Abstract
Ehretia asperula (E asperula) is a well-known traditional medicinal plant in Vietnam with potent activity against a wide range of diseases, including hepatitis B and various cancers. Although E asperula has been used in traditional medicine, the efficacy of E asperula and its bioactive components on retinal degenerative diseases has not been fully evaluated. In present this study, we found that ethanolic extracts of E asperula increased cell viability in retinal precursor cells exposed to glutamate/BSO-induced excitotoxicity/oxidative stress. The major responsible bioactive compounds were rosmarinic acid and methylrosmarinic acid. First, 10 known compounds were isolated from E asperula leaves. Their chemical structures were determined using 1D and 2D nuclear magnetic resonance, and compared with published data. Using high-performance liquid chromatography, we determined the content of 4 compounds in E asperula extract: rosmarinic acid, lithospermic acid B, astragalin, and kaempferol 3-rutinoside. The most abundant of these compounds was lithospermic acid B. The protective effects of the pure compounds and ethanolic extracts against excitotoxicity and oxidative stress-induced retinal cell death were tested in R28 cells. Both 70% and 95% ethanolic extracts of E asperula increased cell viability in these conditions. Rosmarinic acid and methyl rosmarinic acid were more effective at protecting against retinal cell death and elevated reactive oxygen species in cells subjected to glutamate/BSO-induced excitotoxicity/oxidative stress. These findings suggested that E asperula could potentially be used to treat retinal degeneration.
Introduction
As the population ages, an increasing number of people will suffer from aging-related visual impairments, such as decreased visual acuity and visual field loss, and struggle to perform routine activities 1 . Optic neuropathy is typically caused by age-related loss of sensory activity in the central nervous system, and degeneration of the optic nerve eventually leads to glaucoma, which causes visual field loss and irreversible blindness2,3. This condition affects an estimated 60 million people worldwide, with approximately 13% of the affected suffering from bilateral blindness 4 . In fact, because glaucoma can be asymptomatic until a relatively late stage, a much greater number of patients than estimated could suffer optic neuropathy-related visual impairment 5 . Despite the availability of glaucoma medications, these medications are commonly used to treat IOP, and there are no available modalities to treat optic nerve degeneration.
The death of retinal ganglion cells (RGCs), a population of neurons in the inner layer of the retina, is the most important factor in glaucoma-related blindness. RGCs, like many other neurons, are post-mitotic and do not recover or regenerate after being damaged. Glaucoma is classified as a neurodegenerative disorder, similar to Alzheimer's and Parkinson's disease6,7. Oxidative stress is a common pathological mechanism of neurodegenerative diseases 6 , and is caused by imbalanced generation and removal of reactive oxygen species (ROS). An overabundance of ROS results in oxidative damage to cellular macromolecules such as DNA, proteins, and lipids, ultimately leading to neuronal cell death. Since the 1980s, when the link between glaucoma and oxidative stress was first proposed, numerous studies in experimental animal models of glaucoma and clinical samples have supported this link6–8. Therefore, antioxidant substances that inhibit oxidative stress in RGCs may be promising candidates for protection against optic nerve degeneration, and preventing RGC death by correcting the overabundance of ROS characteristic of glaucoma is considered to be a viable potential therapeutic approach9,10.
Ehretia asperula plant is endemic to Northern Vietnam. E asperula leaves have traditionally been used as a folk medicine to treat a variety of ailments such as hepatitis, liver cirrhosis, and cancer 11 . Furthermore, E asperula prevents or alleviates diabetes, hypertension, and acne 12 . Some researchers have recently revealed the potential pharmacological effects of E asperula in cancer treatment 13 , but there are no trial results using E asperula in cancer therapy. One study identified that the leaves of E asperula contained several constituents with anti-cancer properties, but the underlying mechanisms responsible for these effects are unknown 14 . Only a few reports to date have characterized the chemical components of this plant and their biological effects, particularly on eye disease. Therefore, the aim of the present study was to investigate the chemical and chromatographic profiles of E asperula extracts, and their antioxidative activities against oxidative/excitotoxic stress-induced retinal cell death.
Results and Discussion
Ethanolic extract of E asperula (EEEA) was subjected to open column chromatography, isolating 10 known compounds (

Structures of compounds isolated from E asperula.
High-performance liquid chromatography with diode-array detection (HPLC-DAD) analyses identified 4 major compounds,

