Abstract
This is the first report of the high-performance liquid chromatography and gas chromatography–mass spectrometry profile of a herbal mixture (HM) made of Juniperus oxycedrus L. (redberry juniper) berries, inner bark of Betula pendula Roth. (silver birch), and grains of Avena sativa L. (oat), and its effect on the No. of micronuclei (MN) in human lymphocytes and toxicity toward Artemia salina. Constituents represented by over 1000 µg per g of methanol dry extract were gallic acid, protocatechuic acid, and amentoflavone. The methanol extract of the HM at a concentration of 2.0 μg/mL decreased MN frequency by 38.3%, which was more than 3 times greater than that of the radioprotectant amifostine. The essential oil isolated from the HM was composed mainly of β-myrcene (32%) and showed weaker toxicity toward Artemia salina than the positive control after both incubation periods (24 h and 48 h). These findings suggest that the examined HM, beside its ethnopharmacological relevance on the elimination of renal calculi, also significantly reduces the No. of MN in human lymphocytes.
Introduction
Nephrolithiasis is a condition caused by the formation and displacement of crystal agglomerates, also known as stones or calculi, in the urinary tract. 1 The incidence of kidney stone formation is increasing across the world.2,3 In order to eliminate renal colic and calculus many supplements have emerged. Development of new drug formulations from well-known folk remedies may decrease the side effects of drugs, while maintaining the therapeutic effect. 4 A decoction made of the berries of Juniperus oxycedrus L., inner bark of Betula pendula Roth., and grains of Avena sativa L. is used in the Balkan peninsula to eliminate kidney stones.
Although there are numerous studies that validate the medicinal relevance of the individual mixture components5–7 there are no data for the evaluation of this herbal mixture (HM). Therefore, in the present study, high-performance liquid chromatography (HPLC) analysis of the methanol extracts of the HM and individual mixture components was performed, as well as determination of the protective effect of the methanol extracts on human peripheral blood lymphocytes by cytokinesis-block micronucleus assay (CBMN). Furthermore, gas chromatography–mass spectrometry (GC-MS) analysis and acute toxicity to Artemia salina (brine shrimps) were evaluated for the essential oils of J. oxycedrus berries and HM. The headspace (HS) volatiles of the individual herbal materials and the mixture were also determined. The ratio of HM constituents was the same as in the HM traditionally used to treat kidney stones.
Results and Discussion
HPLC Analysis
The chemical composition of the methanol extracts of J. oxycedrus berries, inner bark of B. pendula, grains of A. sativa, and the HM were determined using HPLC-UV analysis. Phenolic compounds were identified according to their retention times and UV spectra, which were compared with the available commercial standards. Quantification was performed using calibration curves obtained for phenolic acids (gallic acid), flavonoids (rutin and quercetin), and procyanidin B2. Results are given in Table 1.
Chemical Composition of Methanol Extracts of Juniperus oxycedrus Berries (M1), Inner Bark of Betula pendula (M2), Grains of Avena sativa (M3), and Herbal Mixture (M4) (μg/g of dry Extract).
