Abstract
Gastric ulcer (GU) is a clinically common gastrointestinal disease with a long disease course that frequently reoccurs. Banxia Xiexin decoction (BXD), a traditional Chinese medicine prescription, has a prominent protective effect against GU. Nonetheless, the therapeutic mechanisms of BXD against GU remain elusive. In this study, a rat model of GU was established by gavage with 95% ethanol, and BXD significantly attenuated the inflammatory effect of GU in rats. An “active ingredient–target” interaction and GU protein–protein interaction networks were constructed based on system biology, which could screen out the crucial active ingredients. The target protein–protein interaction network for the BXD treatment of GU was constructed to identify the key target proteins with network topology parameters. The DAVID database was then used to perform Gene Ontology and Kyoto encyclopedia of genes and genomes enrichment analysis on the proteins targeted by BXD in the treatment of GU. Finally, molecular docking technology was used to study the interactions between key active ingredients and core target proteins. A total of 89 active ingredients of BXD were screened and 63 target proteins of BXD in the treatment of GU were identified. Through the analysis of protein–protein interaction and the active ingredient–target protein network diagram, it was found that tumor necrosis factor-α(TNF-α), AKT1, and PTGS2 may play a key role in the treatment of GU by BXD. Molecular docking showed that these 3 core target proteins had a good affinity with the main components of BXD, including baicalein, norwogonin, and skullcapflavone II. The mechanism of BXD against GU may involve the inhibition of inflammatory response and oxidative stress, involving signaling pathways such as TNF, hypoxia-inducible factor-1, and mitogen-activated protein kinase. Network pharmacology and molecular docking technology indicated the key active ingredients, target proteins, and signal pathways that may be the biological basis of BXD in the treatment of GU.
Introduction
Gastric ulcer (GU) is a clinically frequent gastrointestinal disease that is mainly due to various pathogenic factors, including abnormal gastric acid secretion, alcoholism, Helicobacter pylori (Hp) infection, excessive use of nonsteroidal antiinflammatory drugs, and irregular diet. When the gastric mucosa fails to resist invading factors, tissue necrosis and shedding occur, eventually leading to the formation of ulcers. 1 The clinical manifestations are periodic pain in the upper abdomen, which can cause gastric perforation and cancer, seriously threatening human health and safety. According to statistics, the incidence of GU in the global population is approximately 5% to 10%. 2 The current treatments for GU include the suppression of gastric acid secretion, the protection of gastric mucosa, and the eradication of Hp. However, proton-pump inhibitors have poor durability, lead to frequent relapse, and have a large number of adverse reactions. 3 Long-term application can easily lead to drug resistance and dependence; hence, they are subject to certain restrictions in clinical application. 4
Banxia Xiexin decoction (BXD), which has been used for the clinical treatment of gastrointestinal disease for thousands of years, possesses the characteristics of an effective multiingredient, multiprotein target therapeutic. 5 The traditional Chinese medicine (TCM) kangfuxin exerts anti-GU effects by inhibiting oxidative stress and endoplasmic reticulum stress. 6 Moreover, it can also resist oxidation and inhibit inflammation to reduce the damage caused by GU. 7 BXD prescription consists of several medicinal herbs, including Pinellia Rhizoma, Glycyrrhizae Radix et Rhizoma, Scutellaria Radix, Coptidis Rhizoma, Ginseng Radix et Rhizoma, Zingiberis Rhizoma, and Jujube Fructus. A previous study demonstrated that BXD has a good clinical effect on gastrointestinal diseases. 8 TCM is widely used in the treatment of GU and has shown good clinical effects. 9 However, the targets and signaling pathways of BXD remain unclear, and its mechanism of action in the treatment of GU remains unknown.
Network pharmacology is a systematic method of screening and topological analysis used to highlight the discovery of disease and drug molecular targets and signaling pathways at multiple levels and from different perspectives. A complex biological network of interactions between “disease–gene–target–active ingredients” coincides with the “multicomponent, multitarget and integrity” approach of TCM compounds. Recently, network pharmacology has been used to investigate the mechanisms of action of traditional Chinese medicines.10,11 Therefore, in this study, network pharmacology was used to screen the active ingredients of BXD, virtual targeting technology was used to reverse screen the targets of active ingredients, and the information was examined against multiple databases to systematically investigate the potential targets of BXD in the treatment of GU. Bioinformatic analysis of the biological functions of the target proteins of BXD in the treatment of GU and enrichment analysis was performed to investigate the signaling pathways and mechanism, and the affinity of crucial compounds and key targets were verified by molecular docking technology, thereby providing an important reference basis for the clinical application of BXD.