Representative HPLC fingerprint profile of 70% EtOH (v/v) E asperula leaf extract (detection wavelength = 260 nm). 1 = lithospermic acid B; 4 = rosmarinic acid; 6 = kaempferol 3-rutinoside; 7 = astragalin.
The calibration curves for 4 compounds were calculated using the linear relationships between concentration (x) and peak area (y) of each compound and are shown in Table 1 as regression equations (y = ax + b). All 4 calibration curves demonstrated acceptable linearity (r2 ≥ 0.9996). The limits of detection (LOD) ranged from 0.196367 to 2.447649 μg/mL, and limits of quantification (LOQ) ranged from 0.595053 to 7.417119 μg/mL for 4 standard compounds.
Regression Equation, Linear Range, LOD, LOQ, and Content of 4 Compounds.

LOD, limit of detection; LOQ, limit of quantification.
y, means peak area; x, means concentration.
LOD = 3.3 × (SD of intercept/slope of the calibration curve).
LOQ = 10 × (SD of intercept/slope of the calibration curve).
Quantitative analysis of rosmarinic acid, lithospermic acid B, astragalin, and kaempferol 3-rutinoside in E asperula extract was performed using the analytical method. These compounds were found in concentrations ranging from 0.303 to 13.439 mg/g (Table 1). The most abundant of these 4 compounds was lithospermic acid B (13.439 ± 0.375 mg/g).
To determine the in vitro cytoprotective effect of E asperula on retinal cells, 10%, 30%, 50%, 70%, and 95% ethanolic extracts were tested. These extracts did not significantly affect the cell viability of untreated R28 rat retinal precursor cells at concentrations of 10 and 50 μg/mL (Figure 3A). To determine if the EEEAs were cytoprotective, R28 cells treated with glutamate and l-buthionine-[S,R]-sulfoximine (BSO) were examined in the presence of different types of EEEA. Glutamate is a major retinal excitatory transmitter that induces retinal cell death when present in high concentration 23–25. The addition of glutamate to R28 cell culture causes severe damage in vitro, including decreased viability and cell death, mimicking excitotoxicity in the retina 26 . Furthermore, BSO is a potent inhibitor of glutathione synthesis and is used to enhance the cytotoxic effects of various drugs 27 . As expected, treatment with glutamate and BSO resulted in a decrease in cell viability, but the antioxidant, N-acetylcysteine, reversed the glutamate/BSO-induced cytotoxicity in R28 cells (Figure 3B). Interestingly, the 70% and 95% EEEA effected the cell viability under identical experimental conditions (Figure 3B).

Protective effect of ethanol extract of E asperula (EEEA) against glutamate/BSO-induced cell death in R28 cells. (A) R28 cells were treated with DMSO (vehicle), 10 and 50 µg/mL of 10%, 30%, 50%, 70%, and 95% EEEA or N-acetyl-L-cysteine (NAC) for 24 h. Cell viability of R28 cells was measured by MTT assay. (B) R28 cells were pretreated with DMSO (vehicle), 10 and 50 µg/mL of 10%, 30%, 50%, 70%, and 95% EEEA or NAC (1 mM) as a positive control for 1 h, and then incubated with 10 mM glutamate and 0.5 mM BSO for 24 h. Cell viability of R28 cells was measured by MTT assay (Con: untreated control; Veh: vehicle). Data are expressed as mean ± SEM. ***P<.001.
Next, the bioactive compounds obtained from the 70% ethanol extract were investigated. Compounds

Protective effects of compounds isolated from EEEA against glutamate/BSO-induced cytotoxicity in R28 cells. (A) R28 cells were treated with the indicated concentrations of isolated compounds (EA1-EA10) or NAC for 24 h. Cell viability of R28 cells was measured by MTT assay. (B) R28 cells were pretreated with DMSO (vehicle), indicated concentrations of isolated compounds (EA1-EA11), or NAC for 1 h, and then incubated with 10 mM glutamate and 0.5 mM BSO for 24 h. Cell viability of R28 cells was measured by MTT assay (Con: untreated control; Veh: vehicle). Data are expressed as mean ± SEM. ***P<.001.
Glutamate-induced cytotoxicity is generally accompanied by ROS production
10
, so the antioxidant effects of compounds

