Abstract
Eucalyptus essential oils are well-known and used especially in upper respiratory tract pathologies or diseases as herbal drug preparations. In the present study, the in vitro angiotensin-converting enzyme 2 (ACE2) and lipoxygenase (LOX) enzyme inhibitory potentials of commercial Eucalyptus globulus Labill. and Eucalyptus citriodora Hook. essential oils were evaluated for their potential anti-coronavirus disease 2019 (COVID-19), and anti-inflammatory effects. In addition, the major components, 1,8-cineole and citronellal, were evaluated for their ability to bind at the active site of either human ACE2 or human 5-LOX using an in silico setting. Before activity evaluation, Eucalyptus globulus and E citriodora essential oils were analysed by GC/FID and GC/MS, where 1,8-cineole (30%), and citronellal (80%) were identified as the major components, respectively. The in vitro ACE2 inhibition was calculated as 94.9% for E globulus, and that of E citriodora essential oil as 83.4%. In vitro LOX inhibition experiments for essential oils in the same order showed inhibitions of 71.3 and 91.4%, respectively, at 20 µg/mL test concentrations in microplate-based fluorometric assays. In addition, protein–ligand docking, and interaction profiling was used to gain structural and mechanistic insights into the in silico ACE2 and LOX inhibitory potentials of the major Eucalyptus essential oil constituents, 1,8-cineole as well as citronellal. The resulting data supported the in vitro findings; however, further in vivo studies are needed to confirm the activity.
Introduction
Eucalyptus L. species, family Myrtaceae, as well as their preparations, are well known and have been used as aromatic and medicinal plants for centuries. Eucalyptus essential oils are used due to their antimicrobial and antioxidant activities, especially in pharmaceuticals and cosmetic products, as well as for flavorings and food preparations.
Eucalyptus globulus Labill. is the main source of commercial Eucalyptus essential oil, which is used as an antiseptic and to treat cough, cold, sore throat, and other related conditions. Both the leaf oil and extract have been reported for their antibacterial activity on respiratory system pathogenic bacteria and viruses. 1 Cineole-rich essential oils are inhaled to treat pharyngitis, bronchitis, and sinusitis. A number of in vitro and in vivo 1,8-cineole studies demonstrated anti-inflammatory, antimicrobial 2 and antiviral3,4 properties. Also, a double-blind placebo-controlled trial using the monoterpene 1,8-cineol demonstrated mucolytic, bronchodilation, and anti-inflammatory activities.5,6 Eucalyptus spp. with antioxidant, 7 anthelmintic, 8 antifungal, 9 anti-inflammatory, 10 antibacterial,1,11,12 analgesic and anti-inflammatory2,13 activities have been reported. The in vitro effectiveness of Eucalyptus against herpes viruses was previously evaluated. 14 In silico binding studies involving coronavirus disease 2019 (COVID-19)-related enzymes, such as proteases, were recently reported.15–16
It is well documented that angiotensin-converting enzyme (ACE) and its homolog ACE2 are membrane-associated zinc metallopeptidases, which play opposing roles in the regulation of the renin–angiotensin–aldosterone system through downstream signaling of the angiotensin 2 and angiotensin 1–7 effector peptides via the AT1 and Mas receptors, respectively. 17 Human ACE2, which is highly expressed in the heart, kidneys, and lungs, is not only a peptidase but also a functional receptor in cell membranes facilitating SARS-CoV-2 entry into host cells. 18 The interaction of SARS-CoV-2 with ACE2 is mediated by a self-contained unit of the viral spike protein, known as the receptor-binding domain (RBD). 19 The ACE2 N-terminal peptidase domain switches between an “open” (inhibitor-free) and a “closed” (inhibitor-bound) conformation, and this marked conformational change caused by inhibitor binding results in a shift in the before and after positions of the receptor's critical surface amino acid residues recognized by the SARS-CoV-2 RBD. Therefore, it is stated and suggested that ACE2 serves as a therapeutic target against COVID-19.2,19
Lipoxygenases (LOXs) in humans catalyze oxygen insertions into the 5th, 12th, and 15th positions of arachidonate stereospecifically through 6 discrete soluble enzymes known as 5-, 12-, and 15-LOX, each having A and B subtypes. LOX metabolites include significant pro- and anti-inflammatory mediators like resolvins, lipoxins, and leukotrienes, all of which play roles in the pathobiology of a wide range of respiratory conditions such as SARS-CoV-2 infections.20,21
In this present study, the in vitro ACE 2 and LOX inhibitory potentials of E globulus and Eucalyptus citriodora Hook essential oils were tested after confirming the essential oil chemical composition by GC/FID and GC/MS. In addition, to reveal the mode of action, in silico molecular modeling was performed using the major constituents 1,8-cineole and citronellal, to the best of our knowledge, for the first time.