Materials and Methods
Materials
Pinellia Rhizoma, Scutellaria Radix, Ginseng Radix et Rhizoma, Zingiberis Rhizoma, Coptidis Rhizoma, Glycyrrhizae Radix et Rhizoma, and Jujube Fructus were selected based on the standards specified in the Chinese Pharmacopoeia and were purchased from the Shanghai Kang Qiao Chinese Medicine Tablet Co., Ltd. Enzyme-linked immunosorbent assay (ELISA) kits for interleukin (IL)-2, IL-8, and tumor necrosis factor-α (TNF-α) were obtained from Sinobest Biotech. Omeprazole (OME) was purchased from Luoxin Pharmaceuticals Group Co., Ltd.
Preparation of BXD
In accordance with the prescription, the 7 herbs of BXD, including Pinellia Rhizoma (12 g), Coptidis Rhizoma (3 g), Ginseng Radix et Rhizoma (9 g), Zingiberis Rhizoma (9 g), Glycyrrhizae Radix et Rhizoma (9 g), Scutellaria Radix (9 g), and Jujube Fructus (6 g), were soaked in water for 30 min and then boiled gently twice for 1 h. Finally, the 2 filtrates were combined to a concentration of 1 g·mL−1.
Animal Experiments
Sprague-Dawley rats (160-180 g body weight) were purchased from Beijing Charles River Laboratory Co., Ltd, certificate number: SCXK 2016-0006. All animal experimental procedures were in accordance with the Institutional Animal Committee of Shanghai University of TCM (Permit No. PZSHUTCM190308019). All rats except those in the control group were administered 95% ethanol (2 mL·kg−1) by gavage to induce GU; the control group was administered the same amount of normal saline. After 1 h, the anatomical pathology comprised ulcers and bleeding in the stomach, indicating that the GU model was successfully established. 12 Rats were then randomly divided into 4 groups (n = 8): the control group (rats in the normal control group), the model group (95% ethanol-treated GU rats), the BXD group (rats were administered BXD at a dose of 10 g·kg−1) and OME group (rats were administered OME at a dose of 20 mg·kg−1). The rats in the control group and model group were administered the same dose of saline every day for 7 days.
Histopathological Analysis
After the rats were anesthetized, the whole stomach was separated, which was cut along the greater curvature of the stomach, the stomach contents were rinsed with saline and then fixed in 4% paraformaldehyde for 24 h, and the sections were embedded in paraffin. The sections were deparaffinized and stained with hematoxylin and eosin (HE), and an optical microscope was used to observe the pathological changes in the gastric tissue samples from each group.
ELISA Assay
The serum was separated from 4 mL of blood by centrifugation at 4°C for 15 min at 3000 g. The serum levels of IL-2, IL-8, and TNF-α were measured according to the manufacturer's instructions.
Establishment of Chemical Composition of BXD
BXD is composed of 7 herbs: Pinellia Rhizoma, Glycyrrhizae Radix et Rhizoma, Scutellaria Radix, Coptidis Rhizoma, Ginseng Radix et Rhizoma, Zingiberis Rhizoma, and Jujube Fructus. Through searching the TCMSP (http://lsp.nwu.edu.cn/tcmspsearch.php) and TCMID database (http://www.megabionet.org/tcmid/search/), the information on the ingredients of each herb was screened based on the selection criteria of oral bioavailability (OB) ≥30% and drug-likeness (DL) ≥0.18, 13 and the BXD composition database was established, which included the molecular formula of each chemical composition, the simplified molecular input line entry system (SMILES) chemical structure, molecular weight, OB value, and DL value.
The Target Protein of the Virtual Target Active Ingredient
From the active ingredient information for BXD, the SMILES structure of each active ingredient was imported into the Swiss Target Prediction database (http://www.swisstargetprediction.ch/) to predict the target protein of each compound, and a probability of >0.1 was the screening criterion to establish the target protein information of the active ingredients.
