control group (Figure 5B).

PA inhibits the dynamic migration of ACHN cells. (A) After 12 h treatment with different concentrations of PA, the cell trajectory was dynamically tracked by a high-intensive imaging system for 12 h. (B) Each point corresponds to the displacement of a cell at a given time point.
Effects of PA on the Movement Ability of ACHN Cells
Next, the mean azimuth shift and cell displacement diagrams were drawn. With the increase in PA concentration, the migration trajectory of the cells was shortened and the migration ability was weakened (Figure 6A). In addition, the accumulated traveling distance of the cells was reduced by all PA administrations (Figure 6B), as were the average speed (Figure 6C) and the traveling distance of each cell (Figure 6D).

PA inhibits the dynamic migration of ACHN cells. (A) The mean square displacement of cells at each time point. (B) Accumulated distance (µm) per track. (C) Average speed (µm) per track. (D) Displacement (µm) per track.
Effects of PA on the Expression of EMT-Related Proteins and MMP-Related Proteins in ACHN Cells
EMT is closely related to tumor metastasis and has become a hotspot of current research. Detecting EMT-related markers has also become an important means of determining tumor invasion and metastasis. Therefore, we attempted to further explore the effects of PA on EMT-related proteins at the molecular level. Western blot results showed that the PA administrations upregulated the expression of epithelial marker E-cadherin and downregulated the expression of mesenchymal markers Vimentin and N-cadherin (Figure 7A). Furthermore, EMT-related transcription factors can induce MMP expression. Western blot detection revealed that the PA administrations downregulated the expression of MMP-related proteins in the ACHN cells, including MMP-9, MMP-2, and tissue inhibitor of metalloproteinase-1 (TIMP-1) (Figure 7B). These results suggest that PA can regulate the expression of key MMP proteins of EMT-related proteins in the ACHN cells.

PA affects the expression of EMT-related proteins MMPs key proteins in ACHN cells. (A) The expression of EMT-related proteins was examined by Western blots. (B) The expression of MMP-related proteins was determined by Western blots; β-actin was the endogenous reference. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Discussion
RCC accounts for 90% of renal malignant tumors of the urinary system, with increasing incidence and mortality when it is associated with nephropathy. 19 RCC tends to metastasize and spread in the middle and late stages, and metastasis is a common feature of RCC. When tumor cells metastasize, the diseases are almost incurable, and nearly 90% of cancer deaths are caused by tumor metastasis.20,21 Therefore, inhibiting tumor invasion and metastasis is one of the most important and effective approaches to treat RCC. In the treatment of RCC, Western medicine treatment can induce strong toxic side effects and drug resistance. TCM has been used for thousands of years and is now widely accepted as an alternative therapy for cancers. 22 A line of evidence indicates that TCM has obvious advantages in the prevention and treatment of RCC invasion and metastasis.23–25 Therefore, it is necessary to actively study valuable Chinese medicines to treat RCC.
P. cocos has a long medicinal history and is able to treat various diseases. 26 It is often used as a diuretic for the treatment of edema and urinary dysfunction. 27 An anti-tumor effect of P. cocos was found in recent years, which further expands the scope of research into this natural product. In this study, we explored the effects of PA on RCC cell metastasis and its underlying mechanisms. We first tested the effect of PA on cell survival by cell viability assay and found that PA could inhibit the activity of RCC cells. In addition, we proved by colony formation assay that PA can inhibit the proliferation of RCC cells. This preliminary evidence suggests that PA has an inhibitory effect on RCC cells. After that, we continued to explore the effects of PA on the invasion and metastasis of RCC cells and the possible underlying mechanisms. Cell adhesion plays an important role in maintaining the normal structure, growth, function, movement and metastasis of cells, and other physiological processes. 28 We found that PA decreased cell adhesion, which suggests that PA can inhibit cell adhesion at the onset of cell metastasis. Furthermore, cell migration is an important step and key link in inflammatory response and tumorigenesis. Some scholars have proposed that the inhibition of cell movement may be an effective way to suppress tumor metastasis. 29 The wound healing assay confirmed that PA can inhibit the migration of RCC cells. The Transwell assay also indicated that the invasion and metastasis of RCC cells could be inhibited by PA, which further confirmed the anti-tumor effect of PA. Next, we used a high-content real-time dynamic cell tracking system to record the movement status of the ACHN cells after the PA administrations, and the findings verified our hypothesis that PA can inhibit the dynamic migration of RCC cells. Based on these data, we believe that PA is able to suppress the invasion and metastasis of RCC.
