Abstract
Carvone is one of the most versatile synthons in organic synthesis. It has been used as starting material for a huge number of organic syntheses of natural products with interesting biological activities. This manuscript communicates the antioxidant activity against superoxide of carvone and several derivatives.
The importance of natural products in pharmacy and medicine as a source of bioactive compounds is out of doubt. Our group has tried to use different natural products such as sclareol
1
-3
or

Some building blocks obtained from carvone.
The structure of compounds
The Nuclear Magnetic Ressonance (NMR) data are shown in Tables 1
-3 for the carvone derivatives
1H and 13C NMR, Correlation HSQC and HMBC for Compound 6.

1H and 13C NMR, Correlation HSQC and HMBC for Compound 9.

1H and 13C NMR, Correlation HSQC and HMBC for Compound 10.

2,2-Diphenyl-1-Picrylhydrazyl Radical (DPPH•) Scavenging Activity
To assess the in vitro antioxidant activity, we first examined whether compounds possessed antiradical activity using 2,2-diphenyl-1-picrylhydrazyl radical (DPPH•) scavenging assay.
2,2-Diphenyl-1-picrylhydrazyl radical is not a radical with biological importance, however, this assay is considered a valid accurate, easy, and economic method to evaluate radical scavenging activity of antioxidants, since the radical compound is stable and it has not been generated. 11
In this model, we employed quercetin and curcumin as standards, which have shown a significant antiradical ability against DPPH, with the maximum inhibitory effect of 96.8% ± 0.1% and 98.7% ± 1.8%, respectively (Figure 2).