Construction of GU Target Protein Database
To obtain GU-related target proteins, through database integration, GU-related targets were searched in the GeneCards Database (https://www.genecards.org/) and Comparative Toxicogenomics Database (CTD) (http://ctdbase.org/). The screening criteria were GeneCards ≥8 points and CTD ≥40 points.
Screening of BXD Against GU Target Protein
The target protein of the active ingredients of BXD and the related target of GU was crossed, and the Venn diagram was established. The target protein of these intersections may be the target protein of BXD in the treatment of GU.
Constructing a “Components–Targets” Network
To investigate the interaction between the active components of BXD and the target proteins, the active components were mapped to the target proteins in the intersection of the Venn diagram and the target proteins of BXD in the treatment of GU. The proteins were then imported into Cytoscape 3.7.1 software to construct the “compounds–targets” network; the “Network Analysis” function was used to analyze the network topology attributes, and the crucial active ingredients were identified based on the degree value.
Construction of the Protein Interaction Network of BXD Against GU
According to the target proteins intersected by the Venn diagram, these proteins were imported to the STRING database (https://string-db.org/) to construct a protein–protein interaction (PPI) network. The “Network Analysis” function was used to analyze the network topology. Average values higher than the degree value and betweenness value were considered to be the key target proteins of GU.
Gene Ontology and Kyoto encyclopedia of genes and genomes Enrichment Analysis
Gene Ontology (GO; http://www.geneontology.org/) is used to study the biological functions of the target proteins. Biological processes (BPs), cellular components, and molecular functions (MFs) are part of the GO analysis. The target proteins were imported into the DAVID (https://david.ncifcrf.gov/) database to obtain GO annotation and enrichment information for the target proteins, which would reveal the biological function of the protein and the degree of enrichment.
To investigate the signaling pathways involved in the treatment of GU by BXD, the target proteins of BXD in the treatment of GU were imported into the DAVID (https://david.ncifcrf.gov/) database to obtain the Kyoto encyclopedia of genes and genomes (KEGG) annotation and enrichment information for the target protein.
Molecular Docking Analysis
To study the interaction between the key active ingredients of BXD and the core target protein, the key target proteins, and important active ingredients were obtained through the construction of the above-mentioned “components–targets” network and PPI network diagram. The SMILES format of the active ingredients was imported into Discovery Studio (2016) (DS), the 3D structure of the target protein was imported into the DS software, and the water molecules were removed. The ligand radius range of the corresponding site was then obtained by extending the 5 amino acid sequences near the site, and then the original ligand was removed and docked with the original ligand and compound, respectively, to obtain the target proteins.
Statistical Analysis
All experiments were conducted in triplicate and the data were expressed as the mean ± SD. All statistical analyses were performed with SPSS 20.0. Group comparisons were performed using analysis of variance followed by Tukey's postmultiple test or Student's test. A P-value of <.05 indicated statistical significance.
Results
Data Analysis Flowchart
Figure 1 shows the data analysis flowchart for the whole study.

Experimental flow chart.
The Effect of BXD on the Gastric Tissue of Rats with GU
The analysis of the gross anatomy of the gastric mucosa revealed that there was no ulcer on the gastric mucosal surface of the rats and no obvious congestion or edema in the control group. After gavage with 95% ethanol, the gastric mucosa of the rats in the model group displayed congestion, with scattered spots, patches, and strips of bleeding and erosion. After the administration of BXD, the ulcer symptoms were significantly attenuated (Figure 2A). HE staining analysis revealed that there was lymphocytic infiltration in the lamina propria–submucosa–serosal layer of the gastric tissue in the model group and a small number of lymphocytes scattered in the muscle layer. However, the symptoms of gastric mucosal damage were alleviated in the BXD treatment group as compared to the model group (Figure 2B).

Effect of BXD on gastric tissue of rats with gastric ulcer: (A) gross observation of rat gastric mucosa; (B) HE staining of gastric tissue (200×); a, control group; b, model group; c, BXD group; d, OME group.
BXD Reduced the Expression Levels of Inflammatory Cytokines in GU Rats
The production of the inflammatory factors was detected in serum levels. Our results showed that the production followed an increasing trend from IL-2 to IL-8 and TNF-α in the model group as compared with the control group. However, BXD decreased the expression level of IL-2, IL-8, and TNF-α as compared with the model group (Figure 3).