To understand the molecular mechanism through which PA inhibits the invasion and metastasis of RCC, the effects of PA administrations on EMT were then examined. EMT occurs when cells lose their intercellular adhesion and polarity, which is critical for tumor cells to acquire the ability of invasion and metastasis.30,31 E-cadherin is considered to be a key factor that maintains stability and cell polarity between epithelial cells. 32 Once the expression of E-cadherin is decreased, the adhesion between epithelial cells is directly decreased, which promotes the invasion of epithelial tumor cells. 33 In the present study, we found that PA upregulated the expression of E-cadherin. This finding proves that PA can inhibit the metastasis of RCC by inhibiting the reduction of adhesion between epithelial cells. On the other hand, the acquisition of interstitial characteristics was accompanied by the increase of interstitial markers N-cadherin and Vimentin. 34 The downregulation of E-cadherin could be balanced by the increased expression of N-cadherin, 35 through which cells lose their association with epithelial cells and gain affinity for mesenchymal cells. 36 Vimentin is a typical marker of EMT occurrence, which can regulate a variety of cell types involved in cell migration 37 and enhance cell invasiveness. 38 In this study, we found that PA can downregulate both N-cadherin and Vimentin expression levels. These findings suggest that PA can inhibit the invasion and metastasis of RCC by inhibiting the EMT process. In addition to the above molecular markers, MMP-2 and MMP-9 are believed to be involved in the EMT process of RCC. 39 High expression of MMPs is an important feature of tumor cells escaping from the primary tumor and entering the secondary site. 40 Therefore, MMP-2 and MMP-9 can be used as key effectors for ECM remodeling and potential targets in anti-tumor therapies. In the current study, we found that PA inhibited the expression of MMP-2 and MMP-9. Therefore, it can be speculated that the mechanism of PA inhibiting the invasion and metastasis of RCC cells may be related to the downregulation of MMP key proteins.
Conclusion
The present study shows that PA, the main triterpenoid compound of P. cocos, has anti-RCC effects. We demonstrated that PA could effectively inhibit the adhesion and migration of RCC cells and this may be because PA can inhibit the EMT process and reduce the expression of MMP-related proteins in RCC cells. This study suggests that PA may be an effective active ingredient of P. cocos and can be developed as an anti-tumor drug. These data also provide an experimental basis for the application and research of the anti-tumor effects of P. cocos.
Materials and Methods
Cells
The human renal cell adenocarcinoma ACHN cell line was purchased from China Procell Life Science & Technology Co., Ltd (Cat.no. CL-0021).
Drugs
PA (standard substance, HPLC 97%) was purchased from Shanghai Yuanye Technology Co., Ltd, China (Cat.no. B20400). PA was dissolved in dimethyl sulfoxide (DMSO) (Sigma, Berlin, Germany, Cat.no. D2650) to a concentration of 10 mM and stored at −20 °C.
Reagents
Special culture medium for ACHN cells (Procell, Wuhan, China; Cat.no. CM-0021); phosphate buffered saline (PBS), trypsin (Beyotime Biotechnology, Shanghai, China; Cat.no. C0201, C0221A); FBS (Gibco, Waltham, MA, USA; Cat.no. 10099141); Transwell-permeable supports, 8.0 µm polycarbonate membranes, 96-well plates, 6-well plates (Corning, New York, NY, USA; Cat. no. 356234, 3422, 3599, 3516); MTT (Merck, Berlin, Germany; Cat. no. M5655); MMP-2, MMP-9, E-cadherin, N-cadherin, Vimentin, and β-actin (Thermo Fisher Scientific, Waltham, MA, USA; Cat. no. PA5-85197, PA5-16509, PA5-32178, PA5-29570, MA5-16409, PA146296); TIMP-1 (Abcam, Cambridge, UK; Cat. no. ab211926); anti-rabbit IgG, HRP-linked antibody (Cell Signaling Technology, Danvers, MA, USA; Cat. no. 7074).
Cell Culture
ACHN cells were cultured in special culture medium for ACHN cells at 37 °C in an incubator with 5% CO2. The cell growth status was observed with time, and the solution was changed every day. When the cell growth concentration was above 80%, trypsin was used to digest cells. Cells were passed out every two to three days.
Cell Viability Assay
ACHN cells at the logarithmic proliferation stage were digested and resuspended into single cell suspensions. The cell suspension was taken and 3 × 103 cells per well were inoculated into a 96-well plate. Cells were treated with PA at final concentrations of 0, 20, 40, 80, and 160 µM. The 96-well plate was placed in an incubator for 24 h. Ten microliters of 5% MTT was then added to each well, and the plate was replaced in the incubator for 4 h. The supernatant was discarded, and 100 μL DMSO was added to each well. The 96-well plate was placed in an automatic microplate analyzer and shaken for 10 min to completely dissolve the crystals. The absorbance (A) value of each well was measured at 490 nm. The inhibition rate (%) was calculated as [1 - (cell A in the drug group/cell A in the control group)] × 100%. 41
Colony Formation Assay
ACHN cells at the logarithmic proliferation stage were digested and resuspended into single-cell suspensions. The cell suspension was taken, and 800 cells per well were inoculated into 6-well plates. When the cells began to grow, different concentrations of PA (0, 20, 40, and 80 µM) were added to each well. The cells were cultured continuously for 14 days. During the treatment, the medium containing the drug was changed every 3 days. The cells were fixed with methanol for 30 min and stained with crystal violet for 15 min. The colonies were counted with ImageJ software.
