Abstract
Introduction
Epidemiological surveys have reported that the global incidence of dry eye continues to rise, ranging from 5.5 to 3.7%, with the highest incidence in women, the elderly, and Asians.1–3 The pathogenesis of dry eye presently focuses on ocular surface inflammation, poor tear film stability, tear hypertonic environment, cell apoptosis, abnormal neuromodulation and abnormal sex hormones. Among them, inflammation is considered to be the main pathogenetic mechanism of dry eye. The inflammation in dry eye is usually attributed to an immune abnormality, which often has an impact on the entire tear film functional unit. The main pathological features are characterized by the hypertonicity of the tears, a decrease in the volume of the tears and tear film instability. In turn, all of the features are intensifying the ocular surface inflammation.
Buddleja officinalis Maxim is a natural medicinal plant specially used for the treatment of eye diseases. KAE, the active ingredient of B. officinalis Maxim, is a kind of natural polyhydroxy flavonoids in various plants. 4 It has been proven to have properties of anti-inflammatory, anti-oxidant, anti-viral, anti-tumor, cardioprotection, neuroprotection, and bone protection.5–9 The anti-inflammatory effects of KAE have been extensively shown in various tissues and organs in the human body. For example, KAE could reduce CD3+ T cell infiltration and gene expression of primary pro-inflammatory cytokines (such as interleukin (IL)-6, IL-17A, and tumor necrosis factor (TNF-α)) in psoriatic skin lesions. KAE also downregulates the pro-inflammatory nuclear factor kappa B (NF-κB) signaling in the skin. 10 KAE reduces the expression of pro-inflammatory cytokines (TNF-α, IL-1β, and IL-8) and the production of IL-8 in AGS cells in response to the Helicobacter pylori-caused inflammatory response, as well as inhibits intestinal inflammation.11,12 Furthermore, several studies have reported the underlying mechanism of KAE reducing inflammation. It has been shown that KAE significantly reduces cisplatin-induced apoptosis and inflammatory responses by lowering the STING/NF-κB pathway. 13 KAE improves the progression of chronic airway inflammation by reducing levels of IL-5, IL-13, and granulocyte-macrophage colony-stimulating factor (GM-CSF), as well as eosinophil counts in bronchoalveolar lavage fluid. 14 However, there are few reports on the anti-inflammatory mechanism of KAE in the eye. Considering the high demand for ocular surface diseases for natural medicines, we tried to apply KAE with significant anti-inflammatory effects for treating ocular surface inflammation.
As one of the four subfamilies of the mitogen-activated protein kinase (MAPK) pathway, p38 MAPK plays an important role in inflammation and apoptosis. Studies have found that KAE significantly inhibits the protein expression of interleukin (IL)-1β-induced inflammatory mediators (such as inducible nitric oxide synthase and cyclooxygenase-2), and inhibits IL-1β-induced common matrix-degrading enzymes, including matrix Metalloproteinase MMP-1, MMP-3, and MMP-13. 15 Therefore, the current study, which uses human corneal epithelial cells (HCECs) as research objects, intends to investigate the changes of target genes involved in the p38MAPK signaling pathway, and to observe the effect of kaempferol on hypertonic-induced HCECs.
Materials and Methods
Instruments and Reagents
The instruments and reagents used in this study are as follows: fluorescence quantitative RCP instrument (PikoReal96; Thermo Fisher Scientific, Waltham, MA, USA), fluorescent PCR plate (SPL0960; Thermo), electrophoresis instrument (DYY-2C; Beijing Liuyi Biotechnology Co., Ltd, China), inverted biological microscope (DSZ2000X; Beijing Zhongxian Hengye Instrument, China), multifunctional enzyme label analyzer (MB-530; Huisong Technology, China), transfer membrane instrument (Beijing Liuyi), heating carbon dioxide incubator (DH-160I; Santeng Instruments), KAE (P0013; Pure one), Tunel kit (40306ES50; Yisheng Biotechnology, China), human epidermal growth factor (EGF; AF-100-15-500; Peprotech, USA), SuperECL Plus ultra-sensitive luminescent fluid (K-12045-D50; Advansta), and fluorescence microscope (BA410T; Motic, Fujian, China). HCECs were supplied by the cell bank of Ophthalmology Research Center, Xiamen University.
