Abstract
Introduction
Polycystic ovary syndrome (PCOS) is a common endocrine disease among reproductive women and exhibits a snowballing threat to their health. 1 According to the standardized diagnosis, the incidence of PCOS has increased to 18% in the United States. 2 PCOS is characterized by polycystic ovary, hyperandrogenism, ovulatory dysfunction and even develops into anovulation and infertility. 3 Besides, most patients suffering from obesity, insulin resistance, and abnormal lipid metabolism have a greater risk than normal of developing T2DM, angiocardiopathy, atherosclerosis, and gynecological cancers. 4 The etiology and pathogenesis of PCOS are complex and have not been elaborated clearly. Current medical treatment includes contraceptives, progestin, antiandrogen medication, and Metformin. 5 However, limited efficacy and serious side effects, such as nausea, vomiting, and increased risk of cardiovascular disease, afflict the patients. Therefore, the disorder is plagued by lacking optimal treatment. Recent research proposed that PCOS was a chronic low-grade inflammation with abnormally increased inflammatory markers, including TNF-α, CRP, IL-6, IL-18, and MCP-1. 6 This inflammation is strongly associated with obesity, hyperandrogenism, and insulin resistance. 7 Therefore, eliminating the inflammation might be beneficial to normalizing the various biochemical indicators and relieve the symptoms of PCOS.
TNF-α, which plays an essential role in inflammation, is secreted by adipocytes and is regarded as a significant mediator of IR and androgen expression. 8 Therefore, inhibiting the activity of TNF-α is a potential therapeutic strategy for PCOS. ETA, a TNF-α inhibitor, significantly reduced the weight and downgraded androgen in PCOS rats. 9 Moreover, attention has been attracted to a TNF-α inhibitor for other inflammatory disease treatments. 10 In spite of various TNF-α inhibitors existing on the market, their serious side effects impede their drug use. 11 Exploring and developing natural therapeutic products are necessary for clinical practice.
The dried root of Paeonia lactiflora Pallas (Baishao; radix paeoniae alba, RPA) is classified as the top-grade herbal medicine in Shenong's Herbal Classic, which is the first monograph on materia medica in the history of TCM. 12 RPA has a bitter and sour taste and belongs to “liver and spleen channels.” RPA has the effects of nourishing blood and liver, regulating menstruation, and relieving pain. The chemical constituents of PRA include terpenoids, polyphenolic compounds, volatile oil, monoterpene glycosides, galloylglucoses, and phenols, measured by UPLC and TOF-MS. 13 The major pharmacological effects of RPA include anti-inflammatory, immune adjustment, and anti-oxidative activities. 14 Therefore, RPA has been widely used to treat menstrual diseases and different inflammatory states, including endometritis and cholecystitis. 15 A nationwide matched cohort study revealed that RPA was the common ingredient in products for decreasing the incidence of T2DM in PCOS patients. 16 In an in vivo study, PRA decreased toxic alkaloid absorption into the brain and inhibited oxidative stress, which demonstrated a protective effect of PRA against neurotoxicity in Strychnos alkaloid brain damaged rats. 17 Furthermore, research has found that paeoniflorin could decrease the expression of CYP17A1 and reverse testosterone elevation in primary theca cells of mice. 18 RPA extracts also inhibited LPS-induced inflammatory reaction 19 and decreased the inflammatory response in MPGN rats. 20 However, it is still unknown whether RPA alleviates PCOS by suppressing TNF-α expression and inhibiting inflammation.
In the present study, we primarily utilized network pharmacology to screen possible effective ingredients of RPA in PCOS treatment. Subsequently, targets genes and pathways of effective ingredients were clustered using GO and KEGG. 21 Subsequently, to explore the optimal ingredient binding with TNF-α, molecular docking was applied to assess the binding capacity between the ingredients and TNF-α. Briefly, this study aimed to identify the anti-inflammatory compounds of RPA and provide the theory of treatment in PCOS.