Effect of BXD on the inflammatory cytokines level in GU rats: (A) IL-2; (B) IL-8; (C) TNF-α. ##P < .01 compared with the control group; #P < .05 compared with the normal group; *P < .05 compared with the model group.
Active Ingredient Analysis
Through TCMSP and TCMID databases, using the screening criteria of OB ≥30% and DL ≥0.18, 211 chemical components were identified through screening, including 12 compounds from Pinellia Rhizoma, 5 compounds from Zingiberis Rhizoma, 14 compounds from Coptidis Rhizoma, 37 compounds from Scutellaria Radix, 92 ingredients from Glycyrrhizae Radix et Rhizoma, 22 ingredients from Ginseng Radix et Rhizoma and 29 ingredients from Jujube Fructus. After the exclusion of duplicates, 189 constituents were obtained.
Analysis of BXD in Treating GU Target Proteins
According to the targets of the chemical components of BXD, 869 target proteins were screened using virtual technology. Based on the screening conditions of GeneCards ≥8 points and CTD ≥40 points, targets were selected from the GeneCards and CTD databases. A total of 63 target proteins were identified from the intersection of the active ingredient virtual protein and the GU target proteins in the Venn diagram (Figure 4), which were predicted as the target proteins of BXD in the treatment of GU.

Venn diagram of component targets and gastric ulcer targets of Banxia Xiexin decoction (BXD).
“Active Ingredients–Targets” Interaction Network Analysis
Based on the active ingredients mapped to the target proteins at the intersection of the Venn diagram, 63 targets corresponded to 89 compounds in BXD; therefore, 89 compounds were screened (Table 1), and the “active ingredients–targets” network was constructed with 63 target proteins of BXD for the treatment of GU (Figure 5). Our results showed that there were 152 nodes and 696 edge lines in the network. The compound node relationship analysis found that the average degree value was 9.42, the average value of near centrality (betweenness centrality [BC]) was 0.01315 and the