Cell Culture and Grouping
HCECs were cultured (37 °C, 5% CO2) in DEME/F12 medium containing 10% fetal bovine serum (FBS), human EGF, and 1% double antibody in an incubator with saturated humidity. The cells were randomly allocated into the normal group (cell cultured in the DEME/F12 medium containing 10% FBS, human EGF, and 1% double antibody), model group (cells cultured in 450 mOsm/L medium), vehicle group (cells cultured in 450 mOsm/L medium + dimethyl sulfoxide (DMSO)), and KAE group (cells treated with KAE of different concentrations for 48 hours after 24 hours culturing in 450 mOsm/L medium).
Cell Counting Kit-8 (CCK-8) Detection
The cells in logarithmic growth were inoculated in 96-well plates until cell adherence. Cells in different groups received the corresponding treatments, with three multiple wells per group. HCECs in the vehicle group were treated with 2.6 μL DMSO in the hypertonic medium (450 mOsm/L) for 24 hours and 48 hours; HCECs in the KAE groups were treated with different concentrations of KAE (0 μM, 20 μM, 40 μM, 80 μM, 160 μM, 320 μM, 640 μM, and 1280 μM) in the hypertonic medium (450 mOsm/L) for 24 hours and 48 hours. KAE was prepared as follows. KAE has a molecular weight of 286.24, and 20 mg KAE was added to 139.7 μL of DMSO to dissolve into 500 mM mother liquor 1; 8 μL of mother liquor 1 was added to 92 μL of DMSO to obtain mother liquor 2, with concentration of 40 mM.
The above-grouped cells were detached, counted, and then seeded (1*104 cells/well) in 96-well plates, 100 μL per well, with three replicate wells set. KAE was added for the corresponding time. Each well was added with a 10 μL CCK-8 solution. The drug-contained medium was removed, and each well was added with 100 μL of CCK-8-contained medium. After 4-hour incubation (37 °C, 5% CO2), the absorbance (OD) value at 450 nm was analyzed using a Bio-Tek microplate reader.
TdT-mediated dUTP Nick-End Labeling
The HCEC slides after each treatment were prepared and fixed in 4% paraformaldehyde for 30 minutes. Next, 100 μL of Proteinase K working solution was added to each sample for 20 minutes at 37 °C, followed by three phosphate-buffered saline (PBS) washes, each for 5 minutes. Next, 100 μL 1 × Equilibration Buffer (1:5) was added to each sample for 10–30 minute incubation at room temperature. After that, most of the 1 × Equilibration Buffer was washed off with absorbent paper. Cell slides were then added with 50 μL TdT incubation buffer at 37 °C for 60-minute treatment, followed by three PBS washes. The above two steps were protected from light. The nucleus of the HCEC slides was stained (room temperature, 10 minutes) with DAPI working solution away from light, and then underwent PBS washes three times. Finally, cell slides were mounted with buffered glycerol and photographed under a fluorescence microscope.
Immunofluorescence Staining
The HCEC slides after each treatment were prepared and fixed in 4% paraformaldehyde for 30 minutes. Next, the slides were permeabilized (37 °C, 30 minutes) using 0.3% Triton and blocked (37 °C, 60 minutes) with 5% bovine serum albumin (BSA). After that, the slides were added with the primary antibody Ki67 rabbit antibody (1:50) for overnight incubation at 4 °C and washed with PBS three times for 5 minutes each on the next day. Next, the HCECs were incubated with the Rabbit-IgG-labeled fluorescent secondary antibody for 90 minutes at room temperature. The nuclei were stained with DAPI working solution at room temperature for 10 minutes and washed with PBS three times, 5 minutes for each. Finally, the cell slides were mounted with buffered glycerol and photographed under a fluorescence microscope.
