Abstract
It has been reported that rhein, a Chinese herbal compound, has a potent anti-inflammatory effect on various diseases. However, it remains elusive whether rhein has a protective effect on chronic kidney disease (CKD) and what its underlying mechanism may be. In the present study, we evaluated whether rhein could prevent lipopolysaccharide (LPS)-induced proliferation, expression of extracellular matrix (ECM) proteins, and inflammation in rat mesangial cells (MCs), and whether these effects were mediated by reactive oxygen species (ROS) and Akt signaling. We also investigated the protective effect of rhein on renal function in a rat CKD model. Results showed that rhein significantly suppressed LPS-initiated proliferation in MCs in a cell counting kit-8 (CCK8) assay. Meanwhile, rhein significantly inhibited LPS-induced expression of ECM proteins and inflammation, as indicated by the decreased expression of fibronectin, α smooth muscle actin (α-SMA), tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), interleukin 6 (IL-6), and interferon γ (IFN-γ). Of note, rhein significantly enhanced LPS-induced production of reactive oxygen species (ROS) and inhibited LPS-induced Akt phosphorylation. Furthermore, the inhibitory effect of rhein on Akt phosphorylation was inhibited by H2O2 scavenger catalase. Importantly, the protective effect of rhein was almost totally lost in the presence of phosphatidylinositol 3-kinase (PI3K) protagonist insulin-like growth factor 1 (IGF-1). Finally, rhein significantly decreased 24 h urinary protein, serum creatinine (SCr), and blood urea nitrogen (BUN) in CKD rats. Collectively, these results suggested that rhein improved CKD through inhibition of proliferation, ECM synthesis, and inflammation via ROS/Akt signaling in MCs.
Introduction
Chronic kidney disease (CKD) is defined as 3 or more months of either kidney damage or a decreased glomerular filtration rate (eGFR) (<60 mL/min/1.73 m2). 1 CKD has a high prevalence with an occurrence of 10% to 13% in the general population worldwide. Importantly, CKD is closely related to an increased risk of end-stage renal disease and associated complications and can directly result in death.2,3 Furthermore, CKD can significantly affect life span/quality and places a heavy burden on health care systems, families, and society. 4 Therefore, it is urgent to develop new therapeutic drugs for the treatment of CKD.
Accumulating studies show that mesangial cells (MCs) exhibit increased proliferation, increased extracellular matrix (ECM) deposition, and inflammation in CKD. 5 For example, it has been reported that the proliferation of MCs is significantly increased during glomerular injury progression.6,7 The secretion of ECM proteins, including fibronectin and α smooth muscle actin (α-SMA), was also increased in MCs, 8 leading to kidney fibrosis and CKD. 9 Furthermore, activated MCs also synthesize and secrete various pro-inflammatory cytokines such as tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), interleukin 6 (IL-6), and interferon γ (IFN-γ), 10 which play a critical role in the pathogenesis and progression of CKD. 11 In addition, under pathologic conditions, MCs could express monocyte chemoattractant protein 1 (MCP-1), which then promotes the accumulation of macrophages, leading to the progression of glomerular injury. 12 Therefore, MCs have become a pharmacological target for the prevention and treatment of CKD. 13
Phosphoinositide-3-kinase, or PI3K, generates phosphatidylinositol-34,5-triphosphate (PIP3), a lipid secondary transmitter, which is critically involved in cell growth, apoptosis, and metabolism.14,15 The main downstream regulator of PI3K is serine/threonine kinase (Akt) also called protein kinase B (PKB). 16 In response to lipopolysaccharide (LPS) stimulation in MCs, PI3K/Akt signaling pathways promote cell cycle progression through the regulation of cyclin-dependent kinase (CDK)2 and CDK4. These proteins then act on G1/S phase transition to induce cell proliferation.17–19 In addition to proliferation, Akt signaling also contributes to ECM deposition and inflammation in MCs.17,20 Interestingly, increased production of reactive oxygen species (ROS) can inactivate Akt signaling, thereby leading to blockage of proliferation, ECM deposition and inflammation.21,22
The Chinese herbal compound 9,10-dhydro-4,5-dihydroxy-9.10-dioxo-2-anthracenecarboxvlic acid (rhein) is found in various medicinal herbs, especially in rhubarb (Rheum rhabarbarum). 23 Studies have shown that rhein possesses antioxidation, antiproliferation, and antifibrosis effects in a 5/6 nephrectomied-induced CKD rat model. 24 Specifically, it has been reported that rhein inhibits proliferation and induces apoptosis of renal/colorectal cancer cells through inducing cell cycle arrest in a ROS/Akt-dependent manner.25–27 However, it remains elusive whether rhein has any protective effect on CKD.