Methods
Study Procedure
A scheme of the pharmacology and molecular docking workflow is shown in Figure 1. Compounds of RPA were extracted from the TCMSP database. 22 PCOS correlated disease targets were acquired through GeneCards (https://www.genecards.org/), 23 OMIM (https://www.omim.org/), 24 Pharmgkb (https://www.pharmgkb.org/), 25 TTD (http://db.idrblab.net/ttd), 26 and Drug Bank (https://www.drugbank.ca). 27 After finding these targets, RPA–PCOS possible targets were filtered by the DAVID database. 28 Subsequently, Cytoscape 29 was used to construct an RPA-ingredients-PCOS-target network. Molecular docking was presented by CB-Dock ((http://cao.labshare.cn/cb-dock/). 30

Scheme of pharmacology and molecular docking workflow.
Extracting Active Ingredients of RPA
The potential active ingredients of RPA were searched and extracted from TCMSP. The filter conditions were oral bioavailability (OB) >30% and drug-likeness (DL) >0.18. Thirteen ingredients were collected, and the 2D structures of 8 of the potentially active and PCOS related ingredients of RPA were downloaded from PubChem (http://pubchem.ncbi.nlm.nih.gov). 31 2D structures were transformed into 3D structures with structure optimization in Chemdraw 3D Ultra software. 32
Clustering of RPA and PCOS Correlated Target Genes
The target genes of RPA were collected from TCMSP, and the normalizing names of these genes were calibrated in Uniprot (https://www.uniprot.org/). 33 PCOS-associated target genes were gathered from 5 databases, GeneCards, OMIM, Pharmgkb, TTD, and DrugBank. They were consolidating RPA-correlated potential genes for PCOS treatment through the Venn diagram (http://bioinformatics.psb.ugent.be/webtools/Venn/). 34
Constructing PPI Network of RPA–PCOS-Potential Target Genes
The RPA–PCOS potential target genes were entered into the STRING database (https://string-db.org/) 35 and the organism selected was “Homo sapiens, confidence = 0.900.” Subsequently, a PPI network was constructed to predict the neighborhood, co-expression, and fusion among potential target genes. Each node and edge represent a protein and a functional correlation, respectively.
Visualized Analysis of RPA and PCOS-Potential Target Genes Network
Cytoscape software (version 3.9.1) was used to visualize the correlation of RPA–PCOS-potential target genes. One node symbolically signified a compound or a target gene in the visual representation. Meanwhile, each line symbolizes an intermolecular correlation between one compound and one target gene.
Bioinformatics and Enrichment Analysis
Entrez ID of RPA–PCOS-potential target genes were converted by R packages “org.Hs.egdb version 4.1.2.” GO biological functions annotation and the KEGG Pathway analyses were visualized using the R packages “clusterProfiler,” “pathview,” and “DOSE.” 36
Accessing Combining Capacity Between Active Ingredients and Target Genes by Molecular Docking
Eight potential active ingredients were used to proceed to molecular docking with TNF-α receptor. Accessing combining capacity was conducted in CB-Dock, which is an available online docking tool (http://cao.labshare.cn/cb-dock/). 30 CB-Dock could automatically match the optimal docking pocket and list the Vina scores and related active configuration parameters. Vina scores could preliminarily assess the stability of the combining capability between the selected ingredient and the target protein. The lower the Vina score the more stable is the structure.
Results
Extracting Active Ingredients of RPA
Eighty-five active ingredients of RPA were found from the TCMSP database. After screening, 13 of these matched the required conditions (OB > 30% and DL > 0.18); these are shown in Table 1. The 13 ingredients were paeoniflorigenone, palbinone, mairin, paeoniflorin, kaempferol, ( + )-catechin, β-sitosterol, sitosterol, lactiflorin, albiflorin_qt, benzoyl paeoniflorin, paeoniflorin_qt, and 11α,12α-epoxy-3β-23-dihydroxy-30-norolean-20-en-28,12β-olide.