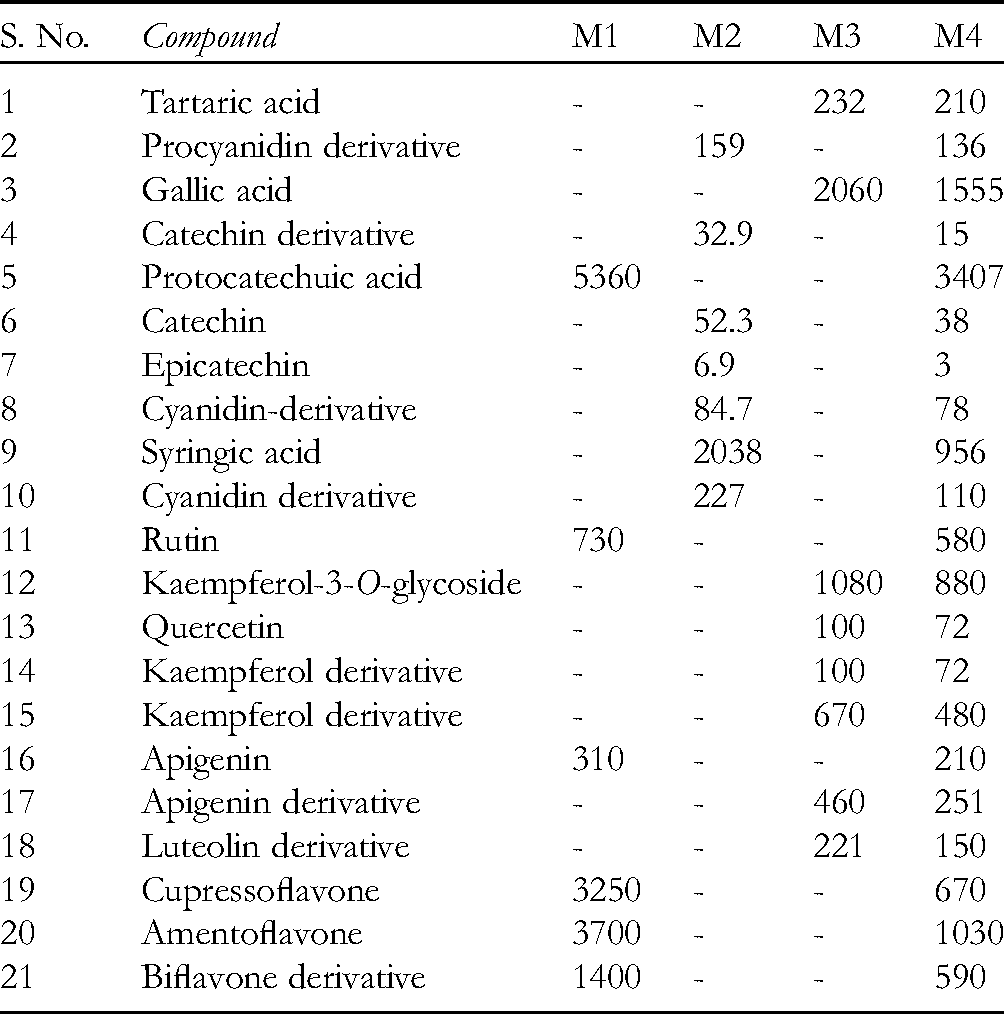
The most significant difference between the methanol extracts was the presence of bioflavonoids, detected only in the methanol extract of J. oxycedrus berries and HM.
Kaempferol-3-O-glycoside and gallic acid were the most abundant compounds of the A. sativa methanol extract (1080 and 2060 μg/g of dry extracts, respectively), while several derivatives of kaempferol, apigenin, and luteolin were detected in lower amounts. It was found that oat products possess an impact on uric acid excretion. In the conducted experiments, patients with elevated uric acid levels were treated by giving them an herbal tea containing 75% of A. sativa, which reduced uric acid levels. 8
Birch bark consists of brown inner bark or cambium (approximately 75%) and white outer bark (approximately 25%). In the sample of inner bark examined, cyanidin and a procyanidin derivative were detected, as well as catechin, epicatechin, and syringic acid.
Since standards of all identified compounds were not available, their identification was achieved by comparison with available standards as follows: compounds 14 and 15 were identified as kaempferol derivatives, compounds 17 and 18 as apigenin and luteolin derivatives, while compounds 19, 20, and 21 were identified according to literature data. 9 Also, compounds 2, 8, and 10 exhibited characteristic UV absorption bands indicating the presence of procyanidin B2 and cyanidin-3-O-glucoside derivatives, according to available standards.