In the present study, we hypothesized that rhein has a protective effect on CKD, and that this effect is mediated by the ROS/Akt signaling pathway. Therefore, we performed in vitro experiments to evaluate whether rhein could inhibit LPS-induced proliferation, ECM deposition, and inflammation in MCs, and whether this protective effect was mediated by the ROS/Akt pathway. We also performed in vivo experiments to investigate the protective effects of rhein in a rat CKD model.
Materials and Methods
Cell Culture
The rat MC cell line (HBZY-1 cells) was purchased from Chinese Center for Typical Culture Collection (CCTCC). Cells were cultured in Dulbecco's Modified Eagle Medium (DMEM, Hyclone) plus 10% fetal bovine serum (Gibco) with 5% CO2 at 37 °C. LPS at 30 μg/mL (Sigma-Aldrich) was used to treat cells. 17 Some cells were pretreated with rhein (100 μmol/L, Sigma-Aldrich), 23 catalase (1000 U/mL, Sigma-Aldrich), 28 insulin-like growth factor 1 (IGF-1, 10 nmol/L, Sigma-Aldrich), 17 or their combinations as indicated 30 min prior to LPS treatment.
Cell Proliferation Assay
Cell counting kit-8 (CCK-8; Beyotime) was used to evaluate cell proliferation. Briefly, cells were cultured at 104 cells/well in 96-well plates for 24 h, then treated with LPS with or without rhein for 0, 24, 48, and 96 h. Afterward, cells were incubated with CCK8 solution for 4 h, the medium was discarded and 100 μL of dimethyl sulfoxide (DMSO) was added. Absorbance was detected at 540 nm with a microplate reader (BioRad).
Flow Cytometry
CCK8 was from Ruian BioTechnologies. After cells were incubated with serum containing rhein for 24 h in 6-well plates, they were harvested and washed 3 times with ice-cold phosphate-buffered saline (PBS). The cells were then incubated in PBS containing 400 μL propidium iodide (PI) dye, 200 μL 0.02% Triton X-100, and 20 μL RNase A for 30 min at room temperature in the dark. Fluorescence was detected using FACSAria (BD Biosciences) and analyzed with FlowJo 7.6.1 software.
Annexin V-FITC/PI apoptosis detection kit was from Dojindo. Briefly, MCs were incubated with 2.5 µg/mL Annexin V-FITC and 50 µg/mL PI, and then cells were analyzed by FACSAria flow cytometry.
Determination of ROS
2′,7′-Dichlorodihydrofluorescein diacetate (H2DCFDA) staining was used to determine ROS production in MCs. On oxidation, H2DCFDA becomes 2′,7′-dichlorofluorescein, which shows highly green fluorescent. Briefly, MCs were incubated with 10 μmol/L H2DCFDA at 37 °C for 30 min, and fluorescence was measured with an AMINCO-Bowman Series 2 (AB2) Spectrofluorimeter (Thermo Fischer Scientific). The excitation and emission were set at 490 and 530 nm, respectively.