Results and Discussion
Chemical Composition of Essential Oils
In the present study, the major components of E globulus essential oil were identified as 30.9% 1,8-cineole, 11.4% α-pinene, and 11.4% β-pinene (see Table 1). The major component for E citriodora essential oil was characterized as 79.9% citronellal. According to previous studies,22–24 the 1,8-cineol content of E globulus essential oil was found to be more than 50%. On the other hand, the percentage of citronellal in E citriodora essential oil was found as 50% to 70% in previous studies .7,25,26 The major monoterpenoid, citronellal, and other E citriodara essential oil components reported in Table 1 for the present study agree with literature findings.
Eucalyptus Essential oil Chemical Compositions.

Abbreviations: RRI, relative retention indices calculated against n-alkanes; %, relative percentages calculated from FID data; tr, trace (<0.1%).
In vitro-In silico Studies
The in vitro ACE2 inhibitory activity of E globulus, and E citriodora essential oils were tested at a concentration of 20 µg/mL, where the inhibition percentages were observed as 94.3% and 83.4%, respectively, as shown in Figure 1. Although E citriodora essential oil has not been studied for its antiviral effect, previous studies have reported its antifungal and antimicrobial properties.27,28 It was demonstrated in previous antiviral studies that citronellal may be the compound responsible for bioactivity.29,30 The antimicrobial and antiviral effects of citronellal-rich essential oils were significant, especially against HSV-type viruses. 31 Also, the E globulus essential oil was evaluated previously for its antiviral effect against H1N1, HSV1, and adenoviruses, where it was found effective against human pathogenic viruses.32,33 In a previous study conducted by our group with 1,8-cineol, the major component of E globulus essential oil, ACE2 enzyme inhibition was found to be 89.2% at a concentration of 5 µg/mL. 34 1,8-cineole is a compound with known antiviral potential being effective against rhinosinusitis viruses. 35 In previous in silico studies, major components of Eucalyptus essential oils were investigated in terms of their antiviral effect potential.15–16
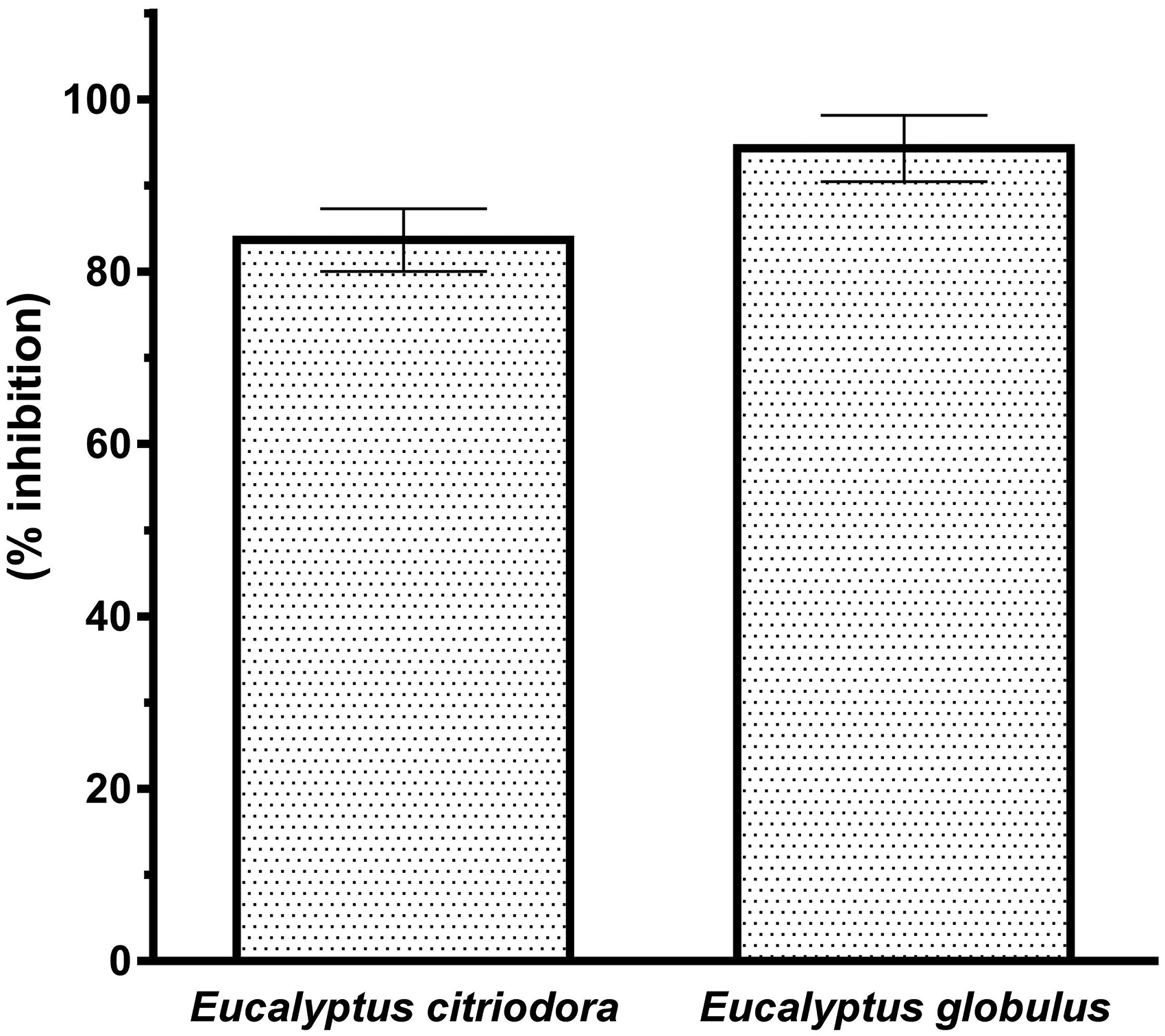
Angiotensin-converting enzyme 2 (ACE2) inhibition by Eucalyptus essential oils.
