Abstract
In the present study, 6,8-dihydroxydaidzein (6,8-DHD or 6,7,8,4′-tetrahydroxyisoflavone) and 6,8,3′-trihydroxydaidzein (6,8,3′-THD or 6,7,8,3′,4′-pentahydroxyisoflavone) were synthesized via a facile and efficient way using commercially available formononetin as starting material. Their structures were confirmed using spectroscopic analyses (infrared, nuclear magnetic resonance, and mass spectrometry). The purity was checked by ultra-high performance liquid chromatography. Their antioxidant activities were evaluated via 1,1-diphenyl-2-picrylhydrazyl radical scavenging assay and reducing power assay using ascorbic acid (vitamin C) as a reference compound. The antihypoxia capacity was determined by a hypoxia injury model in PC12 cells. Our study revealed that 6,8-DHD and 6,8,3′-THD exhibited higher antioxidant activities than that of vitamin C and could protect PC12 cells against hypoxia-induced damage. These results indicate that 6,8-DHD and 6,8,3′-THD are excellent antioxidant agents and could be used for alleviating injury induced by hypoxia.
Introduction
Daidzein (7,4′-dihydroxyisoflavone; Figure 1), abundantly present in legumes, especially in soybeans, is a naturally occurring isoflavone with a variety of pharmacological activities, such as antioxidant, 1 antiinflammatory, 2 antidiabetic, 3 neuroprotective, 4 and cardioprotective 5 activities. The antioxidant activities of daidzein are considered to be responsible in part for its beneficial effects. 6 After absorption, daidzein can be readily metabolized by liver microsomes and converted to hydroxylated metabolites, mainly including 7,3′,4′-, 6,7,4′-, and 7,8,4′-trihydroxyisoflavones. 7 Recently, these hydroxydaidzein derivatives have attracted more attention due to their higher antioxidant, 8 antiinflammation, 9 antitumor, 10 and antibacterial 11 activities than that of daidzein.

Structures of daidzein, 6,8-DHD (6,7,8,4′-tetrahydroxyisoflavone) and 6,8,3′-THD (6,7,8,3′,4′-pentahydroxyisoflavone).
Generally, the hydroxyl groups are the main active groups of isoflavone, and increasing the number of hydroxyl groups can enhance the antioxidant capacity. As shown in Figure 1, 6,8-dihydroxydaidzein (6,8-DHD or 6,7,8,4′-tetrahydroxyisoflavone) and 6,8,3′-trihydroxydaidzein (6,8,3′-THD or 6,7,8,3′,4′-pentahydroxyisoflavone) were 2 hydroxylated daidzein derivatives with 4 and 5 hydroxyl groups, respectively, in the structures. 6,8-DHD was first isolated from biotransformation of daidzein by
Results and Discussion
The aim of this study was to synthesize and assess the antioxidant and antihypoxia activities of 6,8-DHD and 6,8,3′-THD. Formononetin (7-hydroxy-4'-methoxyisoflavone,

Synthetic route to 6,8-DHD and 6,8,3′-THD.
As shown in Figure 3, the purities of 6,8-DHD and 6,8,3′-THD were determined by ultra-high performance liquid chromatography (UHPLC) to be >97%.

UHPLC spectra of 6,8-DHD (A) and 6,8,3′-THD (B).
In this study, the antiradical activity of 6,8-DHD and 6,8,3′-THD was evaluated using a DPPH scavenging assay, which is a facile method and has been widely applicable for measuring the capacity of samples to act as free radical scavengers. 15 As described in Figure 4(A), the DPPH radical scavenging capacity of daidzein was weak and negligible (IC50 » 2 mmol/L). 6,8,3′-THD showed the best DPPH radical scavenging capacity with the IC50 = 0.911 ± 0.033 mmol/L, while 6,8-DHD also showed good scavenging capacity with the IC50 = 1.203 ± 0115 mmol/L. The reference compound vitamin C exhibited moderate potency with the IC50 = 1.667 ± 0.009 mmol/L. 6,8,3′-THD and 6,8-DHD showed significantly higher scavenging capacity on DPPH than that of vitamin C.