Our results are in agreement with those previously published. Emmons and Peterson 10 have reported the following phenolic compounds in fractions of oat (groats and hulls): gallic acid, protocatechuic acid, p-hydroxybenzaldehyde, p-coumaric acid, vanillic acid, caffeic acid, vanillin, ferulic acid, sinapic acid, and avenanthramide. Apigenin, luteolin, and tricin are 3 major flavones found in oat flour and were identified in the vegetative plant only as glycosides. 11 Kaempferol glycosides were isolated from the bran of A. sativa. 12
Biflavonoids were detected as the main compounds of J. oxycedrus berries methanol extract, which is in agreement with previously published results. 9
Phenolic acids (syringic acid 4-β-glucopyranoside), flavanols {( + )-catechin 7-O-β-D-xylopyranoside and ( + )-catechin} and procyanidin dimers and trimers were determined in the inner bark using LC/MS analysis.13,14 It was demonstrated that the inner bark contains high amounts of flavonoids, arylbutanoids, diarylheptanoids, simple phenolic compounds, phenolic acids, lignans, and procyanidins.15,16
Chemical Composition of HS Volatiles and Essential Oils of J. oxycedrus Berries and HM
The qualitative composition and relative abundance of the HS volatile compounds of the berries of J. oxycedrus (HS1), inner bark of B. pendula (HS2), grains of A. sativa (HS3), HM (HS4), essential oils of J. oxycedrus berries (EO1), and HM (EO2) are given in the Table 2.
Composition (%) of Headspace (HS) Volatile Compounds of the Berries of Juniperus oxycedrus (HS1), Inner Bark of Betula pendula (HS2), Grains of Avena sativa (HS3), Herbal Mixture (HM) (HS4), and Essential Oils of J. oxycedrus Berries (EO1) and HM (EO2).

RI—experimental linear retention indices relative to C8–C40 alkanes on an HP-5MS column calculated in accordance with experimental conditions for the GC-MS analysis. bAI—Adam's retention indices. 17 cSalido et al 18 t: traces (<0.1%); (-): not detected.
Abbreviations: EO1, essential oil of J. oxycedrus berries; EO2, essential oil of the herbal mixture; HS1, dried berries of J. oxycedrus; HS2, inner bark of B. pendula; HS3, grains of A. sativa; HS4, herbal mixture;MH, monoterpene hydrocarbons; MO, oxygenated monoterepenes; SH, sesquiterpene hydrocarbons; SO, oxygenated sesquiterpenes; O, Others.
The most dominant component of sample HS1 was β-myrcene (65.4%), accompanied by α-pinene (23.9%) and limonene (6.8%). The dominant component of the essential oil of the berries was β-myrcene (37%), accompanied by α-pinene (13%), β-caryophyllene (8.7%), α-humulene (6%), and germacrene D (7.7%). The amount of β-myrcene was twofold smaller than that of the HS sample. Similarly, the essential oil isolated from the HM was composed mainly of β-myrcene (32%), but the amount of monoterpenes (43.2%) was twofold smaller than that of the HS volatiles of the HM (96.8%). The main monoterpene hydrocarbons of the HM (HS4) were β-myrcene (71.8%), α-pinene (17.2%), and β-pinene (1.2%), followed by a small amount of the sesquiterpene hydrocarbon, germacrene D (1.1%). Dominant HS volatiles of the inner bark of B. pendula (HS2) were primary aldehyde and alcohols n-hexanal (51.0%), n-hexanol (17.9%), and n-pentanol (6.7%), while monoterpenes were detected in significantly lower amounts compared to the other 2 samples (6.6%). The only detected monoterpene was β-pinene (6.6%).
Sesquiterpenes composed the major part of the HS volatiles of A. sativa grains, amounting to 51.6% of the total HS volatiles. The most abundant compounds (HS3) were the sesquiterpenes, germacrene D (46.5%), and α-humulene (5.1%), followed by the monoterpenes α-pinene (9.4%) and β-myrcene (5.6%).
The essential oil of the HM contained similar amounts of monoterpenoids and sesquiterpenoids (43.2% and 37.0%, respectively). Higher amounts of β-caryophyllene (12.7%) and germacrene D (12.1%) were detected in the essential oil of the HM than in the essential oil of the berries (8.7% and 7.7%, respectively), as shown in Figure 1.

Main volatile compounds of essential oils of Juniperus oxycedrus berries (EO1) and herbal mixture (HM) (EO2).
