Abstract
Mentha spp. are used in the food and pharmaceutical industry; the plants are characterized by natural interspecies hybridization. In this study, knowledge of the chemical composition of Mentha spp. was broadened by focusing on plants grown in a geographically small region of Estonia. The antibacterial activity of Mentha spp. essential oils and water extracts was evaluated. Polyphenolic water extracts of M. × villosa Huds., M. × suaveolens Ehrh., and M. × gracilis Sole were tested for the first time on Escherichia coli and Staphylococcus aureus. Leaves of cultivated and wild-grown plants (n = 33) were collected. The microdistilled essential oil composition reflected the diversity within the genus Mentha. Determined by gas chromatography-mass spectrometry (MS), major compounds were cis-piperitone oxide, carvone, linalool, menthol, and menthofuran. Based on high-performance liquid chromatography-ultraviolet-MS/MS analyses of the water extracts, no species-specific polyphenolic compounds could be proposed. Abundant polyphenols were rosmarinic acid, salvianolic acid B, and eriocitrin. Essential oils exhibited antibacterial activity on E. coli and S. aureus by the broth dilution method. Water extracts showed activity only against S. aureus. This study supports the use of Mentha spp. as health-promoting ingredients in food. However, further studies are still needed to widen the knowledge of the chemical composition of these plants.
Mentha spp. belong to the family Lamiaceae, tribe Mentheae, and genus Mentha L. The economic importance of Mentha spp. (mints) is evident from the fact that their essential oils and dried and fresh plant material are in daily use in confectionary, beverages, bakery, cosmetics, pharmaceuticals, and pesticides. 1 Mentha × piperita L. is mainly cultivated in the USA, Morocco, Argentina, France, Hungary, Italy, and Switzerland. 2 Cultivation of Mentha spicata L. is largely located in the USA. 3 Solely in North America, during the year 2004, 3318 tons of M. × piperita and 346 tons of M. spicata were produced. 4
Mints are perennial, fast-growing plants and generally tolerate a wide range of agroclimatic conditions and are thus distributed in all 5 continents. 5 It is believed that the genus originated in the Mediterranean basin and, from there, spread to the rest of the world by both natural and artificial means. 2,3 The taxonomy of mints is complicated and several classifications varying in the number of recognized species have been proposed in the past. 4 Studies have attempted to describe the genetic relationships in Mentha using morphological, 6 cytological, 5 and genetic characteristics. 7 According to the latest taxonomic classification, the genus Mentha contains 18 species and is divided into 4 sections (Tubulosae, Eriodontes, Pulegium, Mentha). Interspecies hybridization exists in the section Mentha that includes 8 species: M. suaveolens Ehrh., M. longifolia (L.) L., M. spicata L., M. arvensis L., M. canadensis L., M. aquatica L., M. dahurica Fisch. ex Benth., and M. japonica (Miq.) Makino. The systematics of these 8 species is especially complex because of the easy hybridization. The 5 basic Eurasian and African species, M. arvensis, M. aquatica, M. spicata, M. longifolia, and M. suaveolens, have produced 11 naturally occurring hybrids of which the best known is M. × piperita L. Furthermore, within the genus, more than 3000 names of species, hybrids, and cultivars have been published since 1753, which is the starting date of modern nomenclature. However, about 95% of these names are either synonyms or illegitimate. 8 Currently, most of the commercially important mints are either hybrids or amphiploids. 2
The pharmacological effects of mints are usually linked to the presence of 2 main groups of secondary metabolites: essential oils and polyphenols. Essential oils of mints are mainly composed of monoterpenes and sesquiterpenes, the content and composition of which varies according to species, 9 plant growth stage, 10 geographical origin, 11 and environmental conditions. 12,13 The main polyphenols reported in Mentha spp. include derivatives of caffeic acid and glycosidic forms of the flavonoids luteolin, apigenin, eriodictyol, and naringenin. Previous studies on the chemical composition and biological activity of Mentha have been focused on the essential oils. 14 -16 However, in recent years, an increasing number of beneficial properties have been associated with polyphenols. 17
Escherichia coli and Staphylococcus aureus are both commensals and many strains are pathogenic for humans and animals. Escherichia coli is the most prevalent inhabitant of the gastrointestinal tract and capable of growing over a wide range of temperature (15-48 °C) and pH (2.0-8.0). 18 Pathogenic strains of E. coli are often associated with urinary tract infections, diarrhea, and persistent intestinal inflammation. 19 About 20%-30% of individuals are persistent and 30% intermittent carriers of E. coli. 20 Staphylococcus aureus is 1 of the main causes of hospital-acquired and community-acquired infections. Staphylococcus aureus can cause bacteremia, pneumonia, and toxic shock syndrome. It also causes serious deep-seated infections, such as endocarditis and osteomyelitis. 21 The control of E. coli and S. aureus infections varies according to strain and clinical manifestations. Trimethoprim-sulfamethoxazole, quinolones/fluoroquinolones, and extended-spectrum cephalosporins are commonly used for the treatment of human E. coli infections. 19 β-Lactam antibiotics, lincosamides, and macrolides are frequently used therapeutics for S. aureus infections. 22 However, antimicrobial resistance in E. coli and S. aureus has been reported worldwide and increasing rates of resistance is a growing concern in both developed and developing countries. 23
The composition of widely cultivated and economically important Mentha spp. is well studied, with the emphasis on essential oils. However, little is known about cultivars and mints from regionally less studied areas. The present study aims to give information on the essential oil and polyphenolic composition of Mentha spp. collected in Estonia. This study will broaden the knowledge about geographical variations and cultivars grown in Estonia and thereby improve the overall understanding of the chemotaxonomy of Mentha. Also, the antimicrobial activity of Mentha essential oils and water extracts is evaluated, supporting the use of mints as a health-promoting component of the diet with regard to their chemical contents.