In the frame of this present study, an attempt was made to predict the ACE2 and 5-LOX inhibitory potentials of Eucalyptus essential oils by in silico docking of them with the major constituents 1,8-cineole, and citronellal, both in different enantiomeric forms. The best docking solutions were selected according to both computed docking scores, and additional structural criteria already described for bona fide ligands. 36
The ether oxygen of 1,8-cineole seemed to complete the tetrahedral geometry about the catalytic active-site Zn2+ ion, which was further coordinated by the imidazole imido nitrogen of each of His-374, His-378, and the carboxylate oxygen of Glu-402, as illustrated in Figure 2. The same coordination pattern is also retained in ACE2-citronellal complexes, with the zinc coordinated by the aldehyde oxygen of either (R)-( + )- or (S)-(-)-citronellal (Figure 2). Multiple Food and Drug Association -approved drugs that target zinc-dependent metalloenzymes are recognized to be coordinated to the Zn2+ ion cofactor through a zinc-binding group such as carboxylate, thiol, or hydroxamate, among others. 37 The Zn2+ coordination in determining the basic pharmacological principles of ACE2 inhibition is also exemplified by the in crystallo binding mode of the potent inhibitor MLN-4760, previously published. 38 It is also worth noting that the partially negatively charged ether, and carbonyl oxygens of 1,8-cineole and citronellal, respectively, can accept a hydrogen bond from the nearby (<2.5 Å) polar residue Tyr-515 observed in the present study.

Angiotensin-converting enzyme 2 (ACE2)-ligand docking calculations.
After ACE in vitro and in silico experiments, initially in vitro LOX inhibition results of E globulus and E citriodora essential oils at a concentration of 20 µg/mL were calculated as 71.3%, and 91.4%, respectively, as also shown in Figure 3. Nordihydroguaiaretic acid (NDGA), which was used as a positive control, inhibited the LOX enzyme by 98.9%. In our previous study, 1,8-cineole was also reported for LOX enzyme inhibition, where 37% inhibition was found. 34 E globulus and E citriodora essential oils were studied in rats for their anti-inflammatory effects in previous studies, and E citriodora essential oil was reported as active and effective. 13

LOX inhibition by Eucalyptus essential oils at 20 µg/ml and NDGA at 5 µg/mL. Abbreviations: LOX, lipoxygenase; NDGA, nordihydroguaiaretic acid.