Biological Activities of J. oxycedrus Berries, Inner Bark of B. pendula, Grains of A. sativa, and HM
Cytokinesis-Block Micronucleus Assay
The analysis of micronuclei (MN) in cultured lymphocytes is applied as a method for genotoxic studies . 19 In this study, the methanol extracts of J. oxycedrus berries (MJO), inner bark of B. pendula (MBP), grains of A. sativa (MAS), HM, and pure compounds at concentrations of 1.0, 2.0, and 4.0 µg/mL were subjected to CBMN assay. The frequencies and distribution of MN in human lymphocytes were scored. The results are presented in Table 3 and Figure 2.

Frequency of micronuclei (MN) in cell cultures of human lymphocytes treated with different concentrations of methanol extracts of Juniperus oxycedrus berries (M1), inner bark of Betula pendula (M2), grains of Avena sativa (M3), herbal mixture (HM) (M4), and pure compounds.
Incidence of Micronuclei (MN), Cytokinesis-Block Proliferation Index (CBPI), Distribution of MN per Cells, and Frequency of MN in Cell Cultures of Human Lymphocytes Treated With Different Concentrations of Methanol Extracts of Juniperus oxycedrus Berries (M1), Inner Bark of Betula pendula (M2), Grains of Avena sativa (M3)
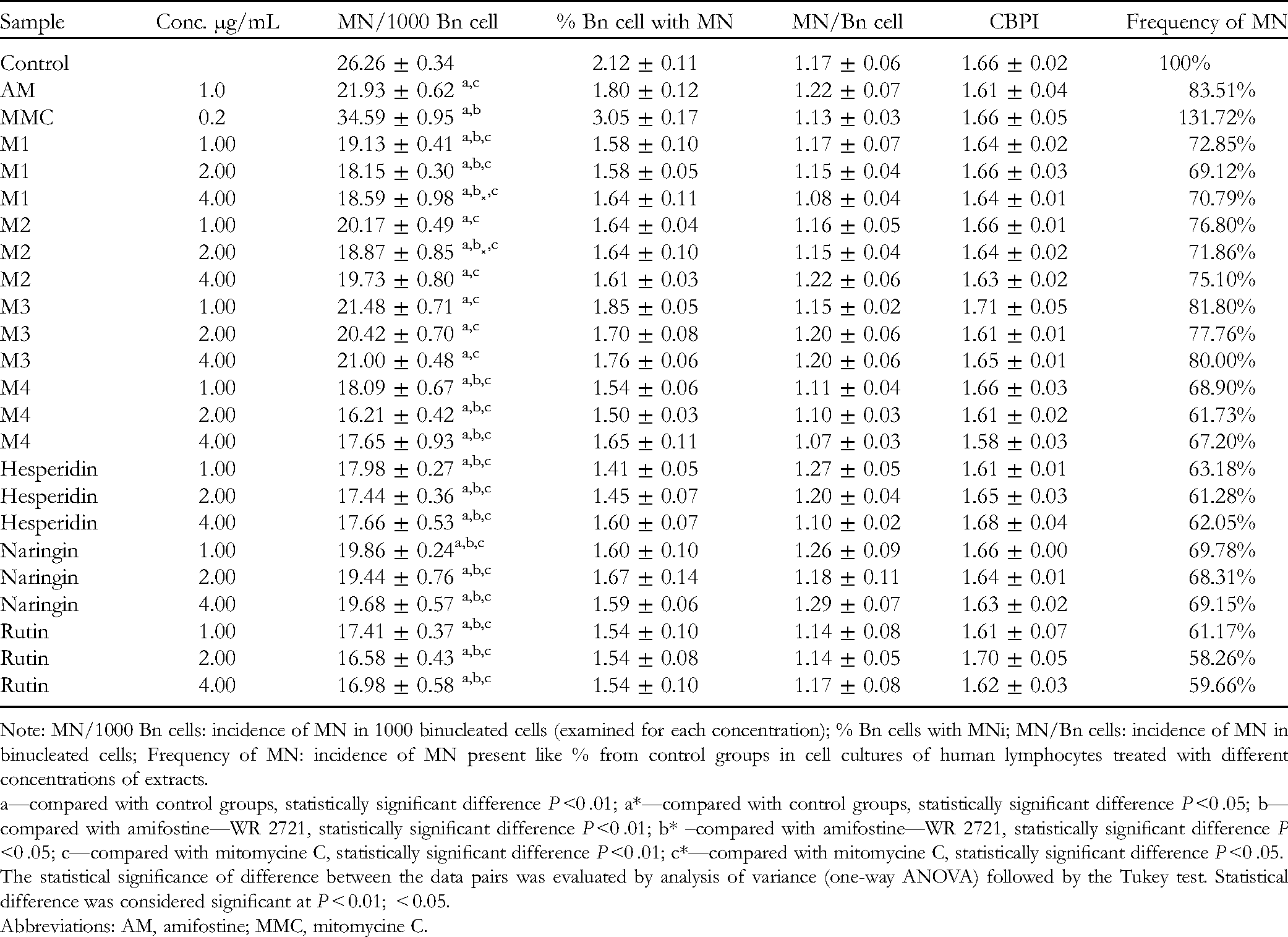
Note: MN/1000 Bn cells: incidence of MN in 1000 binucleated cells (examined for each concentration); % Bn cells with MNi; MN/Bn cells: incidence of MN in binucleated cells; Frequency of MN: incidence of MN present like % from control groups in cell cultures of human lymphocytes treated with different concentrations of extracts.
a—compared with control groups, statistically significant difference P <0 .01; a*—compared with control groups, statistically significant difference P <0 .05; b—compared with amifostine—WR 2721, statistically significant difference P <0 .01; b* –compared with amifostine—WR 2721, statistically significant difference P <0 .05; c—compared with mitomycine C, statistically significant difference P <0 .01; c*—compared with mitomycine C, statistically significant difference P <0 .05.
The statistical significance of difference between the data pairs was evaluated by analysis of variance (one-way ANOVA) followed by the Tukey test. Statistical difference was considered significant at P < 0.01; < 0.05.