Results and Discussion
Quantitative and Qualitative Composition of Mentha spp. Essential Oils
The total content of essential oils isolated from Mentha spp. varied in the range of 0.1%-1.9% (Table 1; Supplemental Table S1). The lowest oil yield was obtained from the sample M. “Black beauty” (No. 8) and the highest from M. × piperita L. “Morrocan” (No. 30).
Total Essential Oil Content (%; w/w) and Major Components (%; ≥3.0%) Detected From the Mentha spp.

In total, 93 compounds were determined in the essential oils (Supplemental Table S1). The most abundant compounds were cis-piperitone oxide (max. 81.4%, No. 13), carvone (max. 72.2%, No. 30), linalool (max. 61.0%, No. 25), menthol (max. 59.1%, No. 6), and menthofuran (max. 52.0%, No. 19).
The essential oils of M. × piperita sample Nos. 1-3, 5, 20 contained menthone and menthol as the major compounds; these are characteristic of peppermint oil and have been reported extensively in previous studies. 24 -28 On the contrary, the essential oil of cultivar “Lime” (No. 26) contained limonene as the most abundant compound. This could be the reason for the cultivar name “Lime” as limonene has been shown to be the major compound of lime (Citrus aurantifolia Swingle) essential oil. 29,30 Higher contents of limonene have been reported also in the M. × piperita oils originating from Brazil. 16 Essential oils of cultivars “Barnes & Cream” (No. 27) and “Morrocan” (No. 30) were rich in carvone. Similar results for M. × piperita were reported by Orav et al 31 for Mentha spp. originating from Estonia. “Lavender mint” (No. 32) contained linalool and linalyl acetate as its most abundant components. These 2 compounds are characteristic of the essential oil of lavender (Lavendula angustifolia L.) 32 and could, therefore, be the basis of the cultivar name. High linalool and linalyl acetate contents were highlighted also for M. × piperita samples Nos. 4 and 31. These 2 compounds have been reported in high content also in M. × piperita samples from Morocco 33 and Senegal. 34 A similarly high content of linalool has been shown to be abundant in the essential oils of basil (Ocimum basilicum L.) 35 and thus could have initiated the cultivar name “Basil mint” of sample No. 31.
Mentha spicata oils No. 17 and 18 contained carvone as the major compound, similar to the essential oils from Algeria, 36 Morocco, 33 India, 37 and Tunisia. 38 On the contrary, in M. spicata oil No. 7, menthofuran was identified as the major compound. Also, sample No. 7 had a high content of an unknown compound with fragment ions m/z 79, 93, 107, and 121. The same unidentified compound was most abundant in the sample M. “Black beauty” (No. 8).
The main constituent of M. longifolia sample Nos. 11 and 16 was cis-piperitone oxide, as shown by Kokkini and Papageorgiou in the oil from Greece. 39 In addition, high piperitone oxide content has been detected also in M. longifolia oils from Brazil 16 and Bosnia and Herzegovina. 40 In contrast, M. longifolia essential oil No. 24 contained in our study α-terpinyl acetate as its most abundant component.
Mentha × gracilis Sole oil No. 33 was rich in carvone. This compound was also found as the major compound in M. × gracilis by Zheljaskov et al. 41 On the other hand, linalool was the major constituent in the M. × gracilis oils Nos. 12, 15, and 25.
The oils of M. arvensis were diverse in composition. Verma et al 42 and de Sousa Guedes 43 reported the oil to contain menthol in the highest concentration in the essential oils originating, respectively, from India and Brazil. Also, the oil from Brazil has been reported to be rich in linalyl acetate. 16 However, in the present study, M. arvensis oils were found to contain 3-octanone (No. 21) and 1,8-cineole (No. 22) as the main components.
Mentha × villosa oil (No. 10) contained carvone as the major compound, contrary to the piperitenone oxide reported by Teles et al 44 and Alves et al 45 in the oil from Brazil. M. aquatica oil (No. 19) was rich in menthofuran, as reported by Mimica-Dukić et al 14 and Boz et al 46 in the essential oils, respectively, from Serbia and Romania. The oil of M. suaveolens (No. 14) had a high content of piperitenone oxide. The same results were found earlier by Civitelli et al. 47
The results of the essential oil composition analyses reflect the diversity among the Mentha spp. Thus, further and wide-scale studies would be needed, especially focusing on cultivars and less cultivated Mentha spp.