The Information of Screening Ingredients in RPA.

Oral bioavailability.
Drug-likeness.
Clustering of RPA and PCOS Correlated Target Genes and Construction of PPI Network
After eliminating repetitions, 3993 target genes were gathered for PCOS therapy from 5 databases. Meanwhile, 91 target genes of RPA were collected using the TCMSP database. The number of unique target genes of RPA was 68. Clustering RPA and PCOS related target genes led to the identification of 54 possible target genes, shown in Figure 2A and Table 2. STRING database was utilized to build a PPI network map of the 54 potential target genes represented in Figure 2B, and a histogram was used to show the clustering results of the top 30 target genes in Figure 2C. The histogram revealed that the target genes contained retinoid x receptor a (RXRA), serine/threonine-protein kinase (AKT1), tumor necrosis factor (TNF), and cytochrome P450 1a1 (CYP1A1).

The target genes and PPI network of RPA–PCOS key target genes. (A) The potential target genes are shown in a Venn diagram. (B) The PPI network of 54 key target genes. (C) The top 30 genes in the PPI network were counted and listed in the histogram.
The 54 Potential Target Genes of RPA for PCOS Treatment.

Visualized Analysis of RPA and PCOS-Potential Target Genes Network
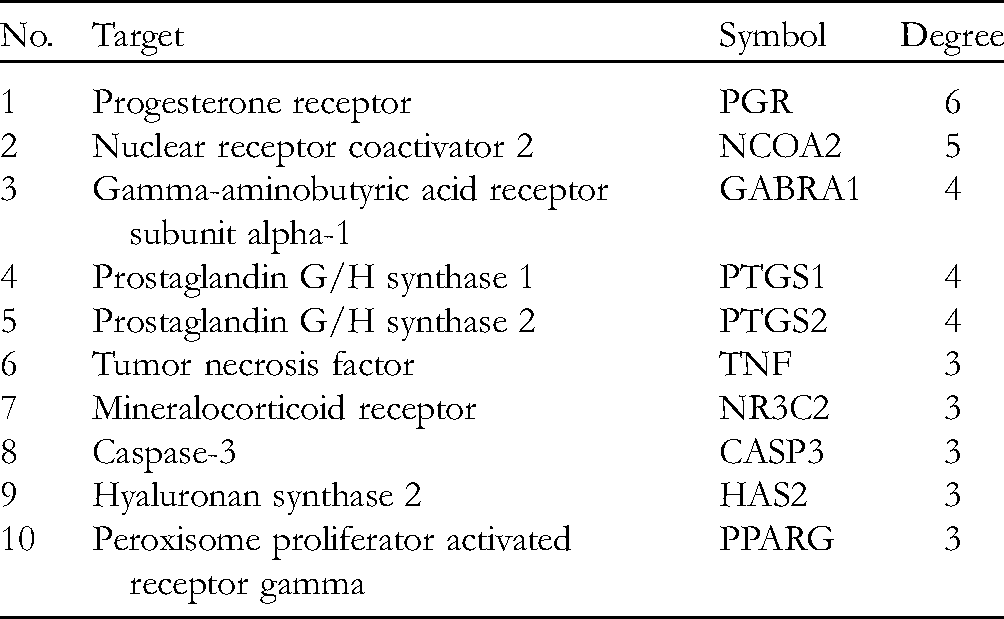
The RPA–PCOS potential target gene visual network is shown in Figure 3. The network includes 64 nodes and 132 lines, which indicate the complex association of the RPA–PCOS-potential target gene. In this network, the key target genes with higher degree values (degree >4) were progesterone receptor (PGR), nuclear receptor coactivator 2 (NCOA2), gamma-aminobutyric acid receptor subunit alpha-1 (GABRA1), prostaglandin G/H synthase 1 (PTGS1), and prostaglandin G/H synthase 2 (PTGS2), which are listed in Table 3.