Antioxidant activities of 6,8-DHD, 6,8,3′-THD, and daidzein: (A) scavenging DPPH radical and (B) reducing power. Data were expressed as the mean ± SD (
The antioxidant activity of 6,8,3′-THD and 6,8-DHD expressed as the capacity of the donating electrons was determined by reducing power assay. As described in Figure 4(B), the reducing power of all compounds increased with increasing concentrations. At a concentration of 1 mmol/L, these compounds exhibited the highest reducing power. The absorbance values at 700 nm of 6,8,3′-THD, 6,8-DHD, daidzein, and vitamin C were 2.501 ± 0.002, 2.296 ± 0.025, 0.314 ± 0.021, and 2.052 ± 0.114, respectively. The order of reduced power was ranked as 6,8,3′-THD > 6,8-DHD > vitamin C > daidzein.
Based on the results of antioxidant assays, 6,8,3′-THD also 6,8-DHD, which had 5 and 4 phenolic hydroxyl groups, were found to exhibit significantly higher antioxidant activity than that of daidzein, which only had 2 phenolic hydroxyl groups. This observation agreed nicely with the theory that the antioxidant capacity of flavonoids was proportional to the number of phenolic hydroxyl groups.
16
In addition the position of the hydroxyl group also affected the antioxidant activity. It has been proved that the
Hypoxia-induced reactive oxygen species overproduction,
18
and administration of antioxidants may exert protection against hypoxia-induced cell death.
19
Therefore, we further evaluate the protective effect of 6,8-DHD and 6,8,3′-THD on hypoxia-treated PC12 cells. As shown in Figure 5(A), treatment with 6,8-DHD, 6,8,3′-THD, or daidzein at a concentration of 1 μmol/L did not exert any cytotoxic or proliferative effects on the PC12 cells under normoxic conditions. However, hypoxia exposure for 24 h significantly reduced the viability of the PC12 cells (Figure 5(B)). Compared to the hypoxia group, pretreated with 6,8-DHD, 6,8,3′-THD or daidzein significantly improved the cell viability (
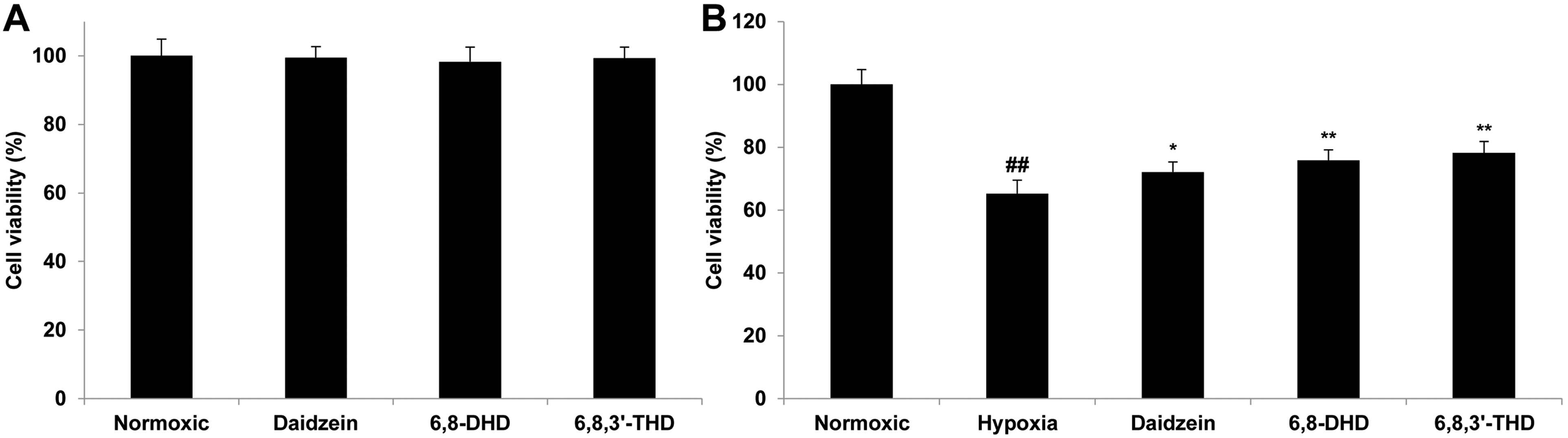
Protective effects on hypoxia-treated PC12 cells. (A) Effects of 6,8-DHD and 6,8,3′-THD and daidzein at the concentration of 1 μmol/L on cell viability of normal PC12 cells. (B) Effects of 6,8-DHD and 6,8,3′-THD and daidzein at the concentration of 1 μmol/L on cell viability of hypoxia-treated PC12 cells. The results are expressed as mean ± SD (
Conclusions
In conclusion, 6,8-DHD and 6,8,3′-THD were synthesized for the first time in a facile and efficient way utilizing formononetin as starting material. 6,8-DHD and 6,8,3′-THD showed stronger antioxidant capacities than that of daidzein and vitamin C. In addition, they also exhibited excellent protective activities against damage induced by hypoxia in PC12 cells. These results indicate that 6,8-DHD and 6,8,3′-THD are outstanding antioxidant agents and can be used for alleviating injury induced by hypoxia.