Quantitative Polymerase Chain Reaction (qPCR)
TRIzol reagent (Invitrogen) was used to extract total RNA from MCs following the manufacturer's protocol. qPCR was performed using Premix Ex Taq DNA polymerase for qPCR (RR039B, Takara). The primers were as follows: TNF-α forward 5′-CTCAAGCCCTGGTATGAGCC-3′, reverse 5′-GGCTGGGTAGAGAACGGATG-3′; IL-1β forward 5′-GGAAGGCAGTGTCACTCATTGTG-3′, reverse 5′-GGTCCTCATCCTGGAAGCTCC-3′; IL-6 forward 5′-TCCTACCCCAACTTCCAATGC-3′, reverse 5′-TAGCACACTAGGTTTGCCGAG-3′; IFN-γ forward 5′-AGGACGGTAACACGAAA-3′, reverse 5′-CTGTGGGTTGTTCACCTC-3′; and GAPDH forward 5′-AGACAGCCGCATCTTCTTGT-3′, reverse 5′-CTTGCCGTGGGTAGAGTCAT-3′. All the sequences were synthesized by Sangon Biotech.
Western Blotting
The proteins of MCs were extracted and quantified as described previously. 17 After separation by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), the proteins were transferred to polyvinylidene difluoride membranes using a mini Trans-Blot (Bio-Rad). After blocking with 5% nonfat milk powder (w/v) for 1 h at room temperature, membranes were probed with primary antibodies against proliferating cell nuclear antigen (PCNA) (1:1000, Cat No. 10205-2-AP, Proteintech), cyclin D1 (1:500, Cat No. 26939-1-AP, Proteintech), pAkt (1:1000, Cat No. 66444-1-Ig, Proteintech), total Akt (1:1000, Cat No. 10176-2-AP, Proteintech), fibronectin (A12932, Abclonal), α-SMA (A7248, Abclonal), and β-actin (AC028, Abclonal). After incubation with horseradish peroxidase (HRP)-coupled antirabbit antibodies at room temperature for 1 h, the membranes were examined with an enhanced chemiluminescence (ECL) kit. The brightness of the protein bands was analyzed with ImageJ 1.51 software.
Animals
All of the procedures were performed in accordance with the guidelines set by the Animal Care and Use Committees of Jinhua Hospital of Traditional Chinese Medicine. Male Wistar rats weighing 339 ± 42 g at the start of the experiment were used. Rats were housed individually in a temperature- and humidity-controlled environment, with a 12 h light:12 h dark cycle. All of the procedures were performed under general anesthesia by intraperitoneal injection (i.p) of sodium pentobarbital (∼50 mg/kg body weight); 5/6 nephrectomy was conducted according to the established CKD rat model, as previously described. 29 In brief, the left kidney was exposed and decapsulated, and the upper and lower kidney poles were removed (about 2/3 of the kidney). After recovery for 14 d, the right kidney was removed. The control group was subjected to a sham operation: during this procedure, the kidneys were exposed, and then a renal capsule was removed. Animals were sacrificed 6 months after surgery under general anesthesia. Some CKD rats were treated with rhein (35, 70, and 140 mg/kg/d) as indicated. Losartan (30 mg/kg/d) was used as positive control.30,31
Biochemical Test
Twenty-four-hour urine, serum creatinine (SCr), and blood urea nitrogen (BUN) were measured using the Hitachi 912 automated clinical chemistry analyzer (Roche Diagnostics Ltd.). The SOD activity was measured as previously described. 32 In the presence of superoxide radical, nitroblue tetrazolium (NBT) was degraded into formazan with the xanthine–xanthine oxidase system. Then, the absorption of formazan was detected at 560 nm. The malondialdehyde (MDA) content in the rat kidneys was measured using a kit from Nanjing Jian Cheng Bioengineering Institute according to the manufacturer's instruction. The MDA concentration was normalized to protein content in each sample.
Statistical Analysis
The data are expressed as mean ± standard error of the mean (SEM). The analysis was performed by one-way analysis of variance (ANOVA) followed by Tukey test with GraphPad Prism 7.0 software. A P value of <.05 was considered statistically significant.
