Abstract
Liver fibrosis is a serious health problem and may lead to advanced liver cirrhosis and hepatocellular carcinoma if left untreated. In this study, a mouse liver fibrosis model was established by the administration of 3,5-diethoxycarbonyl-1,4-dihydrocollidine (DDC), and tanshinone IIA.
Introduction
Liver diseases, such as liver fibrosis, have become a threat to human life worldwide. Acute or chronic liver injury can induce liver fibrosis, which exhibits an increased expression of type I collagen and smooth muscle alpha-actin (α-SMA).
1
In a previous study, it was shown that collagen 1A1 (
The
In the present study, 3,5-diethoxycarbonyl-1,4-dihydrocollidine (DDC) was used to induce liver fibrosis in mice, followed by the administration of tanshinone IIA as a therapeutic agent. The expression of
Materials and Methods
Cell Culture and Tanshinone IIA Treatment
Human liver cell lines (LX-2) were maintained in Dulbecco's modified Eagle's medium (DMEM, Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (Gibco) and cultured at 37 °C under 5% CO2. After LX-2 cells reached 80% to 90% confluency, cells were treated with 40 μM tanshinone IIA (tanshinone IIA, purity 0.98, Yuanye Bio-Technology, Shanghai, China) dissolved in DMSO for 48 h. The Con group was treated with an equal volume of DMSO with DMEM.
Animals and Animal Care
BALB/c mice (N = 30, male, 6-8 weeks) were randomly divided into 3 groups: Con, DDC, and DDC-Tan. DDC was used to establish the liver fibrosis model. The 0.1% DDC diet was used to induce fibrosis in mice in the DDC and DDC-Tan groups for 1 month. Mice in the Con group received normal feeding. Mice in the DDC-Tan group were intragastrically administered with tanshinone IIA (1.6 mg/g/day) dissolved in DMSO via gavage for 2 months when the 0.1% DDC diet was used. The Con and DDC groups were intragastrically administered equal quantities of normal saline solution. The degree of liver fibrosis was detected by HE-staining. Mice were group-housed in the Laboratory Animal Center of Jilin University. Mice were provided
Knockdown and Overexpression of H19 , and let-7a
LX2 cells were transfected with knockdown (siRNA) and overexpression (pcDNA3.1) vectors, let-7a mimics, and a let-7a inhibitor for 48 h. Nonspecific siRNA (Nc) and pcDNA3.1-Con were transfected into the control cells.
Gene Expression Analysis
RNA was extracted from LX2 cells and mouse liver tissues, and cDNA was generated using a cDNA First-Strand Synthesis Kit (Tiangen, Beijing, China). Gene expression analysis was conducted using quantitative real-time PCR (qPCR) as follows: initial denaturation at 94 °C for 3 min; denaturation at 94 °C for 10 s, 35 cycles; annealing at 59 °C for 15 s; and extension at 72 °C for 30 s. The internal controls were
Western Blot Analysis
Total protein was extracted from LX2 cells and mouse tissues using a protein extraction buffer (Beyotime, China). Then, proteins were quantified using the bicinchoninic acid (BCA) protein assay kit (Tiangen, Beijing, China). Next, sodium dodecyl sulfate-polyacrylamide gel electrophoresis was employed for the separation of the proteins. After electrophoresis, proteins were transferred to a polyvinylidene difluoride (PVDF) membrane, which was then incubated with primary antibodies, including anti-COL1A1 (Bethyl Laboratories, Montgomery, TX, USA) and anti-GAPDH (AP0066; Bioworld, USA), overnight at 4 °C. Subsequently, membranes were incubated with horseradish peroxidase (HRP)-conjugated affiniPure goat IgG antibodies (Boster Biological Technology, Wuhan, China) for 1.5 h at room temperature. Target bands were analyzed using ECL Super Signal (Pierce, USA).