average value of closeness centrality (CC) was 0.3820. Moreover, there were 32 compounds with a degree value greater than the average degree value. Our results showed that the top 3 degree values were baicalein (19), norwogonin (17), and skullcapflavone II (17), which were higher than the average degree value of 9.42. These ingredients may be the main active ingredients of BXD against GU. This network showed the comprehensive effects of BXD against GU through multiple components and targets.

The “active ingredients–targets” network of Banxia Xiexin decoction (BXD). In the network, there are 152 nodes and 696 edges, 89 active compounds have related to 63 proteins. Diamonds represent compounds and circles represent targets.
The 89 Chemical Components Information in BXD.
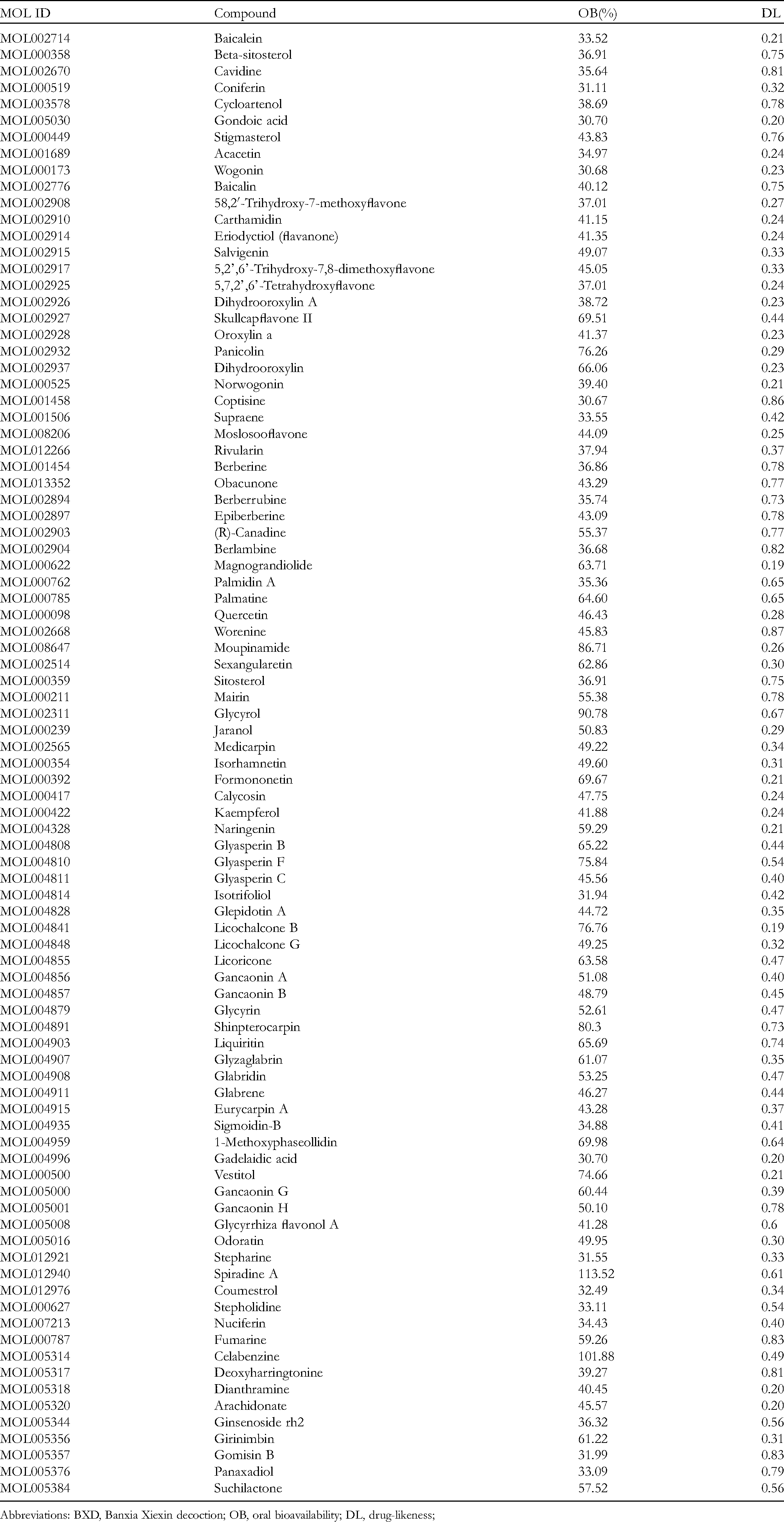
Abbreviations: BXD, Banxia Xiexin decoction; OB, oral bioavailability; DL, drug-likeness;
PPI Analysis
From the analysis of the PPI network (Figure 6), there were 63 nodal proteins and 789 edge lines, with an average degree value of 25.01, an average value of BC of 0.01016, and an average value of CC of 0.6314. There were 29 target points with a higher protein degree. On average, the top 3 target proteins were TNF-α, AKT1, and PTGS2. It could be inferred that these proteins are the core targets of BXD in the treatment of GU.

PPI diagram of BXD in the treatment of GU target proteins. In the network, there are 63 nodes and 789 edges.
GO Enrichment Analysis
The GU target proteins of BXD were imported into the DAVID database. With P < .05 as the screening condition, 412 GO enrichment pathways were obtained, including 305 biological processes (BPs), 34 cell components (CCs), and 73 molecular functions (MFs). Through the enrichment of the first 20 biological functions (Figure 7), BPs, including the cytoplasmic process, oxidative stress process, apoptosis process, recognition protein binding, and enzyme binding participate were identified as the biological process affected by BXD for the treatment of GU.