Abbreviations: AM, amifostine; MMC, mitomycine C.
All tested compounds (in minimal doses of 1 μg/mL) exerted a beneficial effect by decreasing DNA damage of human lymphocytes in the range of 18.2% to 31.1%. Among the tested extracts, the most prominent effect was exhibited by the methanol extract of the HM at a concentration of 2.0 μg/mL, which gave a significant decrease (P <0 .01) in the MN frequency of 38.3%, which was threefold higher than the effect of amifostine (MN frequency of 11.4%). Also, treatment of the cell cultures with the methanol extract of the HM at concentrations of 1 μg/mL and 4.0 μg/mL affected a significant decrease (P <0 .01) in the frequency of MN (31.1% and 32.8%, respectively) compared with the control cell cultures (Figure 2). The methanol extracts of individual samples (M1, M2, and M3) at a concentration of 2.0 μg/mL exhibited a twofold higher activity than that of amifostine, while concentrations of 1 μg/mL and 4.0 μg/mL showed slightly lower activity, but were still more effective than amifostine.
The tested pure substances were selected due to their widespread representation in plant extracts. Among them, rutin exhibited the highest activity at a concentration of 2.0 μg/mL and gave a decrease in the MN frequency of 41.7%, which was slightly higher than that of the methanol extract of the HM (38.3%), even though it was detected as one of the compounds of the mixture. Hesperidin and naringin caused a slightly lower effect than rutin. It was noticed that the pure substances were slightly more efficient than that of the examined methanol extracts in decreasing the MN frequency.
The comparable cytokinesis-block proliferation index (CBPI) for extracts, positive control, and untreated cells confirmed the absence of the impact on cell proliferation.
The methanol extracts of the HM and mixture constituents showed the most significant reduction in the MN frequency at a concentration of 2.0 μg/mL, whereas concentrations of 1 μg/mL and 4 μg/mL were less effective, but still more effective than amifostine.