RT-PCR Detection
The total RNA of HCECs in each group was extracted using the TRIzol kit, with the total mRNA of the tissues used as the template. cDNA was reverse-transcribed, and gene expression was detected by SYBR. The actin served as an internal reference gene. The amplification system consisted of 1 μL upstream primer, 1 μL downstream primer, 2 μL cDNA,15 μL 2X SYBR GREEN PCR Master Mix, and 11 μL enzyme-free water. The amplification conditions were 95°C for 10 minutes, followed by 40 cycles of 95°C for 16 seconds, and 60°C for 30 seconds. The sequence of the target gene was searched from NCBI, and primers were designed using the Primer5 software.
Western Blot
The total protein of the treated HCECs was extracted using RIPA lysate. The protein concentration was determined by the bicinchoninic acid (BCA) reagent. After gel preparation, the protein was separated by electrophoresis. Next, the gel was cut and proteins were transferred to NC membranes. The membranes were sealed with 5% skimmed milk powder at room temperature for 90 minutes, and then incubated (4°C, overnight) with primary antibodies: rabbit anti-TNF-α (1:1000), rabbit anti-IL-6 (1:1000), and rabbit anti-P38 (1:1000), with β-actin as the internal control. After that, horseradish peroxidase (HRP)-labeled goat anti-mouse (1:5000) and goat anti-rabbit (1:6000) IgG antibodies were added for incubation (room temperature, 90 minutes). Enhanced chemiluminescence (ECL) was used for luminescence development, and Image-Pro plus 7.0 was used for gray value analysis.
Statistical Analysis
The data in this study were analyzed using the SPSS 26.0 software, Graphpad Prisim5.0, and Imagine J. The two-sided test was performed. P < .05 represented a statistically significant difference. Data were expressed as mean ± standard deviation (±SD). For data with normal distribution and uniform variances, the one-way analysis of variance (ANOVA) was performed; if the variance was not uniform, a corrected one-way ANOVA would be used. For data that did not conform to normality and the variance was uniform, the non-parametric test was used; if the variance was not uniform, the Turkey method was used for comparison among multiple groups.
Results
CCK-8 Detection
KAE is non-toxic to HCEC and has the effect of enhancing cell viability. In this experiment, 80 μM was selected as the experimental concentration of KAE. After treatment with different concentrations of KAE for 24 hours and 48 hours, the CCK-8 method was used to detect the KAE effect on HCEC growth. The results showed that KAE within the concentration range of 0–80 μmM promoted the growth of HCEC in a concentration-dependent manner; KAE with a concentration of 80 μM caused the highest cell activity (the 24-hour average was 1.22 ± 0.04; the 48-hour average was 1.89 ± 0.02). When treated with KAE within a concentration range of 80–1280 μM, HCEC showed a decreasing growth trend as the concentration increased (Figure 1).

HCEC viability after treatment with different concentrations of KAE for 24 hours and 48 hours was detected. HCECs were treated with different concentrations of KAE. Cell viability after 24/48 hours was observed under a microscope and the OD value at 450 nm was analyzed with a Bio-Tek microplate reader.
TUNEL Test
As revealed by TdT-mediated dUTP Nick-End Labeling (TUNEL) immunofluorescence staining results, HCECs had a significantly increased apoptotic rate in the 450mOsm hypertonic environment, and KAE intervention could notably decrease the apoptotic rate (P < .05); KAE effectively inhibited hypertonic-induced apoptosis (Figure 2).

Effects of KAE on hyperosmolarity-triggered HCEC apoptosis were detected. TUNEL immunofluorescence staining was performed in HCECs treated with normal medium, 450 mOsm/L, vehicle, 450 mOsm/L + DMSO, and 450 mOsm/L + KAE (A). TUNEL fluorescence graphical representation of each group was displayed, and the image was 400 times magnification (B). Statistical analysis was performed using the SPSS26.0 software (*P#.05, **P#.01, ***P#.001, and ****P#.0001) (n ≥ 3).
Immunofluorescence Assay
According to the results of Ki67 fluorescence, HCEC proliferation was remarkably inhibited in the hypertonic environment (450 mOsm/L). KAE intervention could notably improve HCEC proliferation (Figure 3).