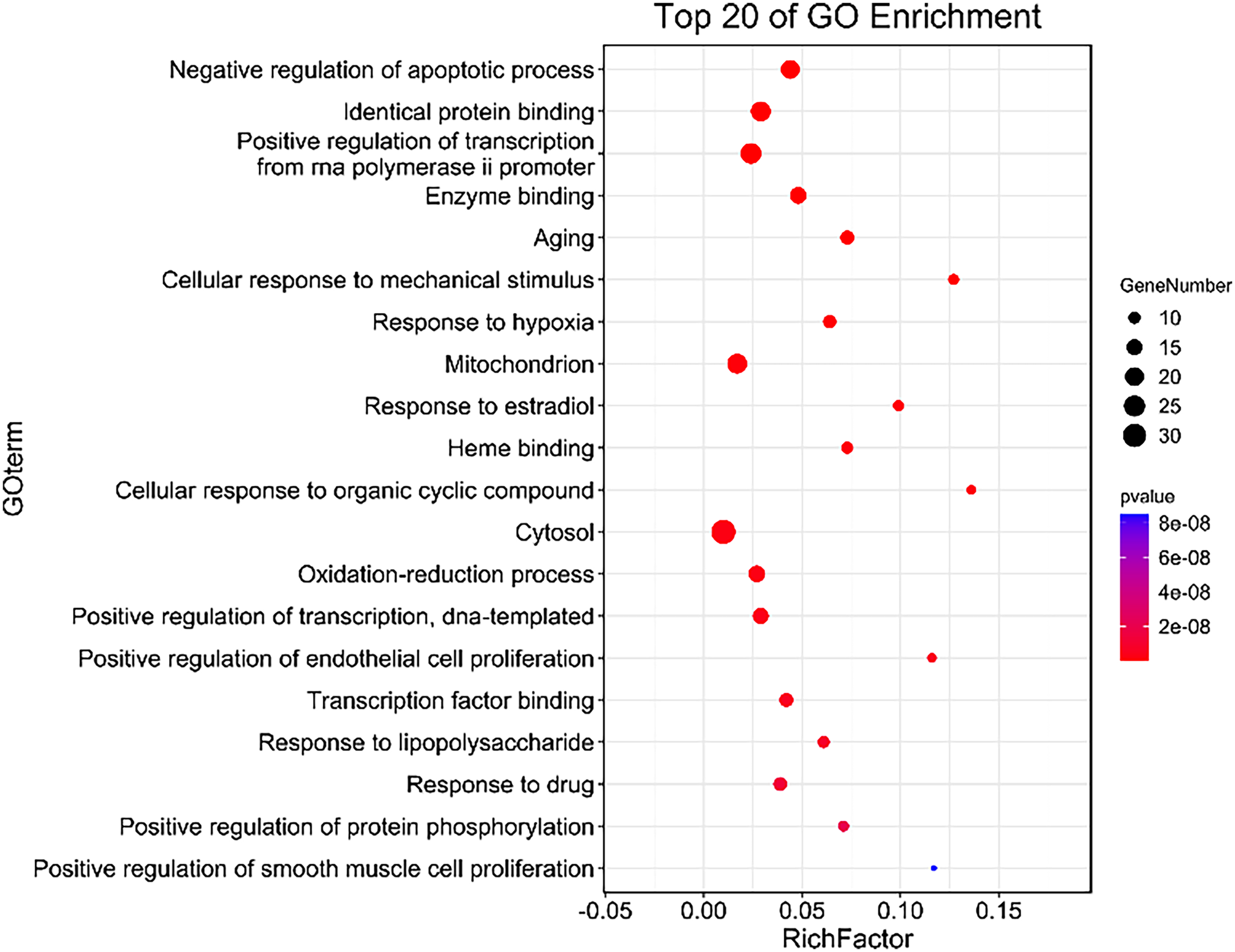
Analysis of GO enrichment of BXD treating GU target proteins. The size of the circle represents the amount of protein enrichment, color represents the significance of enrichment, and the right-hand color gradient represents P-value.
KEGG Enrichment Analysis
Based on the information on the target proteins of BXD in the treatment of GU, information was imported into the DAVID database. With P<.05 as the screening condition, 102 KEGG-enriched entries were obtained. The top 20 entries showed that the signaling pathways including gastric cancer, TNF, hypoxia-inducible factor-1 (HIF-1), apoptosis, and mitogen-activated protein kinase (MAPK) were involved in the effect of BXD on GU (Figure 8).

Analysis of KEGG enrichment of BXD treating GU target proteins. The size of the circle represents the amount of protein enrichment, color represents the significance of enrichment, and the right-hand color gradient represents the P-value.
Molecular Docking Analysis
According to the “active ingredients–targets” network, the top 3 compounds by degree were baicalein, norwogonin, and skullcapflavone II. These compounds were combined with the core target protein TNF-α in the PPI network. Our analysis found that the active ingredient with the highest affinity for TNF-α (PDB: 6OP0) and PTGS2 (PDB: 5F1A) was norwogonin and the active ingredient with the highest affinity for AKT1 (PDB: 1UNQ) was baicalein. The molecular docking situation examined the active cavity of the compound and the protein. Figure 9 presents 3D and 2D docking modes.

Molecule docking mode of compounds and target proteins: (A) Baicalein and AKT1; (B) Norwogonin and TNF-α; and (C) Norwogonin and PTGS2.