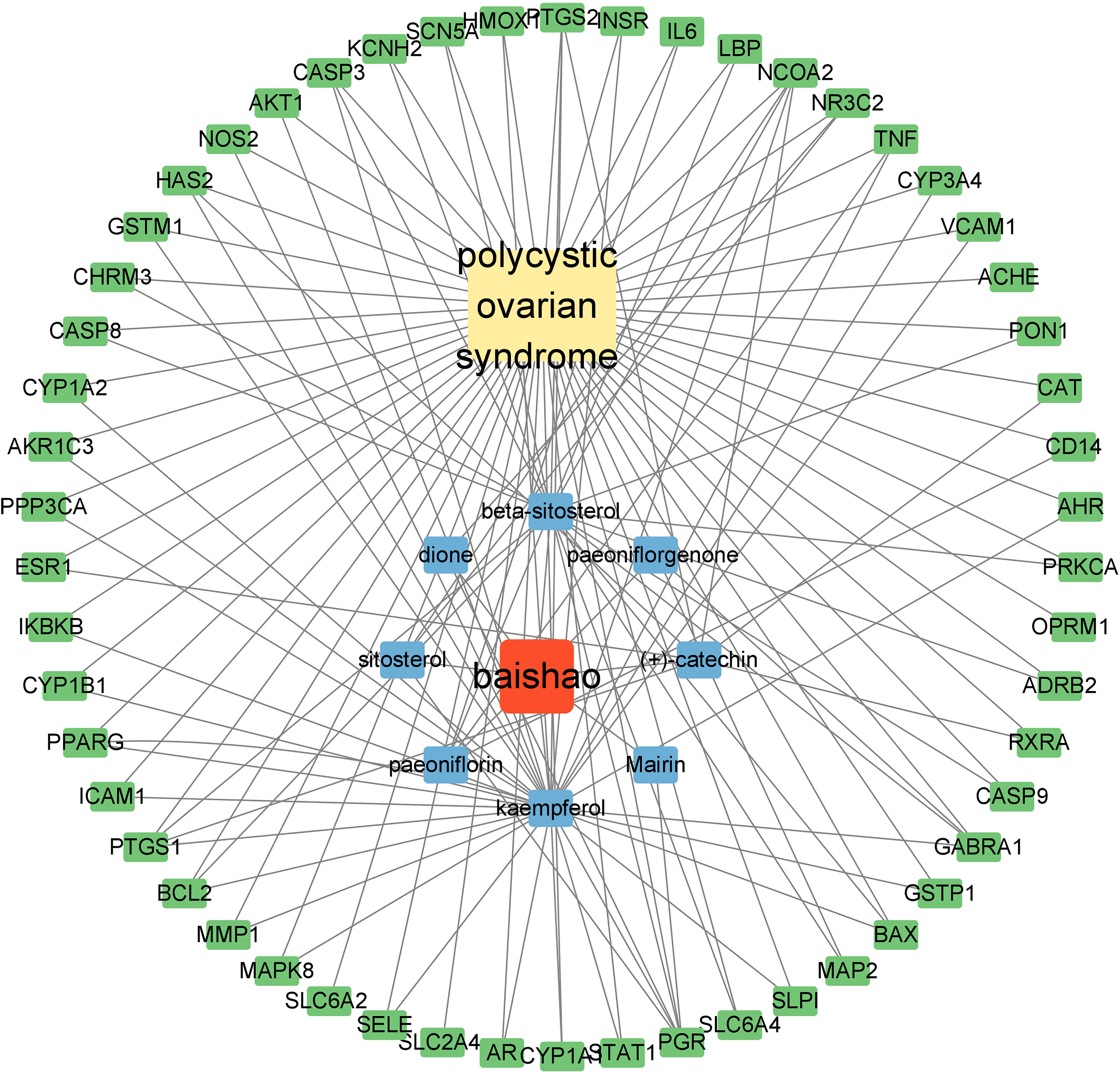
The visual network of ingredients-disease-target genes. The yellow node indicates PCOS. The red node represents RPA. The blue and green nodes indicate bioactive ingredients and target genes, respectively. Interaction of each node is shown with gray lines.