Acute Toxicity—Artemia Salina Test
Acute toxicity against Artemia salina is used for the preliminary assessment of the safety and potential pharmacological application of natural products. The results of the bioassay could provide information about the cytotoxic properties of the tested extract considering good correlation between the assay and cytotoxic activity against some human solid tumors.20,21 The results of the acute toxicity assay of the tested essential oils (berries—EO1 and HM—EO2) are summarized in Table 4. The final concentrations of the tested EO1 and EO2 in the Petri dishes were in the range of 4.57 to 73.125 μg/mL and 16.25 to 127.5 μg/mL (respectively), while the final concentration of Dimethyl sulfoxide (DMSO) was less than 1% (v/v).
Acute Toxicity of Examined Samples and SDS (Positive Control) on Artemia salina, Expressed as LC50 in µg/mL After 24 h and After 48 h.

Abbreviations: EO1, essential oil of the berries; EO2, essential oil of the herbal mixture; LC50, lethal concentration to 50% of nauplii; SDS, sodium dodecyl sulfate.
The statistical significance of difference between the data pairs was evaluated by analysis of variance (ANOVA), followed by the Tukey test. Statistical difference was considered significant at P < 0.0001. There is a statistically significant difference (P < 0.0001) in activity between each of the oils and the SDS, as well as between the oils, SDS, and control sample (dimethyl sulfoxide [DMSO]).
To the best of our knowledge, there is no study regarding the acute toxicity of the essential oils of J. oxycedrus berries and the HM in the Artemia salina test. Based on the survival of nauplii after 24 h of incubation, EO1 showed higher toxicity than both the remaining samples, EO1/EO2 (almost twofold stronger) and EO1/SDS (1.2 times higher). Higher amounts of α-pinene and β-myrcene were detected in the essential oil of the berries, EO1 (13% and 37%, respectively) than in the essential oil of the HM, EO2 (4.1% and 32%), while the amounts of β-caryophyllene and germacrene D (8.7% and 7.7%, respectively for EO1) were smaller (12.7% and 12.1%, respectively for EO2). Previously, the methanol extract of the berries of Juniperus drupacea from Turkey was found to be moderately toxic against brine shrimps, with a Lethal concentration to 50% of nauplii (LC50) value of 489.47 ± 27.8 µg/mL. 21 For the incubation period of 48 h, both oils exhibited lower toxicity than SDS, EO1 being about 3 times less and EO2 about 5 times lower.
Experimental
Plant Materials
Berries of J. oxycedrus were collected from Klobuk, province of Trebinje (coordinates 42° 42′ 28.19″ N and 18° 31′ 33.59″ E, R. Srpska). Red-brown mature fruits were air-dried, lyophilized, and stored in a dark place. Grains of A. sativa were purchased at a health food store. Inner bark of B. pendula was collected in city park Vranje (coordinates 42.5521° N and 21.8989° E, Serbia). The voucher specimens have been deposited in the Herbarium collection at the Department of Biology and Ecology, Faculty of Science and Mathematics, University of Niš under the acquisition numbers: 14611-F, 14612-F, 14613-F, respectively.
Preparation of Methanol Extracts of Berries, Inner Bark, Grains, and HM
The extractions of individual species were performed in triplicate. Two g of each sample was chopped and subjected to ultrasound-assisted extraction with 20 mL of methanol (Sigma-Aldrich; purity ≥ 99.9%) using an ultrasound bath (UZK 8; Maget,) for 30 min; after that, the extracts were left in the dark (room temperature) overnight. Dry residues of the extracts were obtained using a rotary evaporator (KNF Laboxact) with the water bath set at 40 °C. The mixture of chopped dried berries (1.1 g), inner bark of B. pendula (0.5 g) and grains of A. sativa (1.6 g) was extracted with 30 mL of methanol in the same way as the individual components of the mixture. The extract yields were 17.4 ± 0.1% for J. oxycedrus berries, 1.33 ± 0.05% for B. pendula inner bark, 1.78 ± 0.04% for grains of A. sativa, and 18.01 ± 0.1% for mixture. The dry methanol extracts were prepared prior to HPLC analysis and MN assay.