Effects of KAE on the proliferation of HCECs in the hypertonic environment were detected. Representative Ki67 immunofluorescence staining of HCECs treated with normal medium, 450 mOsm/L (69 mM NaCl), vehicle, 450 mOsm/L + DMSO, and 450 mOsm/L + KAE was displayed (A). Fluorescence graphical representation of Ki67 fluorescent staining histogram of IOD/AREA mean value of each group was displayed, the image was 400 times magnification (B). Statistical analysis was performed using the SPSS26.0 software (*P#.05, **P#.01, ***P#.001, and ****P#.0001) (n ≥ 3).
KAE can Significantly Reduce the Production of Inflammatory Mediators through the p38MAPK Signaling Pathway
RT-PCR Detection
PCR results showed that KAE can effectively inhibit inflammatory cytokines. The mRNA relative expressions of TNF-α, IL-6, and p38 in the hypertonic environment (450 mOsm/L) were remarkably increased (P < .05), which could be effectively decreased by KAE intervention for 48 hours (Figure 4).

Effects of KAE on the hypertonic environment-induced ocular surface inflammation were detected by real-time PCR. Graphical representation of the quantified real-time PCR depicted the relative mRNA expressions of TNF-α, IL-6, and p38. TNF-α, IL-6, and p38 mRNA transcript levels in HCECs with different treatments were compared, with HCECs treated with DMSO as the vehicle control. Statistical analysis was performed using the SPSS26.0 software (*P#.05, **P#.01, ***P#.001, and****P#.0001) (n ≥ 3).
Western Blot Test
As revealed by Western blot analysis results, KAE can effectively suppress the expression of inflammatory cytokines. TNF-α, IL-6, and p38 protein levels in the hypertonic environment (450 mOsm/L) were significantly increased (P < .05), while KAE intervention for 48 hours could dramatically decrease the protein level of these inflammatory cytokines (Figure 5).
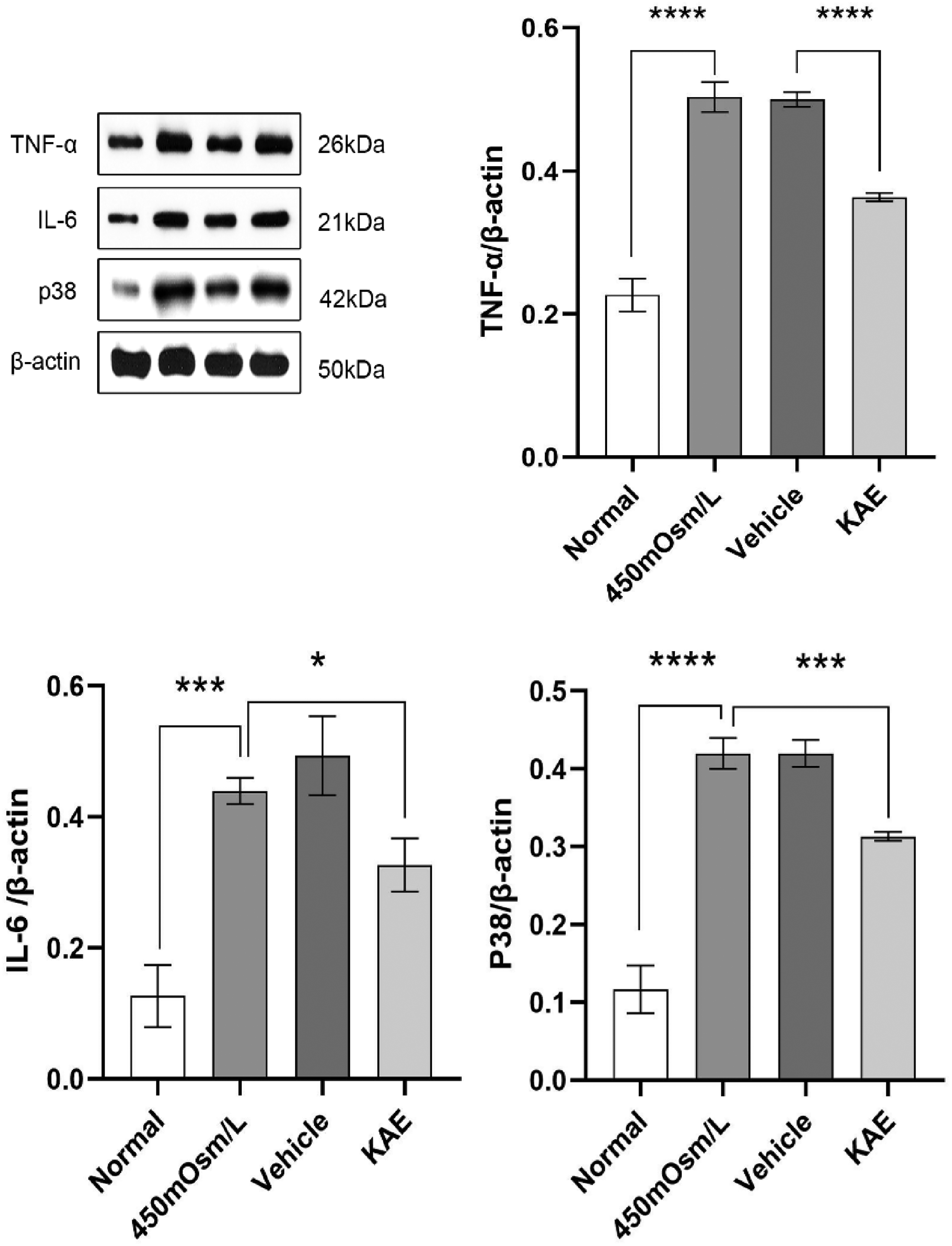
Effects of KAE on the hypertonic environment-induced ocular surface inflammation were detected by western blot. Representative western blot results of TNF-α, IL-6, and p38 under different treatments were displayed (A). Densitometric analysis of the protein levels of TNF-α (B), IL-6 (C), and pP38 (D) regarding the GAPDH levels was plotted graphically. TNF-α, IL-6, and p38 protein levels in HCECs with different treatments were detected, with those DMSO as the vehicle control. Statistical analysis was performed using the SPSS26.0 software (*P#.05, **P#.01, ***P#.001, and ****P#.0001) (n ≥ 3).