The top 10 Target Genes in the Visual Network (Degree >3).
GO and KEGG Pathway Enrichment Analysis
The Entrez IDs of 54 target genes are listed in Table 2. For GO analysis, the 71 biological functions of genes were gathered (P < .05). The histograms and scatterplots representing the top 20 gene biological functions were significantly gathered using GO analysis (Figure 4A and Supplementary Figure 1). The therapeutic effect of RPA on PCOS involved heme binding, amide binding, tetrapyrrole binding, oxidoreductase activity, acting on paired donors, with incorporation or reduction of molecular oxygen and peptide binding, which were the key biological functions. Furthermore, KEGG pathway enrichment uncovered a deeper mechanism in RPA for PCOS therapy. The top 20 signaling pathways are contained in the scatterplots and histograms shown in Figure 4B and Supplementary Figure 2. Lipid and atherosclerosis, diabetic complications related to the AGE-RAGE signal pathway and TNF signal pathway were the crucial signal pathways that were correlated with the protective mechanism of RPA in PCOS. The crucial target genes that correlated with the TNF signal pathway in RPA–PCOS are shown in Figure 5.

Clustering analysis of GO (A) and KEGG (B).

The network of the TNF signal pathway and the target genes of RPA for PCOS treatment are shown in red.
Binding Capacity Between the Key Ingredients With TNF-α Receptor Using Molecular Docking
Eight key ingredients were used to access the binding affinity with the TNF-α receptor, and the results are represented in Table 4. Paeoniflorigenone had lower Vina scores than the other 7 components. This result manifested the most stable binding capacity between paeoniflorigenone and TNF-α with the lowest Vina score. Thus, paeoniflorigenone might be the natural product acting as a TNF inhibitor. The 3D docking models of the 8 components with TNF-α are shown in Figure 6.

The 3D structure of the molecular docking of (A) β-sitosterol, (B) ( + )-catechin, (C) palbinone, (D) sitosterol, (E) mairin, (F) kaempferol, (G) paeoniflorigenone, and (H) paeoniflorin with TNF-α.
Molecular Docking Parameters and Results of TNF-α and 8 Active Ingredients of RPA.

Coordinates of docking pocket center.
The size in the X, Y, and Z directions of the docking pocket.
Discussion
Polycystic ovary syndrome is an intricate and unmanageable health issue, which afflicts more than 5% of premenopausal women worldwide. 37 The exact etiology and underlying mechanism have not been illustrated. 38 Currently, PCOS has been regarded as chronic low-grade inflammation. Despite this, there are no effective anti-inflammatory drugs for PCOS. Therefore, the efficient treatment of PCOS is still impeded. In China, treatment based on syndrome differentiation is the principle of TCM, and Chinese herbs and formulas have been widely used to relieve symptoms in the PCOS process.