Results
Rhein Suppressed LPS-Induced Proliferation in MCs
First, we evaluated whether rhein has a protective effect against LPS-induced proliferation in MCs. As shown in Figure 1A, rhein significantly inhibited LPS-induced increase of cell number in MCs in CCK8 assay. Rhein was also shown to significantly inhibit LPS-induced expression of PCNA in Western blot assay (Figure 1B). We further evaluated whether the inhibitory effect of rhein on proliferation involves G1/S cell cycle progression. As shown in Figure 1C, rhein treatment resulted in G1/S phase delay. Meanwhile, the protein level of cyclin D1, a proliferation marker, was significantly inhibited by rhein (Figure 1D). In contrast, Figure 1E and F shows that rhein treatment had no effect on LPS-induced apoptosis. These results suggested that rhein suppressed the proliferation of MCs by promoting G1/S delay.

Rhein suppressed LPS-induced proliferation of MCs by G1/S phase delay in MCs. (A) Effect of rhein on LPS-induced proliferation of MCs in CCK8 assay. (B) Representative Western blot images (upper panel) and summarized data of band intensity (lower panel) showing the inhibitory effect of rhein on LPS-induced PCNA expression in MCs. (C) Representative flow cytometric images (upper panel) and summarized data (lower panel) showing the inhibitory effect of rhein on LPS-induced G2 increase. (D) Representative Western blot images (upper panel) and summarized data (lower panel) showing the inhibitory effect of rhein on LPS-induced expression of cyclin D1 in MCs. Representative flow cytometric images (E) and summarized data (F) showing the inhibitory effect of rhein on LPS-induced cell apoptosis in MCs. n = 4. *P < .05 versus vehicle group; #P < .05 versus LPS group.
Rhein Inhibited LPS-Induced ECM Deposition and Inflammation in MCs
Next, the inhibitory effect of rhein on ECM deposition in MCs was evaluated. As shown in Figure 2A and B, LPS significantly increased mRNA level of fibronectin and α-SMA, and this stimulatory effect was significantly inhibited by pretreatment with rhein. Consistently, LPS significantly increased the protein level of fibronectin and α-SMA, and this stimulatory effect was significantly inhibited by rhein (Figure 2C and D). These results suggested that rhein inhibited LPS-induced ECM deposition.

Rhein suppressed LPS-induced expression of fibronectin/α-SMA and inflammation in MCs. Summarized data showing the effect of rhein on LPS-induced fibronectin (A) and α-SMA (B) mRNA level in MCs. Representative Western blot images (upper panel) and summarized data (lower panel) showing the inhibitory effect of rhein on LPS-induced expression of fibronectin (C) and α-SMA (D). Summarized data showing the inhibitory effect of rhein on LPS-induced increase in mRNA level of TNF-α (E), IL-1β (F), IL-6 (G), and IFN-γ (H) in MCs. FN, fibronectin. n = 4. *P < .05, **P < .01 versus vehicle group; #P < .05 versus LPS group.
It has been reported that inflammation plays a central role in the initiation and development of CKD. Therefore, we investigated whether rhein has an anti-inflammatory effect on MCs. Figure 2E to H shows that LPS significantly increased the expression of TNF-α, IL-1β, IL-6, and IFN-γ. Notably, the stimulatory effect of LPS was significantly inhibited by rhein. These results suggested that rhein suppressed LPS-induced inflammation.
Rhein Prevented LPS-Induced Alteration of ROS/Akt Signaling in MCs
Previous studies show that ROS production and Akt phosphorylation play a central role in LPS-induced pathological changes. Therefore, we evaluated whether rhein would have any effect on LPS-induced ROS production and Akt phosphorylation. As shown in Figure 3A and B, LPS induced significant ROS production, and ROS content was further enhanced by rhein. Figure 3C shows that LPS significantly increased Akt phosphorylation in MCs, which were significantly inhibited by rhein. Interestingly, the inhibitory effect of rhein on LPS-induced Akt phosphorylation was almost totally lost in the presence of the H2O2 scavenger catalase (Figure 3D). These results suggested that rhein inhibited LPS-induced Akt phosphorylation in a ROS-dependent manner.