Experimental
General
Flash column chromatography was performed on silica gel (200-300 mesh). The melting points were measured on an X-4B melting point instrument without correction. Infrared (IR) spectra were recorded using Bruker ALPHA II Fourier transform infrared spectroscopy. The 1H-NMR and 13C-NMR spectra were recorded using a Bruker Avance III HD spectrometer using CDCl3 or dimethyl sulfoxide-
Reagents and Materials
Formononetin (purity >98% by high-performance liquid chromatography) was purchased from Ci Yuan Biotech Co., Ltd. Other commonly used reagents were purchased from commercial sources, and no further purification was carried out.
6,8-Dibromo-7-Hydroxy-4′-Methoxyisoflavone (5 )
To a stirring solution of formononetin (2.68 g, 10 mmol) in DMF (40 mL) at 25 °C was added NBS (3.60 g, 25 mmol). The resulting mixture was stirred for 1.5 h and then poured into 100 mL of cold 2 M hydrochloric acid (HCl). Then 250 mL of distilled water was added. The precipitate was filtered, washed with distilled water, and dried under reduced pressure to afford compound
6,8,3′-Tribromo-7-Hydroxy-4′-Methoxyisoflavone (6 )
Compound
7-Hydroxy-6,8,4′-Trimethoxyisoflavone (7 )
CuBr (1.75 g, 12.5 mmol) was suspended in 4 mL of DMF and stirred in the dark for 0.5 h at 25 °C. A solution of 25% sodium methoxide in methanol (60 mL) was added to the mixture, which was stirred in the dark for another 1 h. The resulting mixture was added to a stirring solution of compound
7-Hydroxy-6,8,3′,4′-Tetramethoxyisoflavone (8 )
Compound
6,7,8,4′-Tetrahydroxyisoflavone (6,8-DHD, 2 )
To a stirred solution of AlCl3 (269 mg, 2.25 mmol) in CH2Cl2 (12 mL) at 5 °C was added dropwise 150 µL of dimethyl sulfide (DMS, CH3SCH3). Then compound
6,7,8,3′,4′-Pentahydroxyisoflavone (6,8,3′-THD, 3 )
Purity Test
Purity was analyzed using a Thermo Ultimate 3000 UHPLC system equipped with an Acclain-C18 column (particle size = 2.2 µm, dimensions = 100 mm, and pore size = 2.1 nm) maintained at 25 °C. The mobile phase was composed of water (0.01% acetic acid)/methanol (30:60, v/v). Then 10 µL of the sample dissolved in methanol was injected. The ultraviolet detection wavelength was 254 nm and the flow rate was 0.2 mL/min.
DPPH Radical Scavenging Assay
The DPPH radical scavenging activity of 6,8-DHD and 6,8,3′-THD was evaluated according to the reported method with slight modifications.