Baishao is frequently used to relieve PCOS-related symptoms including decreasing the level of LH, T, and E2 and improving ovarian function. 39 In this study, we filtered 8 ingredients of RPA, which showed good anti-inflammatory effects. The 3 monoterpenes paeoniflorigenone, paeoniflorin, and albiflorin 40 showed noteworthy inhibiting effects on TNF-α-induced damage of endothelial cells. 41 Paeoniflorin also recuperated the abnormal signal pathway related to inflammation and regulated immune cytotic function in several disorders such as rheumatoid arthritis 42 and psoriasis. Albiflorin attenuated anxiety, neuropathic aches, and depressive disorder by reducing the activity of NLRP3 inflammasome in rats. 43 β-Sitosterol is a natural phytosterol with potential effects against various disorders. It alleviated adipocyte triggered inflammation and inhibited the level of pro-inflammatory factor such as IL-6 and TNF-α through decreasing the expression of JNK and the IKKβ/NF-κB signaling pathway. 44 Furthermore, in the HFD-induced intestinal inflammation model, β-sitosterol restrained the transposition of NF-κB and blocked the combination of LPS with TLR4. 45 Kaempferol significantly decreased the level of I-κB and NF-κB in the LPS-induced IBD vitro model. 46 For other compounds, (+)-catechin could regulate the expression of TLR2/4, MAPK, and NF-κB in mice infected with Porphyromonas gingivalis. 47 In brief, the widespread use of Baishao and the anti-inflammatory effects of its ingredients indicated the possibility of these components for PCOS treatment.
RPA for PCOS treatment is a multicomponent and multitargeted therapeutic strategy. There were 54 proteins related to RPA and PCOS in this study, and, simultaneously, different degrees of connection and interaction existed between these proteins. According to the visual network, the potential and vital targets of RPA for PCOS contained PGR, NCOA2, GABRA1, PTGS1, PTGS2, TNF, NR3C2, CASP3, HAS2, and PPARG. These potential targets are correlated with regulating the level of gonadal hormone, inflammation, and oxidative stress. In GO and KEGG enrichment analysis, 71 biological functions and 132 pathways were potentially associated with the mechanism of RPA for PCOS treatment. Inflammation and TNF-α were important biofunctions and pathways in GO and KEGG clustering analysis. Meanwhile, research revealed that anti-inflammation would be a crucial mechanism in PCOS. 7
The molecular docking parameters and results indicated that paeoniflorigenone was the best ingredient of RPA for binding to TNF-α protein. The previous studies indicated that the level of TNF-α was associated with IR and inflammation in PCOS patients.48,49 In another study, paeoniflorigenone inhibited apoptosis and increased autophagy in human HNSCC cells by regulating the PI3K/AKT signaling pathway. 50 Moreover, paeoniflorigenone was the top compound with the best oral bioavailability in Xijiao Dihuang Tang. The formula downregulated the levels of pro-inflammatory factors IL-1β and TNF-α in Kawasaki disease mice. 41 This research revealed that paeoniflorigenone possessed various bioactive functions including anti-inflammation and inhibiting the expression of TNF-α. The present study expanded the scope of basic research on paeoniflorigenone.
Conclusion
RPA is a preferred Chinese herb in PCOS treatment, which has been verified in clinical practice and previous research. The GO and KEGG analysis of RPA bioactive ingredients and PCOS target genes were explored through network pharmacology. Paeoniflorigenone was the optimal ingredient for binding to TNF-α using the molecular docking approach. In a word, paeoniflorigenone could be the optimal compound of RPA acting as a natural TNF-α inhibitor for PCOS treatment.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221129136 - Supplemental material for Identification of the Anti-Inflammatory Compound, Paeoniflorigenone, in Radix Paeoniae Alba for the Treatment of Polycystic Ovary Syndrome Through Network Pharmacology and Molecular Docking
Supplemental material, sj-docx-1-npx-10.1177_1934578X221129136 for Identification of the Anti-Inflammatory Compound, Paeoniflorigenone, in Radix Paeoniae Alba for the Treatment of Polycystic Ovary Syndrome Through Network Pharmacology and Molecular Docking by Juntong Li, Gaoxiang Wang, Shufang Chu, Deliang Liu, Hengxia Zhao and Huilin Li in Natural Product Communications
Footnotes
Authors’ Note
Juntong Li and Gaoxiang Wang contributed equally to this work. LJT and WGX collected the data and wrote the original draft. CSF, LDL, ZHX, and LHL revised the manuscript. The relevant data and code used in this study are accessible upon reasonable request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NO.82104759), and the Guangdong Natural Science Foundation (2020A1515010775).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