Discussion
Inflammation, as an important process of human healing response, plays a crucial role in the pathogenesis of dry eye. The activation of the p38MAPK pathway is one of the common pathways for ocular surface inflammation. Accumulating studies16–19 have shown that KAE can inhibit the expression of inflammatory cytokines in cells and the phosphorylation of protein kinases activated by various extracellular stimuli through the p38MAPK signaling pathway. However, little is known about the effect of KAE on HCEC and the underlying mechanism.
As has been evidenced previously, hypertonic pressure-induced experimental dry eyes in mice can stimulate the expression and production of IL-1β, TNF-α, and MMP-9, as well as activate the ocular surface MAPK signaling pathway. 20 IL-1α, IL-1β, and TNF-α mRNA expressions in mice with experimentally induced dry eyes are also significantly increased. At the same time, the phosphorylation of JNK1/2, ERK1/2, and p38MAPK in the corneal epithelium is stimulated, and the MAPK signal pathway of corneal epithelial cells is activated. 21 Additionally, in vitro experiments also prove that hypertonicity exerts great effects on inflammation and ocular surface damage. The hypertonic environment affects the morphology of corneal epithelial cells and reduces cell viability and even death. 22 A study has found that the expressions of TNF-α, IL-1β, IL-6, and IL-8, as well as autophagy-related genes (Ulk1, Beclin1, Atg5, and LC3B) in primary HCECs cultured in hypertonic medium (450 mOsm/L) are remarkably increased. 23 In this experiment, the hypertonic medium was used to stimulate HCEC inflammation. It was observed that the hypertonic environment caused the elevation of the production and expression levels of TNF-α, IL-6, IL-6, and p38, showing a long-lasting effect. This effect may be attributed to the hypertonicity promoting the expression of inflammatory mediators, leading to abnormal tear secretion and unstable tear film, which in turn promotes inflammation, forming a vicious circle.