Effect of terpene compounds evaluated on 2,2-diphenyl-1-picrylhydrazyl radical. Different concentrations of compounds and patterns, quercetin and curcumin, were evaluated (1 × 10–9-1 × 10–4 M).
The evaluated compounds showed variable scavenging activities for this free radical. So, in Figure 2(a), in which we collected a group of evaluated monoterpenes, we observed that these compounds had no significant antiradical activity when we compared them with positive controls. Activities obtained with these terpenes were between 20% and 30%.
The compounds that presented the higher antiradical activity against DPPH were
Nitric Oxide Radical Scavenging Activity
Nitric oxide radical (NO•) plays an important role in the control of various physiological and pathophysiological pathways. 12
Although NO in low concentrations is not toxic, during pathological conditions, it can become harmful due to its high reactivity with other free radicals, such as superoxide anion (O2 •). In anaerobic conditions, the NO is highly unstable and reacts with the oxygen to produce intermediate products (NO2, N2O4, and N3O4). In this condition, the NO could react with the O2 • and generate peroxynitrite (ONOO−), a strong oxidant that reacts with most biological molecules, causing cell damage.
This delicate balance between the physiological and the pathological role must be achieved through the inhibition of excess NO. It can also be achieved by reducing O2 • levels, thus preventing the production of reactive nitrogen species (RNS).
The model of antiradical activity against NO applied in this assay was an indirect method to predicting the ability of a compound to capture the excess of NO, and in this way, we prevent the generation of ONOO-.
Table 4 shows the results obtained when the assayed compounds were evaluated against the NO radical. The percentage of NO scavenger developed by the pattern compounds quercetin and curcumin was 31.3 ± 2.5 and 29.2 ± 2.0, respectively, which could be considered as a discrete activity. For quercetin, the maximum activity was registered by the concentration of 1 × 10–8 M, while for curcumin the maximum inhibitory effect was observed with the 1 × 10–9 M.
Maximal Inhibitory Effect and Half Maximal Inhibitory Concentration (IC50) of Terpene Compounds Evaluated on NO and O2 • Radicals.
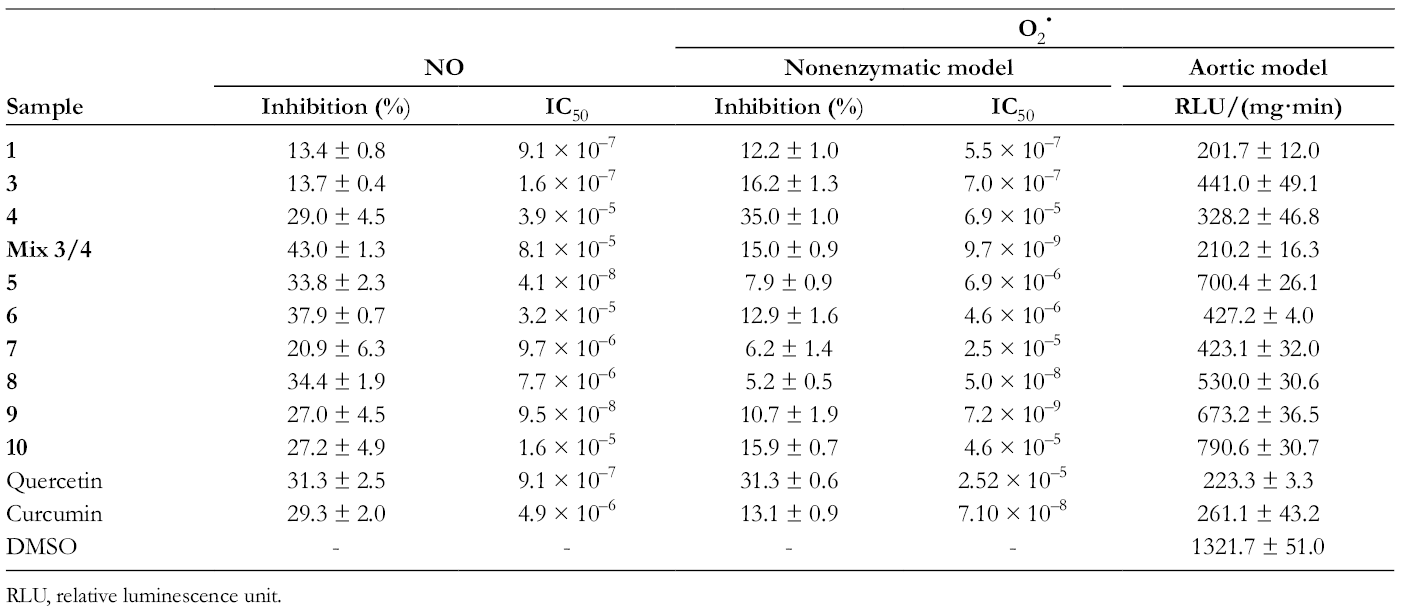
RLU, relative luminescence unit.
Same as the pattern compounds, the activity of all terpenes to captain the NO was lowered to 50%. We observed that the mixture
Assay of Superoxide Radical Scavenging Activity
Superoxide anion is one of the free radicals of higher biological importance since it is in greater concentration on the organism, with the initiator of oxidative stress chain, constituting the main precursor of other reactive species of oxygen and nitrogen.
In the model tested, O2− was produced chemically without the presence of enzymes (nonenzymatic model) which determines the percentage of superoxide anion uptake. Thus, in this system, the evaluated compounds can exert an antioxidant activity by reducing the production of O2−, or by a stabilizing action of the radical when donating or receiving electrons to the O2− radical.
In Table 4, the results obtained by the evaluation of set of compounds are shown. The monoterpenes that were derived from Carvone,
Vascular Superoxide Production Assay
The antioxidant activity evaluation against O2
• in the presence of lucigenin is achieved by the chemoluminiscence measurement of the formation of O2
• in a biological medium as the aorta rings. It was determined by taking into account that the diminution of the chemoluminiscence signal indicates less O2
• concentration, therefore a higher antioxidant activity. This system permits the evaluation of another mechanism of activity antioxidant in an indirect way as the possible modulation of pro-oxidants’ enzymatic systems. It has been demonstrated that the incubation of the rings with the inhibitors of NADPH oxidase, nitric oxide synthase, xanthine oxidase, and the cyclooxygenase stops the anion superoxide production and lucigenin is not able to generate a good signal. Although lucigenin can potentiate that production of the superoxide anion radical without the pro-oxidative enzymes previous action in the production of that radical, lucigenin, it is not able to generate superoxide anion
Our results show that the baseline production of O2
• in aortic rings incubated with lucigenin was significantly inhibited in the presence of the reference substances quercetin and curcumin, whose relative luminescence unit (RLU) values at maximum concentration were 223.3 ± 3.3 and 261.1 ± 43.1, respectively (Figure 3; Table 4). At the same concentration, monoterpenes