Isolation of Essential Oils
The air-dried samples (11 g J. oxycedrus berries, 5 g B. pendula inner bark, and 16 g A. sativa grains) were cut into small pieces and subjected to hydrodistillation using a Clevenger-type apparatus for 2 h. The obtained essential oil was extracted with diethyl ether and dried over anhydrous sodium sulfate. After filtration, the solvent was removed under reduced pressure and stored at +4 °C prior to analysis. The essential oil of J. oxycedrus berries was obtained in the same way as the HM. The yield of essential oils was 0.66% and 0.61%, respectively, based on the dried weight of the samples.
Sample Preparation for HS Analysis
For HS analysis of the HM, chopped berries of J. oxycedrus (0.5 g), inner bark of B. pendula (0.25 g), and grains of A. sativa (0.8 g) were placed in a vial for HS analysis and moistened with 1 mL of distilled water. Samples of the individual ingredients of the mixture were prepared in the same way using 1 g of each ingredient.
GC-MS Analysis
The samples were analyzed by a 7890/7000B GC/MS/MS triple quadrupole system in MS1 scan mode (Agilent Technologies) equipped with a Combi PAL sampler and HS for G6501B/G6509B. Details about GC-MS analyses, identification, and calculation of relative amounts of volatile compounds have been reported previously. 22
In short, a fused silica capillary column HP-5MS (5% phenylmethylsiloxane, 30 m × 0.25 mm, film thickness 0.25 µm) was used. The injector and interface operated at 250 and 300 °C, respectively. Temp. program: from 50 to 290 °C at a heating rate of 4 °C/min. The carrier gas was helium with a flow of 1.0 mL/min. The 500 µL HS volatile components and 1 µL essential oil solutions in hexane (1:100), respectively, were injected. Split ratio was 40:1. Post-run: back flash for 1.89 min, at 280 °C, with helium pressure of 50 psi. MS conditions were as follows: ionization voltage of 70 eV, acquisition mass range of 40 to 440, and scan time 0.32 s. The percentage composition of the samples was computed from the TIC peak areas without any corrections. Constituents were identified by comparison of their linear retention indices (relative to C8–C40 n-alkanes on the HP-5MS column) with literature values and their MS with those from Wiley 6, NIST11, and Agilent Mass Hunter Workstation B.06.00 software by application of the AMDIS software (Automated Mass Spectral Deconvolution and Identification System, Ver. 2.1, DTRA/NIST, 2011).
HPLC Analysis
HPLC analysis was performed on an Agilent, Zorbax Eclipse XDB-C18, 5 μm, 4.6 mm × 150 mm column using an Agilent 1200 series liquid chromatography (equipped with a diode array detector [DAD]), Chemstation Software (Agilent Technologies), a quaternary pump, an online vacuum degasser, autosampler, and a thermostated column compartment. The mobile phase consisted of 2 solvents: water–formic acid (1%) (A) and methanol (B), starting with 30% B at 5 min, and using a gradient to obtain 70% B at 20 min and 90% B at 25 min and finished with 90% B at 40 min. The flow rate was 0.5 mL/min, the injection volume 20 μL, and the column was thermostated at 25 °C. Spectral data from all peaks were accumulated in the range of 190 to 400 nm, and chromatograms were recorded at 254 and 350 nm. Identification was conducted using retention times and UV spectra, which were compared with the corresponding commercial standards. The following standards were used: tartaric acid, procyanidin B2, catechin, gallic acid, syringic acid, protocatechuic, rutin, kaempferol-3-O-glycoside, quercetin, kaempferol, apigenin, and luteolin (all standards were supplied by Sigma-Aldrich). Quantification was performed using calibration curves obtained of any standard except for derivatives of the corresponding compound for which calibration curve of that compound was used and also for cupressoflavone, amentoflavone, and biflavone derivative for which quercetin calibration curve was used. The amount of identified compounds was expressed as μg per g of dry extract weight (μg/g).
Cytokinesis-Block Micronucleus Assay
CBMN was performed as previously described.19,23 The cell culture lymphocytes were treated with 1.0, 2.0, and 4.0 μg/mL of the examined methanol extracts and available pure compounds (hesperidin, naringin, and rutin; Sigma-Aldrich).