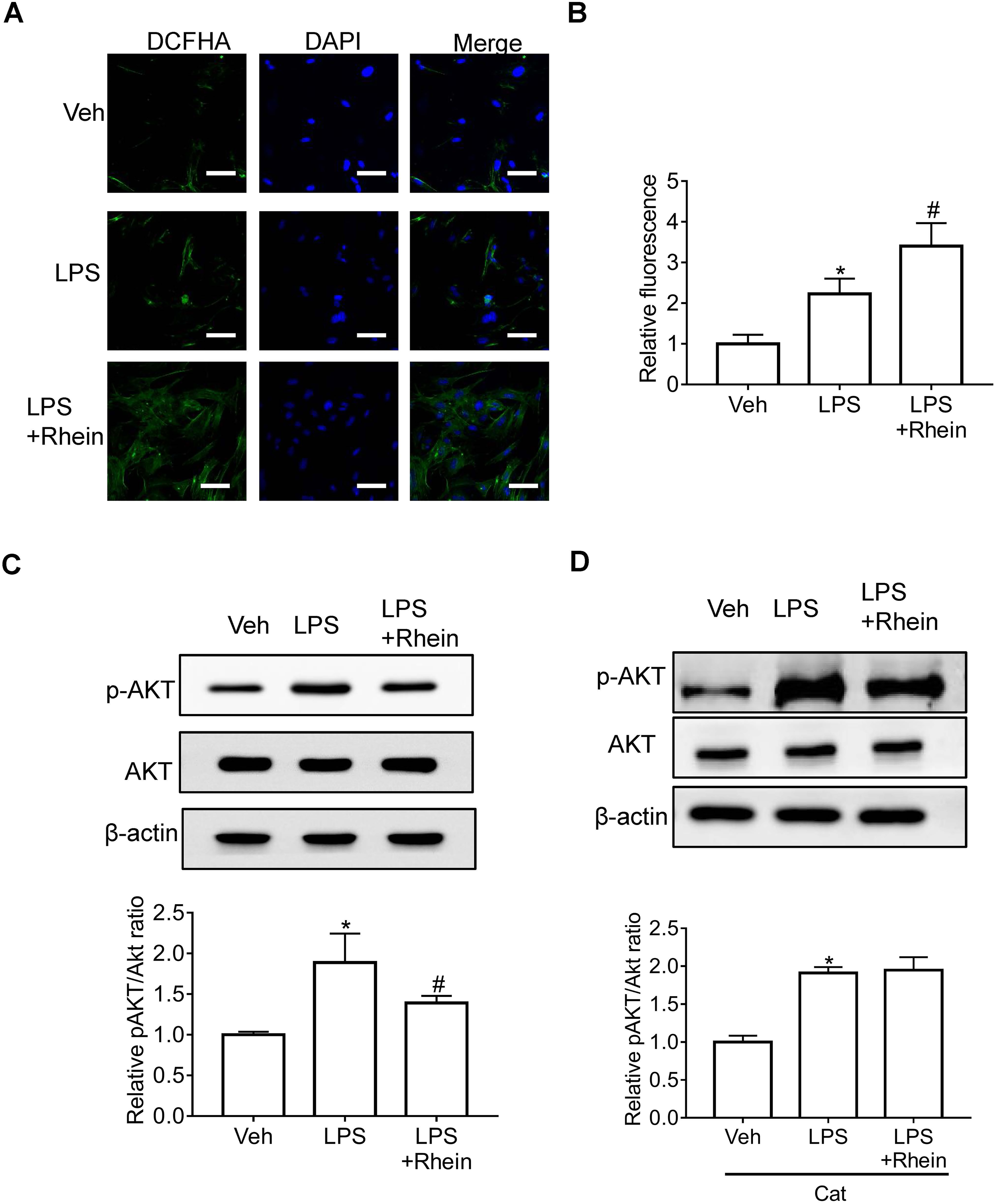
Rhein prevented LPS-induced changes of ROS and Akt phosphorylation in MCs. Representative fluorescence images (A, scale bar = 100 μm) and summarized data (B) showing the enhancing effect of rhein on LPS-induced ROS production. (C) Representative Western blot images (upper panel) and summarized data (lower panel) showing the inhibitory effect of rhein on LPS-induced Akt phosphorylation. (D) Representative Western blot images (upper panel) and summarized data (lower panel) showing the inhibitory effect of rhein on LPS-induced Akt phosphorylation in the presence of catalase. n = 4. *P < .05 versus vehicle group; #P < .05 versus LPS group.