Effect of quercetin and terpene compounds on the vascular superoxide anion (O2
•) production in aorta rings (
In Figure 3, we can observe that, although to a lesser extent than compounds
The antioxidant efficacy of the compounds evaluated does not present a pattern, but, like the reference compounds, differs depending on the radical to be inhibited. It should be noted that despite the variability in the antioxidant activities developed by monoterpenes in different trials, it is the chemoluminescence model that offers the most reliable data. It is acknowledged that chemoluminescence can be very sensitive in the detection of low-level reactions because it provides a detectable response below the detection limit of most chemical assays.
If we think the structure activity relationship can be deduced for more substituted monoterpenes, the activity diminished in a biological media with the halogenated ones being most active.
Conclusion
Three new compounds have been synthesized from (
Experimental
General Methods
Unless otherwise stated, all chemicals were purchased with the highest purity commercially available and were used without further purification. Infrared (IR) spectra were performed on a Genesis II ATR spectrophotometer. The samples were placed on the diamond, previously cleaned with isopropanol. The spectra were performed using the OMNIC software. 1H and 13C NMR spectra were performed in CDCl3 and referenced to the residual peak of CHCl3 at δ 7.26 ppm and δ 77.2 ppm, for 1H and 13C, respectively, using a Bruker Avant 400, 400 and 100 MHz spectrophotometer. Chemical shifts (δ) are reported in parts per million (ppm) and coupling constants (
Synthesis of Compound 6
In a round-bottom flask equipped with a reflux condenser, (
Compound
HRMS (EI) calc for C17H18BrNO5Na requires (M+Na) 283.0341; found 283.0307. NMR data, see Table 1.
Synthesis of Compound 9
In a round-bottom flask equipped with a reflux condenser, (
Compound
Synthesis of Compound 10
In a round-bottom flask equipped with a reflux condenser, (
Compound
IR (film): 3499, 2941, 1645, 1597, 1385, 1373, 1217, 1141, 1010, 904, 745, 630 cm−1. NMR data, see Table 3.
2,2-Diphenyl-1-Picrylhydrazyl Radical (DPPH•) Scavenging Activity
The DPPH free radical scavenging activity was estimated by assay based on the method described in the literature. 13 All compounds were dissolved in dimethylsulfoxide (DMSO) at different concentration solutions (1 × 10–9-1 × 10–4 M). Quercetin and curcumin were used as positive controls. The reaction mixtures contained 100 µL of sample solution and 100 µL DPPH methanol solution (0.3 mM), and then, the absorbance was measured at 515 nm after incubation at 37°C for 30 minutes. The DPPH radical scavenging effect was calculated using the following formula:

where
Nitric Oxide Radical Scavenging Activity
Nitric oxide scavenging activity was estimated by assay based on Griess Illosvoy reaction. 14 About 50 µL of the test sample (1 × 10–91 × 10–4 M in DMSO) was mixed with 50 µL of sodium nitroprusside (10 mM), and 50 µL of Griess reagent prepared in saline phosphate buffer (pH 7.4). The microplates were incubated for 150 minutes at room temperature and the absorbance was recorded at 546 nm. Curcumin was used as a standard. The percentage of NO• radical scavenging was calculated according to Eq. (1).
Superoxide Radical Scavenging Activity
Measurement of O2 • scavenging activity of terpene compounds was based on the method described by Lin and Chou. 15 Superoxide radicals were generated in a (Phenazine Methosulfate - Reduced Nicotinamide Adenine Dinucleotide) PMS-NADH system by the oxidation of NADH and assayed through the reduction of NBT. The superoxide radicals were generated with 50 µL of different concentrations of the sample (1 × 10–9-1 × 10–4 M), 50 µL of NBT (150 µM) solution, and 50 µL of NADH (936 µM) solution. The reaction started by adding 50 µL of PMS solution (120 µM) to the mixture, it was incubated at 25°C for 5 minutes, and the absorbance at 560 nm was then measured by an automated microplate reader (GloMax-Multi + Detection System with Instinct Software). Decreased absorbance of the reaction mixture indicated increased superoxide anion scavenging activity. The percentage of superoxide radical scavenging was calculated according to Eq. (1).
Vascular Superoxide Production Assay
Vascular O2 • production was measured in thoracic aorta segments using lucigenin-enhanced chemiluminescence, as previously described. 16 Vessels were incubated in a (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) HEPES buffer (in mM: NaCl, 119; HEPES, 20; MgSO4, 1; KCl, 4.6; KH2PO4, 0.4; Na2HPO4, 0.15; NaHCO3, 5; CaCl2, 1.2; glucose, 5.5; pH 7.4) aerated (95% O2/5% CO2) and maintained at 37°C for 30 minutes. After an equilibrated period, samples were then transferred into fluorescence microplates containing 200 µL of the HEPES buffer, 5 µM lucigenin, and supplemented with the corresponding tested samples, positive controls (10−7 -10−5 M) and the solvent DMSO. Under these conditions, superoxide levels were measured by chemiluminiscence using a luminometer (Glomax Multi Detection System). Luminescence units were recorded every 30 seconds for 10 minutes. The relative values of •O2 – production were expressed as RLU per milligram and minute of dry tissue (RLU/(mg·min)).
Statistical Analysis
Results of antioxidant activity are presented as mean ± standard deviation (mean ± SD). Minimum of 3 independent experiments were carried out. Statistical analysis was performed using (one-way analysis of variance) one-way ANOVA and significanct difference between the treatments was accepted at the level of
Footnotes
Acknowledgment
AMR and IET thank MINECO (FPU) and Junta de Castilla y León, respectively, for their grants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Secretaría Nacional de Ciencia, Tecnología e Innovación (INF11-054 and FID11-090), MINECO CTQ2015-68175-R, FEDER, and University of Salamanca.