Amifostine WR-2721 (98% S-2[3-aminopropylamino] ethylphosphothioic acid; Marligen-Biosciences) at a concentration of 1 μg/mL was used as a positive control. The alkylating agent mitomycin-C (MMC, Bristol-Myers Squibb), prepared by diluting the drug in phosphate buffer (PBS), was added at a final concentration of 0.2 µg/mL to the lymphocytes cultures and used as a negative control. Three experiments were performed for each sample. The results are expressed as the means ± standard deviation (SD). The statistical analysis was performed using Origin software package version 7.0. The statistical significance of difference between the data pairs was evaluated by analysis of variance (one-way ANOVA), followed by the Tukey test. Statistical difference was considered significant at P < .01 and P < .05.
Acute Toxicity—Artemia Salina Test
Acute toxicity in Artemia salina (brine shrimps) was evaluated using the method previously described by Radulović and colleagues. 24 Two teaspoons of lyophilized cysts of Artemia salina were added to 1 L of the artificial seawater. The suspension was thermostated at 28 °C, aerated with a strong airflow, and kept under constant illumination for 48 h, after which most of the cyst hatched. Freshly hatched nauplii (20 individuals) were transferred into Petri dishes containing 20 mL of artificial seawater. One hundred microliters of tested oil of each concentration (14.62; 7.31; 3.65; 1.83; 0.91 mg/mL for EO1 and 25.5; 13; 6.5; 3.25; mg/mL for and EO2) was added into Petri dishes as well. So, the final concentrations of the tested EO1 and EO2 in the Petri dishes were in the range of 4.57 of 73.125 μg/mL and 16.25 of 127.5 μg/mL, respectively. Brine shrimps were not fed during the test. The tested samples were not aerated, and the test dishes were left at room temperature, under constant illumination. DMSO was inactive against A. salina nauplii and it was used as a negative control, whereas varying concentrations of sodium dodecyl sulfate (SDS) were used as the positive control. Further, 3 Petri dishes were filled only with the equal volume (as for all other tests) of seawater and all nauplii survived after 24 h and after 48 h. Brine shrimps were not fed during the test. The tested samples were not aerated, and the test dishes were left at room temperature, under constant illumination. All the tests were performed in triplicate. Dead nauplii were counted after 24 h and 48 h. LC50 was determined after statistical analysis according to Hamilton et al 25
Conclusion
Results obtained by HPLC analysis of the methanol extract of the HM and individual mixture components revealed the presence of phenolic acids, flavonols, flavonols glycosides, bioflavonoids, flavones, and flavan-3-ols derivatives. Major volatile components of the HM essential oil were β-myrcene (32%), β-caryophyllene (12.7%), and germacrene D (12.1%). Methanol extracts of the HM and its constituents reduced the frequency of MN in a concentration-dependent manner, with the medium concentration of 2.0 μg/mL being the most effective and even more effective than the positive control. The essential oil of the HM showed weaker toxicity than the positive control after both time periods (24 h and 48 h). These findings suggest that this HM, beside its ethnopharmacological relevance on the elimination of renal calculi, also significantly reduces the No. of MN in human lymphocytes. As the No. of MN serves as an indicator of DNA damage, the results indicate that the tested extracts at a concentration of 2 μg/mL show a protective effect on DNA.
Footnotes
Acknowledgments
The authors acknowledge the Ministry of Education, Science and Technological Development of Serbia for financial support (Agreement No. 451-03-9/2022-14-200124).
Author’s Contribution
Dr Ivana Zlatanović: Literature research; Designing a study; Experimental work (plant collecting, HPLC analysis); Analysis of the results; and Writing the paper. Dr Miroslava Stanković: cytokinesis block micronucleus test. Jovana Ickovski: acute toxicity in Artemia salina (brine shrimps) test. Ivana Zrnzević: GC/MS analysis. Dr Gordana Stojanović: Analysis of the results; Writing the paper; And Supervision of all of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Science and Technological Development of Serbia (grant number No. 451-03-68/2022-14/200124).
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