Cell Adhesion Assay
ACHN cells were inoculated in 6-well plates and treated with PA (0, 20, 40, and 80 µM) for 24 h after cell adherence. The Matrigel gel was diluted 8-fold with serum-free medium, and 300 μL of the diluted matrix gel was added to the 24-well plate, which was placed in an incubator for 4 h to make the Matrigel gel solidify. After cleaning the uncured gel with PBS, each hole was sealed with serum-free medium for 90 min. The cells were inoculated with 2 × 104 cells in each well on 24-well plates and incubated for 90 min in a 37 °C incubator. Unadhered cells were washed with PBS; then, 300 μL methanol was added to each well for cell fixation and 300 μL crystal violet for cell staining. The number of adherent cells was recorded under an inverted microscope. The number of adherent cells was analyzed by ImageJ software.
Wound Healing Assay
ACHN cells at the logarithmic proliferation stage were taken and digested by trypsin. The cells were seeded into 6-well plates with 3 × 105 per well and cultured in an incubator with 5% CO2 at 37 °C. When the degree of cell fusion was greater than 80%, lines were drawn on the monolith of fused cells using the tip of a 200 µL pipetting tube. PBS was used to clean the scratched cells. Next, the cells were treated with PA with final concentrations of 0, 20, 40, and 80 µM. The cells were further cultured in the incubator. The scratched area was photographed under an inverted microscope at 0 and 24 h. Cell scratch healing rate {(%) = [(0 h scratch width - scratch width after culture)/0 h scratch width] × 100%}, and cell migration ability were measured. The scratch width was measured by ImageJ software.
Transwell Chamber Assay
Matrigel gel was diluted 8 times in serum-free medium and added to the Transwell chamber. The 24-well plate was placed in an incubator at 37 °C for 4 h to solidify the gel. ACHN cells in logarithmic proliferative phase were taken and cultured in serum-free suspension after trypsin digestion. The resuspended cells were seeded in the upper chamber at 2 × 104 cells per well, adding PA at a final concentration of 0, 20, 40, and 80 µM in the lower chamber. The cells were cultured for 24 h. The culture was terminated, and PBS was used to clean the cells, which were then fixed in methanol for 30 min, and stained with crystal violet for 15 min. The number of submembrane cells in 5 fields, randomly selected from each well, was counted using an inverted microscope. The Transwell migration assay does not require the inner surface of the membrane at the bottom of the chamber, and the remaining steps were the same as the Transwell invasion assay.
High-Content Imaging Technology
ACHN cells at the logarithmic proliferation stage were digested and inoculated into 96-well plates at a density of 3 × 103 cells/well. After cell adherence, the cells were treated with PA at final concentrations of 0, 20, 40, and 80 µM. The cells were cultured for another 12 h. The board was placed in the high-content imaging system machine for further incubation for 12 h, and Harmony 4.1 software was used for data collection and analysis.
Western Blot Analysis
ACHN cells were inoculated into 6-well plates and treated with PA (0, 20, 40, and 80 µM) for 24 h before total cell protein was extracted and quantified. After the target protein was separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), the proteins separated from the glue were completely transferred to a polyvinylidene fluoride (PVDF) membrane. TBS+Tween (TBST) solution containing 5% skim milk powder was used to seal the membrane at room temperature for 2 h. The membrane was incubated with primary antibody for 12 h at 4 °C. 42 The membrane was incubated with a second antibody at room temperature for 2 h. The primary antibodies used in the present study included N-cadherin, E-cadherin, Vimentin, MMP-2, MMP-9, TIMP-1, and β-actin. The protein bands were detected by a gel imaging analysis system. β-Actin was used as the endogenous control.
Statistical Analysis
All data were averaged from at least three independent trials. Graph Prism 8.0 software was used for ordinary one-way analysis of variance to determine statistically significant differences. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 were considered statistically significant.
Footnotes
Authors’ Contributions
Conceptualization: YL, ZC; methodology: ZC, ZW, JH, HW; formal analysis: ZC; investigation: YL, ZW, JH; writing - original draft preparation: YL; writing - review and editing: YL, ZC, HW; project administration: HZ; funding acquisition: HZ; resources: HZ. All authors agreed to be held accountable for the current study in ensuring that the questions related to the integrity of any part of the work are appropriately investigated and resolved. All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Medical College of Yangzhou University, Jiangsu Province, China.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Research and Development Projects of Yangzhou City (No. YZ2020089), Practice Innovation Program of Jiangsu Province (No. SJCX20_1386), and Administration of Traditional Chinese Medicine of Jiangsu Province (No. YB201841).
Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