Rhein Inhibited LPS-Induced Proliferation, Matrix Deposition, and Inflammation of MCs Through Akt Signaling
We then further evaluated whether Akt mediates the protective effect of rhein. As shown in Figure 4A, the inhibitory effect of rhein on LPS-induced proliferation of MCs was significantly attenuated in the presence of the PI3K agonist IGF-1 (Figure 4A). Similarly, IGF-1 significantly prevented the inhibitory effect of rhein on LPS-induced expression of fibronectin and α-SMA (Figure 4B and C). Also, IGF-1 significantly prevented the inhibitory effect of rhein on LPS-induced inflammation, as indicated by altered expression of TNF-α, IL-1β, IL-6, and IFN-γ (Figure 4D-G). These results suggested that Akt was involved in the protective effect of rhein in LPS-induced proliferation, ECM deposition, and inflammation.

Rhein inhibited LPS-induced proliferation, expression of fibronectin/α-SMA, and inflammation through targeting ROS/Akt signaling in MCs. (A) Effect of rhein on LPS-induced proliferation of MCs in the presence of PI3K agonist IGF-1 in CCK8 assay. Representative Western blot images (upper panel) and summarized data (lower panel) showing the inhibitory effect of rhein on LPS-induced expression of fibronectin (B) and α-SMA (C) in the presence of IGF-1. Summarized data showing that IGF-1 prevented the inhibitory effect of rhein on LPS-induced increase of mRNA level of TNF-α (D), IL-1β (E), IL-6 (F), and IFN-γ (G). FN, fibronectin. n = 4. *P < .05, **P < .01 versus vehicle group; #P < .05 versus LPS group.
Rhein Improved CKD
CKD rats were treated with different doses of rhein, losartan, or vehicle by intragastric administration. After treatment, 24 h urinary protein, SCr, and BUN were measured to evaluate kidney function. We found that rhein at 35 mg/kg significantly improved kidney function in CKD rats, and this effect was comparable to that of losartan at 30 mg/kg (Figure 5A-C). Importantly, the protective effect was enhanced as the dosage was further increased (Figure 5A-C). Meanwhile, rhein significantly increased SOD and reduced MDA (Figure 5D and E). These results suggest that rhein improved kidney function and reduced oxidative stress in CKD rats.

Rhein improved CKD. The 24 h urinary protein level (A), SCr (B), BUN (C), SOD (D), and MDA (E) in rat CKD models with or without rhein (35, 70, and 140 mg/kg/d) or losartan (30 mg/kg/d) treatments. n = 8. *P < .05 versus sham group; #P < .05, ##P < .01 versus CKD group.
Discussion
In the present study, we evaluated whether rhein has any protective effects on kidney function in CKD and attempted to determine the underlying mechanisms. Our in vitro study showed that rhein inhibited LPS-induced proliferation, ECM deposition, and inflammation in MCs, and this protective effect of rhein was mediated by ROS/Akt signaling. Further in vivo study shows that rhein dose-dependently improved kidney function in the rat CKD model. These results suggest that rhein may have clinical usage in the prevention and treatment of CKD.