In “Thunder Lord concocts herbal medicine comments,” B. officinalis Maxim is recorded as sweet, slightly cold in nature, and non-toxic, and it enters the liver meridian. It mainly treats bluish blindness and corneal opacity, conjunctivitis redness and eye discharge with tears, drooping pannus, as well as children's chickenpox and keratomalacia; it is soaked in wine overnight, steamed with honey, and dried in the sun. It is specially used in the liver meridian. Therefore, there is no other longness beyond the treatment of eyes, and the essential medicine for ophthalmology.” 24 “Yu Jiu Yao Jie” has recorded that B. officinalis is sweet and slightly cold; it enters the liver meridian, clears the lungs and moistens dryness, improves vision and removes nebula; it clears the liver and treats redness, swelling, corneal opacity, and vision obstruction. 25 According to Liu et al, 26 Erxian Decoction plus B. officinalis could effectively prolong tear film breakup time (BUT), and improve the Schirmer test (SIT) and clinical symptoms of patients with menopausal dry eye. It is extensively believed that B. officinalis clears heat, enriches yin, and improves vision without hurting the yang, which is an essential medicine for treating dry eyes and dim vision.
Accumulating studies have found that Buddleja officinalis can inhibit lacrimal gland cell apoptosis, reduce inflammatory factor expressions in the lacrimal gland cells, and improve the ultrastructure of the lacrimal gland to maintain the basal secretion of tear fluid. Among them, the total flavonoids of Buddleja officinalis play the most important role.27–29 KAE, a natural flavonol found in different plant species, has been evidenced to possess effective anti-inflammatory properties. 1 With potent anti-inflammatory activities, KAE has been used to treat many acute and chronic inflammation-induced diseases, including intervertebral disc degeneration and colitis, as well as postmenopausal bone loss and acute lung injury.
In this study, extracellular stimulation (sodium chloride hypertonic solution) increased the expression of inflammatory mediators (TNF-α and IL-6) of HCEC. Pro-inflammatory factors and other in vitro stimulations led to p38 activation, and p38 can be selectively activated by MKK3 and MKK6. Although whether MKK3 or MKK6 is implicated in the specific mechanism in the in vitro model of hypertonic-induced dry eye is worthy of further study and exploration, KAE certainly plays a regulatory role in the activation of the p38MAPK pathway by this stress. KAE could decrease inflammatory factor expressions, promote cell proliferation, and control cell apoptosis; KAE may even reverse the damaged state of HCECs after inflammation.
Kaempferol Processing.

RT-qPCR Primer Sequence of Each Gene.

Footnotes
Acknowledgments
This study was supported by the National Natural Science Foundation of China (Nos. 30772824 and 81574031), the Science and Technology Department of Hunan Province (No. 2015SF2016-6), the open Foundation of Hunan Engineering Technology Research Center of Medicinal and Food Homology Functional Food (2018YSTY04), Construction Project of Hunan Provincial Key Laboratory of TCM Prevention and Treatment of ENT Diseases (2017TP1018), Academician Liu Liang workstation Scientific Research Guidance Project in 2021 (21YS002) and Key Project of Scientific Research Fund of Education Department of Hunan Province (21A0238).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the National Natural Science Foundation of China, Academician Liu Liang workstation Scientific Research Guidance Project in 2021, the open Foundation of Hunan Engineering Technology Research Center of Medicinal and Food Homology Functional Food, Construction Project of Hunan Provincial Key Laboratory of TCM Prevention and Treatment of Ent Diseases, Key project of Scientific Research Fund of Education Department of Hunan Province (grant number Nos. 30772824, 81574031, 21YS002, 2018YSTY04, 2017TP1018, and 21A0238).
Date Availability
The data used to support the findings of this study are available from the corresponding author upon request.