20
Then 100 μL of different concentrations (0.0625-2.0 mmol/L) of samples in DMSO and 100 μL of DPPH solution (100 µM) in methanol were added to a 96-well plate, shaken vigorously, and reacted in the dark for 0.5 h at 25 °C. The absorbance of the resulting solution was recorded on a microplate reader (Spectramax i3 Multi-Mode Detection Platform, Molecular Devices) at a wavelength of 517 nm. Vitamin C was used as the reference compound. The DPPH radical inhibition ratio was assessed using the following equation: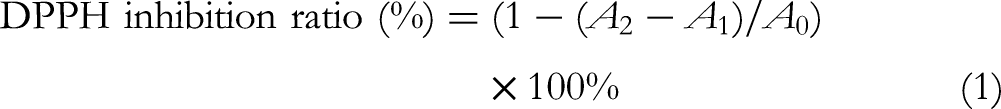
Reducing Power Assay
The reducing power of 6,8-DHD and 6,8,3′-THD was determined according to the previously reported method with slight modification. 21 Then 100 μL of different concentrations (0.03125-1.0 mmol/L) of samples in DMSO were mixed with 2 mol/L sodium phosphate buffer (pH = 6.6, 2.5 mL) and 1% (w/v) potassium ferricyanide (2.5 mL). The resulting mixture was incubated at 50 °C for 0.5 h. After cooling to 25 °C, 10% trichloroacetic acid (2.5 mL) was added to the mixture, which was then centrifuged at 3000 r/min for 10 min. The upper layer (2.5 mL) was mixed with distilled water (2.5 mL) and 0.1% FeCl3 solution (0.5 mL) at 25 °C. After 0.5 h, the absorbance was recorded on an ultraviolet-visible spectrophotometer (NanoPhotometer-NP80, Implen) at a wavelength of 700 nm. Vitamin C was used as the reference compound. The assay was performed in triplicate.
Cell Culture and Treatment
The PC12 cells were obtained from the Cell Bank of the Chinese Academy of Sciences and cultured in Dulbecco’s modified Eagle’s medium (DMEM; Bioss) with 10% (v/v) fetal bovine serum (Bioss), and 1% penicillin-streptomycin (Bioss) in a humidified atmosphere containing 95% N2 and 5% CO2 at 37 °C. To establish a hypoxia damage model, the PC12 cells (passage 4-6) were subject to hypoxic conditions (94% N2, 1% O2, and 5% CO2) at 37 °C for 24 h. To evaluate the protective effects of hydroxydaidzein derivatives, PC12 cells were incubated with 1 μmol/L of samples for 1 h and then cultured under normoxic or hypoxic conditions for 24 h at 37 °C. Samples were dissolved in DMEM containing 1% DMSO.
Cell Viability Assay
The cell viability was assessed by a cell counting kit-8 (CCK-8). PC12 cells were added to 96-well plates (1 × 104 cells/well) and cultured. Following treatment as described above, 90 µL of media and 10% CCK-8 solution (Bioss) was added to each well via media exchange modes. Then, the plates were incubated for an additional 2 h at 37 °C. The optical density was recorded on a microplate reader (Spectramax i3 Multi-Mode Detection Platform, Molecular Devices) at a wavelength of 450 nm. Cell viability was expressed as a percentage of the normoxic control.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221126042 - Supplemental material for Synthesis, Antioxidant, and Antihypoxia Activities of 6,7,8,4′-Tetrahydroxyisoflavone and 6,7,8,3′,4′-Pentahydroxyisoflavone
Supplemental material, sj-docx-1-npx-10.1177_1934578X221126042 for Synthesis, Antioxidant, and Antihypoxia Activities of 6,7,8,4′-Tetrahydroxyisoflavone and 6,7,8,3′,4′-Pentahydroxyisoflavone by Hongqiang Tan, Jin Shao, Jie Zhang, Huiping Ma and Linlin Jing in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China (Grant No. 81872796, 81202458, 81571847) and the Logistics Research Program of PLA (Grant No. CWH17J010).
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