In this present study, E citriodora essential oil showed remarkable results in terms of LOX enzyme inhibition. Citronellal, which is the major component of E citriodora essential oil, was examined for its anti-inflammatory activity, which was also reported in previous studies, supporting our findings. 39 The better LOX enzyme inhibition percentages of E citriodora essential oil compared to that of E globulus essential oil may be due to its relatively high citronellal content.
As is well known, human 5-LOX oxygenates arachidonate at carbon 5. Although arachidonate containing 20 carbons is the most frequent substrate for LOXs in mammals, linoleate, and linolenate (both 18 carbons) are 2 substrates for LOXs in plants as well. Thus, due to this 2-carbon difference in the lengths of mammalian, and plant fatty acyl chains, a plant 13-LOX enzyme is functionally similar to a human 15-LOX enzyme. Soybean 13-LOX, and human 5-LOX share a mean sequence identity of ∼21% and a mean sequence similarity of ∼67%, with a root-mean-square deviation of 1.1 Å over 368 aligned atoms. Furthermore, soybean 13-LOX is one of the most widely used LOX enzymes implemented in drug screenings.40–42 These justify our selection of soybean 13-LOX as a model enzyme to perform the LOX activity inhibition assays in this present work.
Most LOXs require a catalytic Fe2+ ion at their active site, although it can occasionally be replaced with an Mn2+ ion. In human 5-LOX, this non-heme iron is coordinated by 3 conserved histidine residues (His-367, His-372, His-550) and the carboxylate group of the C-terminal residue Ile-673. The results of the present in silico docking calculations reveal that both 1,8-cineole, and citronellal favor binding at a site rather distant (>10 Å) from the Fe2+ ion cofactor, as shown in Figure 4. The ether oxygen of 1,8-cineole is able to form a hydrogen-bonding interaction with the guanidino group of Arg-596. The hydrogen bond to Arg-596 appears to be the only substantial protein–ligand interaction observed also in the x-ray crystallographic structure of NDGA-bound stable 5-LOX. 43 Correspondingly, the aldehyde oxygen of citronellal can be engaged in a bifurcated hydrogen bond with Arg-596.

5- lipoxygenase (LOX)-ligand docking calculations.
It is worth mentioning that in the present study, the minor Eucalyptus essential oils constituents were also docked in silico. The selected minor components, (1R)-( + )-α-pinene, (1S)-(-)-α-pinene, and p-cymene, were docked onto human ACE2, and human 5-LOX enzymes. However, interestingly, the fully automated protein–ligand docking tool JAMDA failed to generate docking poses for the minor evaluated constituents in silico. This may be explained by the fact that none of the compounds contain a polar functional group, which may act as a hydrogen-bond acceptor, and thus enhance binding with the key hydrophilic residues lining the active-site cleft and/or the metal ion cofactor at the active site.
Overall, in the present study, 2 different Eucalyptus essential oils were evaluated experimentally for their potential in vitro and in silico LOX and ACE2 enzyme inhibitory activities. Also, to expand the findings and knowledge, major and minor Eucalyptus constituents were evaluated in this frame. All the experimental in vitro and designed in silico data reported in this present work are reported for the first time, to the best of our knowledge.
Conclusion
In silico data are valuable when supported by experimental data, such as in the present work. However, based on the initial experimental results, it is strongly suggested that Eucalyptus essential oils may be further evaluated in vivo as a potential anti-inflammatory, and antiviral tool against viruses and in particular against coronaviruses.
Experimental
Materials
Commercial E globulus and E citriodora essential oils were kindly supplied by Doallin Ltd, Istanbul; voucher samples are deposited at IMEF Herbarium (Herbarium No: IMEF 1313-1314). 13-LOX from Glycine max (soybean) (product no. L7395), and all other complementary chemicals were procured from Sigma-Aldrich. The angiotensin II-converting enzyme (ACE2) Inhibitor Screening Kit was acquired from BioVision (product no. K310).