From the analysis of the molecular docking results, it was found that the hydroxyl group in the core structure of baicalein formed hydrogen bonds with the amino acids ARG15, GLY16, GLU17, and LYS20 in the AKT1 protein, and the chromone core formed a π–π interaction with the amino acid ILE74. The binding energy of docking with AKT1 was −26.69 kJ/mol, indicating that baicalein could bind to the active site of the protein and that the active cavity had a good match. The hydroxyl and carbonyl groups in the core structure of norwogonin formed hydrogen bonds with the amino acids ILE58 and GLY122 of TNF-α, and the core structure formed a π–π interaction with the amino acids LEU57 of the A and B chains, which was empty and formed the protein activity. The cavity had a good match, and the docking binding energy of this compound with TNF-α was −31.63 kJ/mol. The carbonyl group in the core structure of norwogonin formed a hydrogen bond with the amino acid SER530 in the PTGS2 protein, and the hydroxyl group formed a hydrogen bond with the amino acid TYR385. In addition, the core structure formed a π–π interaction with the amino acids VAL349, LEU352, and ALA527. The hydroxyl group in the nuclear structure and the amino acid MET522 exerted an interaction force between the receptors. The docking binding energy of this compound with PTGS2 was −20.00 kJ/mol, and the norwogonin molecule fit completely into the active cavity of the protein.
Discussion
GU is a common disease affecting people throughout the world. 14 The occurrence of GU is the result of an imbalance between aggressive and protective factors in the gastric mucosa. Excessive ingestion of ethanol is one of the main causes of GU. 15 Recently, chemical drugs were used to treat GU; however, their long-term use may cause undesirable side effects, including nausea, abdominal pain, diarrhea, headache, sleep deprivation, and pneumonia. BXD has been used for the treatment of GU for many years. It is inexpensive and exhibits a remarkable curative effect with few side effects. However, the therapeutic compounds in BXD and their mechanisms of action against GU remain unknown. In this study, the administration of 95% ethanol by gavage was used to establish a rat model of GU. Treatment with BXD significantly reduced the pathological changes in the gastric tissue of rats with GU, by decreasing the expression of IL-2, IL-8, and TNF-α, which significantly attenuated the degree of inflammation. Network pharmacology and molecular docking technology were then used to systematically study the mechanism of action on GU, which screened the target protein to construct the interaction relationship between the active ingredients of BXD and the target protein. Through the analysis of the BPs and signal pathways enriched by the target proteins, BXD was shown to be a comprehensive treatment for GU targeting multiple targets and multiple signal pathways.
Network pharmacology uses network analysis to systematically and comprehensively analyze diseases, targets, and signal pathways, offering a means to clarify the pathogenesis of diseases. This study found that the main active ingredients of BXD were baicalein, norwogonin, and skullcapflavone II in the analysis of the “active ingredients–targets” network. There is growing evidence for the antiinflammatory activity of baicalein.16,17 Additionally, as a phenolic antioxidant, it also has biological activity against oxidative stress and inhibits the proliferation of gastric cancer cells.18,19 Baicalein could significantly attenuate the degree of GU damage, which reduced the oxidative stress factor-lipid peroxide malondialdehyde (MDA) and the serum inflammatory factors TNF-α, IL-2, and IL-6. The expression of the protective factor epidermal growth factor is upregulated, thereby promoting the repair of the gastric mucosa.20 Norwogonin is an important flavonoid compound with antioxidant activity, which has the effects of antioxidative stress and inducing cell apoptosis, 21 and has been reported to reduce the levels of MDA and the inflammatory factors TNF-α, cyclooxygenase-2, and NO, which inhibited oxidative stress and inflammation, thereby reducing the degree of GU damage. 22 It was speculated that norwogonin may be an active compound in the treatment of GU. Skullcapflavone II is also associated with the inhibition of reactive oxygen species, 23 which has an antiinflammatory effect. 24 Therefore, it can be speculated that these 3 active ingredients may be important in the treatment of GUs owing to their antioxidative stress effects.