It has been estimated that MCs comprise about one-third of the cell population within the glomerulus, and that MCs are the main source of mesangial matrix, which plays a critical role in maintaining the structural architecture of the glomerulus. 13 MCs also regulate phagocytosing apoptotic cells and immune complexes. 8 Indeed, increased proliferation, ECM deposition, and inflammation in MCs are common features of CKD, and these pathological changes in MCs have become an important target for the prevention and treatment of CKD. 33 Of note, rhein has shown potent protective effects through inhibition of proliferation, ECM deposition, and inflammation in various cell types. For example, it has been shown that rhein can induce cell cycle arrest, thereby inhibiting proliferation and inducing apoptosis in renal/colorectal cancer cells.25–27 Rhein prevented the development of glomerulosclerosis and the progression of IgA nephropathy (IgAN) effects that may be mediated by decreased expression of fibronectin and α-SMA in renal tissues. 34 In another study, rhein inhibited inflammatory injury through activation of NOD-like receptor protein 3 (NLRP3) inflammasome.35,36 Consistent with these findings, we here present data showing that rhein inhibited LPS-induced proliferation, ECM synthesis, and inflammation in vitro, and rhein improved kidney function in vivo.
It has been reported that rhein exerts protective effects through distinct mechanisms in different diseases and/or models. For example, it has been demonstrated that rhein inhibits the proliferation, migration, and invasion of human renal carcinoma cells, and that these effects were mediated by the mitogen-activated protein kinase (MAPK) signaling pathway. 25 Rhein reduced cell viability, induced cell apoptosis, and induced cell cycle arrest at the G2/M phase in colorectal cancer cells, and these effects were mediated by the inhibitory effect on the phosphorylation of signal transducer and activator of transcription 3 (STAT3). 26 In other research, Tu et al found that rhein inhibits the AMP-activated protein kinase/mammalian target of rapamycin (AMPK/mTOR)-mediated autophagy, thereby inhibiting tubular fibrosis in adenine-induced renal tubular injury rats. 37 Finally, Wu et al reported that rhein protected CKD through the sirtuin 3 (SIRT3)/forkhead box O3α (FOXO3α)-dependent signaling pathway in CKD. 24
Of note, studies have revealed that increased generation of ROS could inactivate PI3K/Akt signaling. For example, Wen et al demonstrated that the thioredoxin reductase-1 inhibitor chaetocin inhibited the proliferation of gastric cancer cells through increased generation of intracellular ROS and subsequent inactivation of PI3K/Akt. 38 In another study, Liu et al reported that rhein inhibits proliferation and induces apoptosis through ROS-PI3K/Akt inactivation in pancreatic cancer cells. 27 Besides proliferation, ROS-PI3K/Akt may also play a role in ECM deposition and inflammation, since Akt was critically involved in ECM deposition and inflammation.39,40 Consistent with these findings, we here present data showing that rhein inhibited LPS-induced proliferation, ECM deposition, and inflammation through ROS-PI3K/Akt signaling. In contrast, there are studies showing that ROS induces proliferation in various cell types, including oral cancer cells 41 and acute myeloid leukemia. 42 Therefore, ROS may have distinct roles in the proliferation of various cell types in different contexts.
In summary, the present study demonstrated that rhein improved kidney function in CKD in rats, which may be mediated by the decreased proliferation, ECM deposition, and inflammation of MCs through the ROS/Akt signaling pathway. The present study provides a theoretical basis for the potential clinical usage of rhein in the prevention and treatment of CKD.
Footnotes
Authors’ Note
RX and XF conceived the concept of the study and generated data. RX, XH, SH, MY, and XF performed the experiments and collected the data. RX and XF did the statistical analysis. RX drafted the manuscript. XF edited the manuscript. All coauthors have read and approved the final manuscript. This research project was approved by the Research Ethics Committee of Shanghai University of Traditional Chinese Medicine (Approval No: SZY201508002).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of Zhejiang Province (grant number LY15H270001).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Jinhua Hospital of Traditional Chinese Medicine, China and approved by the Administration Committee of Experimental Animals, Jinhua Hospital of Traditional Chinese Medicine, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