GC-FID and GC/MS Analysis
Agilent 6890N GC equipment was used with the temperature of the FID at 300 °C. Simultaneous auto-injection on the same column under the same operational conditions was used to acquire the same elution order with the coupled GC/MS system. FID chromatograms were used to calculate relative percentages (%) of the separated substances.
GC/MS analyses were carried out using an Agilent 5975 GC-MSD system, with an Innowax FSC column (60 m × 0.25 mm, 0.25 µm film thickness) and helium as carrier gas (0.8 mL/min). The relative retention times (RRT) of essential oils were compared to those of authentic samples, or their relative retention index (RRI), which was according to a series of n-alkanes. Computer matching against commercial (Wiley GC/MS Library, MassFinder Software 4.0) and in-house “Başer Library of Essential Oil Constituents” built up with genuine compounds and components of known oils was used for the identification as previously reported. 34
ACE2 Enzyme Inhibition Assay
The experiments were performed using the “Angiotensin II Converting Enzyme (ACE2) Inhibitor Screening Kit”, as previously described.34,44 Eucalyptus essential oils were initially dissolved in DMSO (< 1%, v/v). Test samples (20 μg/mL) were added to each well, then the prepared enzyme solution (0.5 ng/μL ACE2) was added to all wells except the blank. Following the experimental period, a multimode microplate reader (SpectraMax i3) in fluorescence mode using Ex/Em = 320/420 nm wavelength was used for screening. All results are from triplicate experiments, and the percentage inhibition (%I) values were calculated as the mean.
LOX Enzyme Inhibition Assay
LOX activity inhibition assays were performed according to the modified spectrophotometric method described by Baylac and Racine, 45 and our previous work.34,44 The concentration of Eucalyptus essential oils was 20 μg/mL. As the positive control, NDGA was used. Experiments were carried out in triplicates, and results are reported as means.
Computational Analyses
The 3-dimensional conformers of (i) 1,8-cineole (PubChem Compound ID: 2758), (ii) (R)-( + )-citronellal (PubChem Compound ID: 75427), and (iii) (S)-(-)-citronellal (PubChem Compound ID: 443157) in SDF format were downloaded from the PubChem open chemistry database. 46 The x-ray crystallographic structures of (i) the zinc metallopeptidase domain of human recombinant ACE2 complexed with the dipeptide mimetic inhibitor MLN-4760 (also known as ORE1001 or GL1001) (PDB ID: 1R4L) and (ii) human stable 5-LOX complexed with the natural product inhibitor NDGA (PDB ID: 6N2W) were downloaded from the RCSB Protein Data Bank and used as input to UCSF Chimera v1.11.2 47 for protein preparation purposes. The proteins were prepared by preserving only the highest occupancy sets of alternate location atoms and replacing incomplete amino acid side chains with complete ones using the Dunbrack rotamer library. 48 The ligands were docked in the presence of metal (Zn2+/Fe2+) ion cofactors and structurally relevant water molecules into the active-site clefts of the proteins using the JAMDA molecular docking tool49–51. The appropriate protonation states and hydrogen atom coordinates were assigned to the proteins before docking using the inherent Protoss hydrogen prediction tool. 52 The binding site was defined to include all protein atoms within a radius of 6.5 Å from any atom of the reference ligand (MLN-4760/NDGA). Molecular docking was executed with medium precision. Interaction profiling after docking was achieved using the GeoMine geometric search engine.53,54
Statistical Analyses
GraphPad Prism software Version 7.02 (La Jolla) was used for all statistical analyses. Enzymatic assay data were expressed as mean ± standard deviation (mean ± SD). Different groups were compared by 1-way analysis of variance (followed by Dunnett's post hoc test) or paired samples t-test. A P < .05 was considered statistically significant.
Footnotes
Acknowledgments
This PhD and research project was supported by Anadolu University Scientific Research Projects Commission (BAP 2005S058 and BAP 2103S017). Part of this work was presented online at third Annual Conference of the Centre for Natural Products Discovery (3. CNPD), Liverpool John Moores University (UK). The authors would like to dedicate this work to the liverwort expert Professor Yoshinori Asakawa and celebrate his 80th birthday.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Anadolu Üniversitesi (grant number 2103S017).