PPI analysis found that the core proteins targeted by BXD in the treatment of GU included TNF-α, AKT1, and PTGS2. Among them, TNF-α is involved in the nuclear factor-κB-mediated inflammation pathway, which is closely related to ethanol-induced GU. 25 TNF-α can induce endothelial cell apoptosis and tissue inflammation, and a reduction in the content of TNF-α in tissues is closely related to the protection of GU. 26 TNF-α inhibitors can reduce the levels of prostaglandin E2 and heme oxygenase-1 in the serum and decrease the degree of GU loss in rats. 27 Herein, baicalein was shown to be an active compound in BXD, and BXD inhibited the production of TNF-α; these findings showed that the anti-GU effect of BXD may be based on these main components. AKT is a key target protein for multiple signaling pathway cascades. Reports show that AKT1 is a target protein for intervention in rats with liver dysfunction, spleen deficiency, and GU. 26 There is an abundance of evidence indicating that downregulation of the mRNA and protein expression of phosphatidylinositol 3-kinase (PI3K) and AKT, inhibition of the PI3K/AKT signaling pathway, and reducing the abnormal secretion of cytokines IL-2R and IL-4 have protective effects on the gastric mucosa. 28 PTGS2 is an inducible enzyme and its expression is activated by various factors. It plays an important role in promoting tumorigenesis, angiogenesis, and metastasis and is also involved in the process of gastric cell apoptosis. It is a key target protein for GU 29 ; as an inflammatory mediator, it is a rate-limiting enzyme that can catalyze the body's production of prostaglandin, which has a proinflammatory effect. 30 , 31 In this study, it was found from the molecular docking analysis that the key active components of BXD, such as baicalein, norwogonin, and skullcapflavone II, have a good affinity for these core proteins TNF-α, AKT1, and PTGS2.
GO enrichment analysis found that BXD targeted the cytoplasmic process, oxidative stress process, apoptosis process, protein binding, and enzyme binding. KEGG enrichment found that the target proteins involved multiple signaling pathways, including gastric cancer, TNF, HIF-1, apoptosis, and MAPK. This could promote the healing of GU by inhibiting the expression of TNF-α. 32 HIF-1α could regulate multiple target genes, including endothelial growth factors, thereby regulating various hypoxia-induced adaptive responses, such as angiogenesis, metabolism, and apoptosis.33,34 HIF-1α is activated under hypoxic conditions. Reactive oxygen species in gastric tissues act as a signal molecule that stimulates HIF-1α protein synthesis, which can activate the PI3K/AKT signaling pathway involved in the occurrence of inflammation of the stomach tissue.35 The apoptotic pathway was involved in the pathogenesis of GU, and regulating the FoxO3a/FasL/Caspase-8 pathway may inhibit apoptosis in gastric mucosal cells, which would assist in repairing the gastric mucosa. 36 MAPK could be stimulated by various extracellular factors to mediate signal transduction from the cell surface to the nucleus. P38MAPK is a key protein in the MAPK signaling pathway, which could be activated by inflammation. It may be expressed at a significant level in the gastric mucosal tissue of rats with GU and induce abnormal secretion of the inflammatory factors TNF-α, IL-1β, and IL-6, which subsequently trigger inflammation. 37 Therefore, it can be speculated that these signaling pathways are closely related to the mechanism of BXD in the treatment of GU.
Conclusions
In this study, network pharmacology and molecular docking technology were used to investigate the active ingredients, targets, BPs, and signaling pathways associated with the treatment of GU by BXD. Our results revealed that the important active components of BXD, including baicalein, norwogonin, and skullcapflavone II, may act on the target proteins TNF-α, AKT1, and PTGS2, leading to the inhibition of inflammation and oxidative stress. Moreover, BXD may affect the TNF, HIF-1, and MAPK signaling pathways. Our findings indicate that BXD exerted anti-GU effects through multiple components, multiple targets, and multiple pathways. This provides an alternative perspective to support the use of BXD as a general TCM prescription with clinical applications, including relieving GU.
Footnotes
Author Contributions
WJ wrote the original draft. XZ performed the experiments. CH and YZ conceived the study and designed the study. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 21904022), and sponsored by the Scientific Research Foundation of Shanghai University of Medicine and Health Sciences (grant no. E3-0200-22-201006-42).
Ethical Approval
All animal experimental procedures were in accordance with the Institutional Animal Committee of Shanghai University of Traditional Chinese Medicine (Permit No. PZSHUTCM190308019).
Statement of Human and Animal Rights
All animal experiments were conducted in accordance with protocols approved by the Institutional Animal Committee of Shanghai University of Traditional Chinese Medicine (Permit No. PZSHUTCM190308019).
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