ELISA
Alanine aminotransferase (ALT), bilirubin, aspartate aminotransferase (AST), and albumin levels were detected using an ELISA kit (Yibo Biotechnology, China). Blood was collected, and mouse serum was isolated from mice in the Con, DDC, and DDC-Tan groups. Levels of ALT, bilirubin, AST, and albumin were analyzed according to the manufacturer's instructions. The optical density of each well was determined at a wavelength of 450 nm. The standard curve was constructed, and the concentration of each sample was calculated.
HE-Staining
Liver tissues of mice in the Con, DDC, and DDC-Tan groups were fixed. The tissues were cut into 4-μm slices, dewaxed with xylene, and washed with either ethanol or water. Staining with hematoxylin was performed for 5 min, after which stained tissue sections were washed with distilled water. The differentiation time was 30 s. Sections were then soaked in either distilled water for 15 min or warm water (50 °C) for 5 min and incubated in Yihong solution for 2 min. The staining was observed under the microscope, and ImageJ software was used for image analysis.
Immunohistochemistry
Liver tissues of mice in the Con, DDC, and DDC-Tan groups were fixed with 4% paraformaldehyde. The tissues were cut into 4-μm slices, dewaxed, and washed with distilled water. Then, sections were incubated with primary antibodies (anti-collagen I, anti-α-SMA, and Sirius red) overnight at 4 °C. Tissue sections were then incubated with a biotinylated secondary antibody at 37 °C for 20 min. Following a 3,3´-diaminobenzidine staining step, nuclei were stained with hematoxylin. Then, sections were sealed with neutral resin. ImageJ software was used for image analysis.
Statistical Analysis
Data obtained by qPCR and ELISA were analyzed using GraphPad Prism 5.0 (GraphPad Software, Inc., San Diego, CA, USA). The non-pairing student T-test and two-way ANOVA were used to evaluate the differences in this study. Data are expressed as the mean ± standard deviation.
Results and Discussion
Tanshinone IIA Inhibits Liver Fibrosis in Mice
In the present study, DDC was used to induce liver fibrosis in mice. These mice were treated with tanshinone IIA for 2 months (Figure 1A and B). Subsequently, levels of ALT, AST, bilirubin, and albumin in mouse serum were evaluated using enzyme-linked immunosorbent assay (ELISA). The results showed that levels of ALT, AST, and bilirubin were increased in the DDC group (Figure 1C to E), while levels of albumin decreased (Figure 1F). This indicates that tanshinone IIA may play a role in regulating ALT, AST, bilirubin, and albumin levels in liver fibrosis.

Analysis of mouse liver function after tanshinone IIA treatment. (A-B) The chemical structure of Tanshinone I and Tanshinone IIA. The red area indicates the difference between tanshinone IIA and tanshinone I. (C) ALT content determined by ELISA. (D) AST content determined by ELISA. (E) Bilirubin content determined by ELISA. (F) Albumin determined by ELISA. Con, normal mouse; DDC, DDC module; DDC-Tan, tanshinone IIA treatment group. **** (
Tanshinone IIA antagonizes the expression of TGF-β1, which induces liver fibrosis and inhibits the activation of hepatic stellate cells.