Reduced ROS production by rosmarinic acid (RA) and methylrosmarinic acid (MRA) isolated from EEEA. (A-C) R28 cells were treated with DMSO (vehicle), indicated concentrations of EEEA, RA, MRA, or NAC for 1 h (C: untreated control; V: vehicle). Then, ROS production was induced by (A) 1 mM H2O2, (B) 1 mM KO2, or (C) 1 mM H2O2 and 100 μM ferrous perchlorate (II) in R28 cells. Data are expressed as mean ± SEM. *P<.05, ***P<.001. Experiments were independently repeated thrice.
Rosmarinic acid (
In conclusion, 10 known compounds (
Materials and Methods
Chemicals and Apparatus
The 1D and 2D nuclear magnetic resonance spectra were obtained using a 500 MHz Bruker Avance DRX spectrometer, and chemical shifts were recorded in parts per million. Silica gel (Merck, 63-200 μm particle size) and RP-C18 (Merck, 75 μm particle size) particles were used for column chromatography. Thin-layer chromatography (TLC) was performed using Merck 60 F254 and RP-C18 F254 silica gel plates. Isolated compounds were visualized after spraying with aqueous 20% H2SO4 and heating for approximately 5 min. Analytical-grade acetonitrile and distilled HPLC-grade water were purchased from Fisher Scientific. Open column chromatography was performed using silica gel (Merck) and Sephadex LH-20 (Pharmacia).
Plant Material and Extraction
Leaves of E asperula (5 kg) were collected in Northern Vietnam in 2018. Botanical identification was performed by Msc. Trong Duc Nghiem. A herbarium specimen was deposited at the Department of Botany, Hanoi University of Pharmacy. Air-dried leaves were ground and extracted with 70% ethanol at room temperature 3 times (3 h × 20 L). The ethanol extract was obtained by solvent evaporation.
Isolation and Identification
The ethanol extract (180 g) was resuspended in distilled water and successively partitioned using hexane (Hx) (12 g), methylene chloride (MC) (28 g), ethyl acetate (EtOAc) (15 g), and n-butanol (BuOH) (20 g). The MC-soluble fraction was subjected to silica gel column chromatography using a stepwise gradient of Hx–EtOAc (10:5 to 0:1) to yield an isomeric mixture of
HPLC Analysis
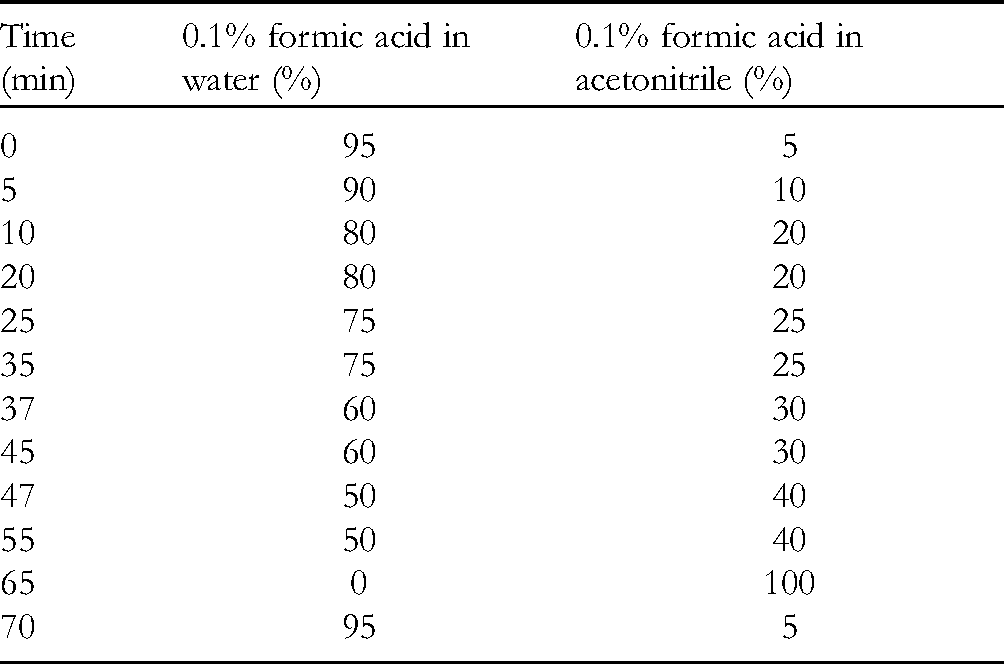
The analyses were conducted using an Agilent series 1200 liquid chromatography system and a YMC pack pro C18 column packed with 5 µm particles (4.6 × 250 mm) maintained at 25 °C in a column oven. The HPLC method is presented in Table 2. Formic acid (0.1%) was added to both acetonitrile and water mobile phases to obtain a better peak shape. The injection volume was 10 µL. The flow rate was 0.7 mL/min, and UV detection was performed at 260 nm.
HPLC Method.
Sample Preparation for HPLC Analysis
Ethanol extract of E asperula leaves (10 mg) was dissolved in methanol (1 mL) and filtered through a syringe filter (0.45 µm). Four standard isolated compounds with purities ≥98% as evaluated using HPLC analysis, were dissolved in methanol to make stock solutions (1 mg/mL), and then further diluted with methanol to obtain various concentrations for quantitative analysis.
Calibration Curve, LOD, and LOQ
The calibration curves for 4 standard compounds were determined from the peak areas of the standard solutions at various concentrations: rosmarinic acid (7.8125-250 μg/mL), lithospermic acid B (7.8125-250 μg/mL), astragalin (1.953125-62.5 μg/mL), and kaemferol 3-rutinoside (1.953125-62.5 μg/mL). All measurements were performed in triplicate to produce calibration curves. The LOD and LOQ for the 4 standard compounds were determined using the standard deviation (SD) of the intercept and slope of the calibration curve, as derived by the following equations:

Cell Culture
Immortalized rat retinal precursor cells (R28 cells) were purchased from Kerafast (USA). R28 cells were cultured in 75 cm2 culture flasks in Dulbecco's modified Eagle's medium (DMEM; HyClone) supplemented with 10% fetal bovine serum (FBS; HyClone) and 100 U/mL penicillin/streptomycin (HyClone). The cultures were maintained at 37 °C in a humidified atmosphere containing 5% CO2.
Cell Viability
R28 cells were seeded in 96-well plates and incubated for 24 h. After 1 h of pretreatment with various concentrations of EEEA, glutamate (10 µM) and 1-buthionine-(S,R)-sulfoximine (BSO; 0.5 mM) mixture (glutamate/BSO) was added to the culture and kept for 24 h. To measure cell viability, MTT (3-[4,5-dimethylthiazol-2-yl]2,5-diphenyltetrazolium bromide) solution was added to the cells (final concentration: 0.5 mg/mL) at 37 °C and maintained for 1 h. The optical density of the solubilized formazan product was measured using a spectrophotometer (BioTek Instruments, VT, USA) at a test wavelength of 570 nm and reference wavelength of 690 nm.
Assessment of ROS Production
Intracellular ROS levels were determined using dichlorodihydrofluorescein diacetate (DCFH-DA) and ROS levels were assayed as previously described 10 . Briefly, R28 cells were pretreated with the indicated concentrations of the compounds or extracts for 1 h, and the cells were then loaded with the DCFH-DA radical probe (10 µM) and incubated for 20 min at 37 °C. After removing the excess probe, 1 mM H2O2 (H2O2 radical), 1 mM H2O2 plus 100 µM iron (II) perchlorate hexahydrate (•OH), or KO2 at 1 mM (O2•−) were added to generate the radical species. Fluorescence was measured after the ROS-generating compounds were present for various time periods, using excitation/emission wavelengths of 485/535 nm (luminescence spectrometer LS50B, PerkinElmer, UK).
Statistical Analysis
Data are expressed as mean percentage of the control value ± standard error of the mean (SEM). Statistical comparisons were performed using one-way analysis of variance followed by Dunnett's test. Statistical analyses were conducted using GraphPad Prism, version 7.0 (GraphPad). Differences were considered statistically significant at P < .05.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211067986 - Supplemental material for Protection Against Oxidative Stress-Induced Retinal Cell Death by Compounds Isolated From Ehretia asperula
Supplemental material, sj-docx-1-npx-10.1177_1934578X211067986 for Protection Against Oxidative Stress-Induced Retinal Cell Death by Compounds Isolated From Ehretia asperula by Tam Thi Le, Tae Kyeom Kang, Ha Thi Do, Trong Duc Nghiem and Wook-Bin Lee, Sang Hoon Jung in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by an intramural grant (2Z06481) from the Korea Institute of Science and Technology (KIST), Republic of Korea and by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education(2021R1C1C100700711).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