12
Previous reports have shown that lncRNA
Tanshinone IIA Regulates the Expression of COL1A1
To confirm the regulatory role of tanshinone IIA in liver fibrosis, hematoxylin-eosin (HE) staining was employed. The results showed that tanshinone IIA slowed the progression of liver fibrosis (Figure 2A). Further, Sirius red and α-SMA staining results showed a decrease in the number of positive cells in the tanshinone IIA administration (DDC-Tan) group (Figure 2B and C) compared with the control (Con) group. To investigate further the effect of tanshinone IIA on liver fibrosis, immunohistochemistry (IHC) was employed to analyze the expression of

Analysis of liver tissue after tanshinone IIA treatment in mice. (A) HE-staining. (B) Immunohistochemical analysis of Sirius red staining. (C) Immunohistochemical analysis of α-SMA staining. (D)
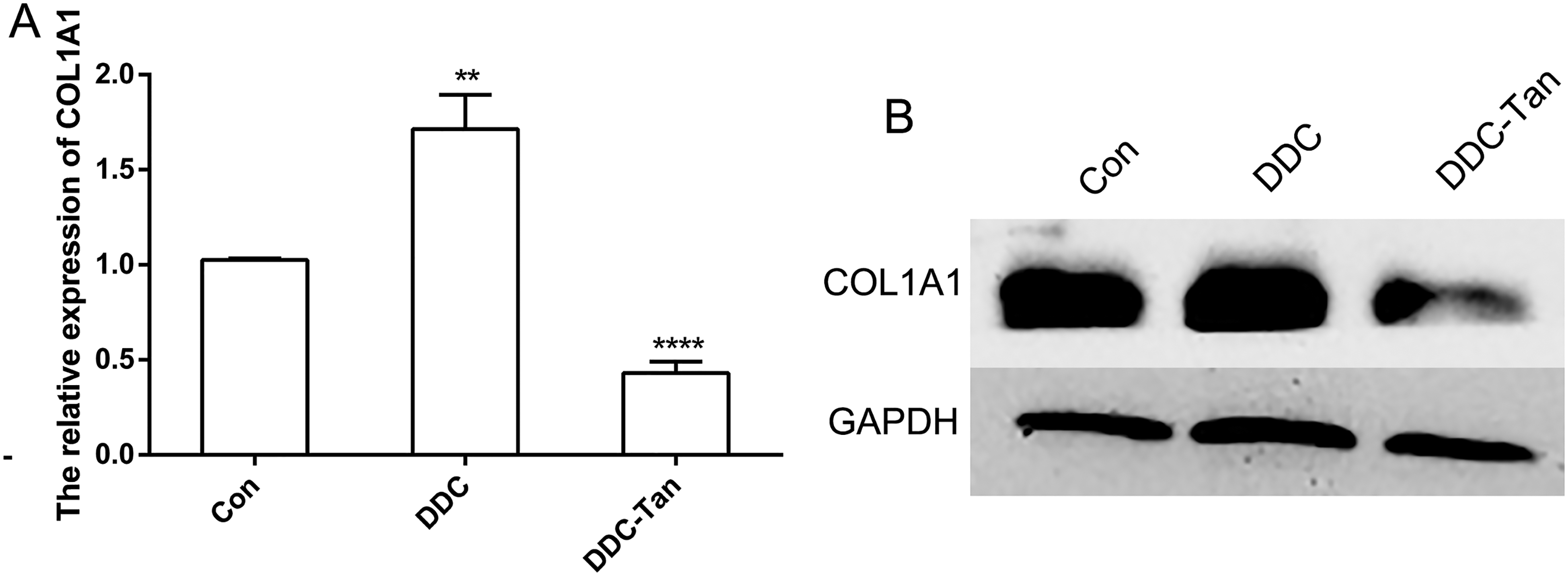
Analysis of
Previous reports have shown that
H19/let-7a Regulates COL1A1 in Liver Fibrosis
Bioinformatic analysis revealed that
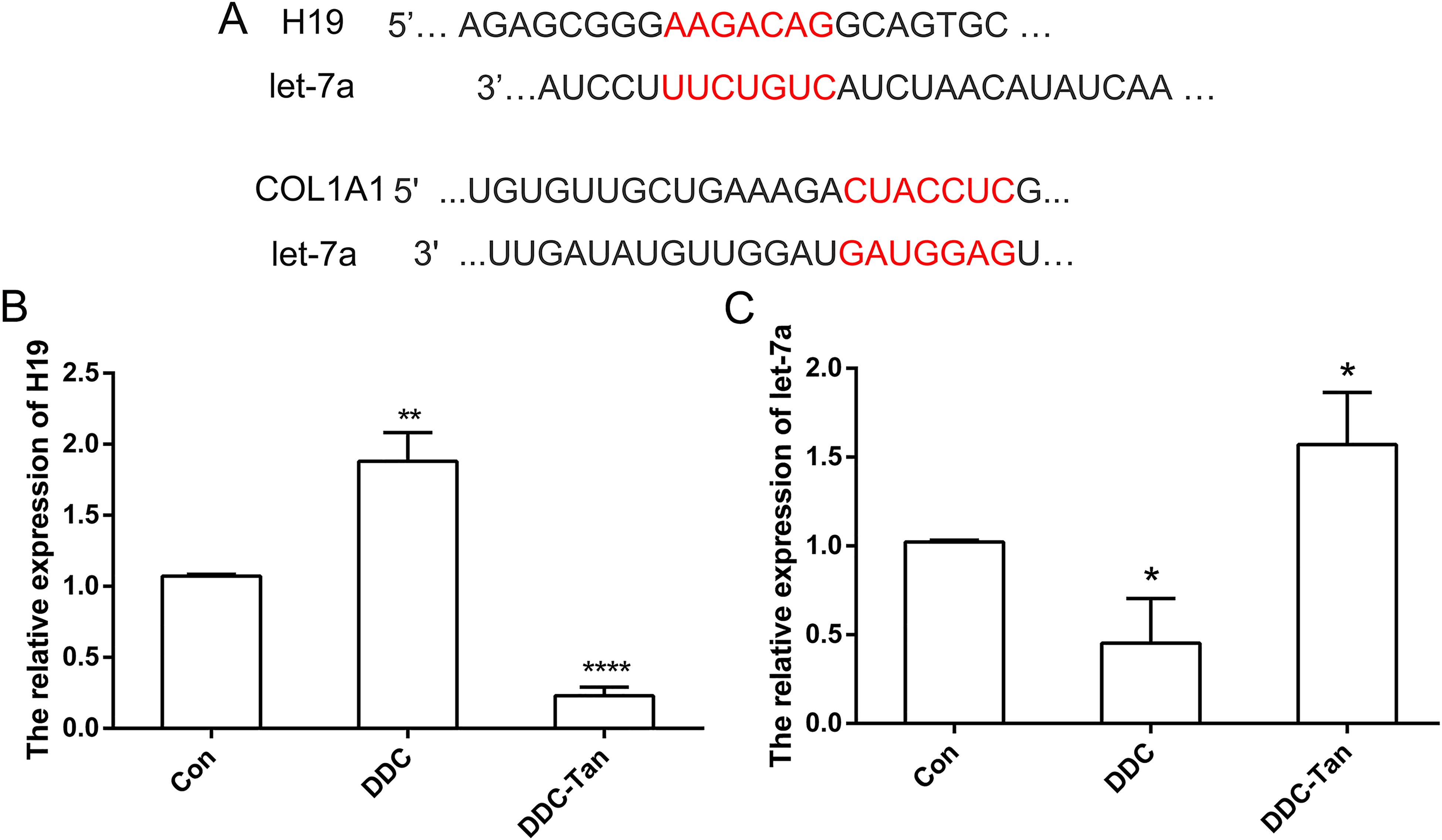
The expression pattern of

Conclusions
The expression of
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221123698 - Supplemental material for Tanshinone IIA Inhibits Liver Fibrosis by Regulating COL1A1 Expression Through
H19/ let-7a in Mice
Supplemental material, sj-docx-1-npx-10.1177_1934578X221123698 for Tanshinone IIA Inhibits Liver Fibrosis by Regulating COL1A1 Expression Through
H19
Footnotes
Acknowledgements
The authors express their gratitude to Ming Hao at the Jilin University for technical assistance.
Authors’ Contributions
Chao Lin designed the experiments and wrote the manuscript. Ning Yin and Jianming Xing performed cell experiments and gene expression analysis. Liqun Sun and Ziping Jiang contributed reagents and materials. Yongjian Gao and Shuo Yang carried out animal experiments. Dongxu Wang analyzed data and prepared figures. All authors reviewed and approved the manuscript.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animals were cared for in accordance with the Guide for the Care and Use of Laboratory Animals in China. All experimental procedures were approved by the Animal Care and Use Committee of Jilin University (grant number. SY201705007).
Funding
This work was supported by the Fundamental Research Funds for the Central Universities under grant number 2019JCKT70, the Jilin Province University Key Laboratory Scientific Research Project under Grant [2019] No. 004, and the Jilin Scientific and Technological Development Program under grant numbers 202002006JC and 20210101010JC.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