Quantitative and Qualitative Composition of Mentha spp. Polyphenols
The content of polyphenols identified in the water extracts of the Mentha spp. varied, but the composition profile was rather similar (Tables 2 and 3). In total, 32 compounds were identified. Only 3 polyphenols were present in all the Mentha spp. extracts, namely danshensu, medioresinol, and rosmarinic acid. Of the detected compounds, the most seldom detected were narirutin and apigenin-O-glucuronide. Sample high-performance liquid chromatography (HPLC)-ultraviolet (UV)-MS/MS chromatograms are presented in Supplemental Table S2.
Liquid Chromatography-Mass Spectrometry (MS)/MS Characteristics of the Compounds Identified in the Mentha spp.
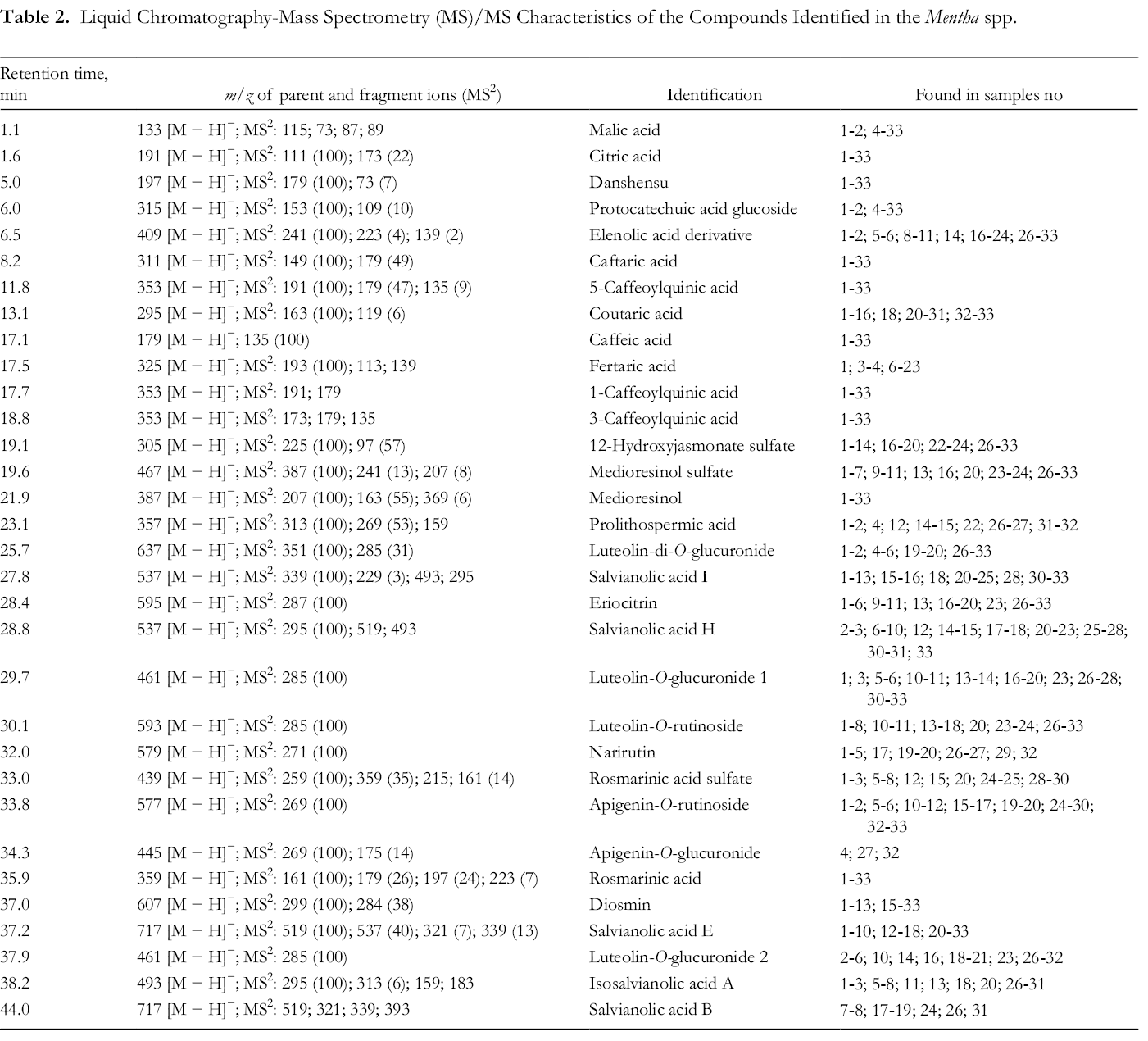
Quantitative Composition (%) of the Polyphenolic Compounds in the Water Extracts of Mentha spp.

Abbreviations: AG, apigenin-O-glucuronide; AR, apigenin-O-rutinoside; D, diosmin; DA, danshensu; ER, eriocitrin; ISAA, isosalvianolic acid A; LDG, luteolin198 di-O-glucuronide; LG1, luteolin-O-glucuronide 1; LG2, luteolin-O-glucuronide 2; LR, luteolin-O-rutinoside; M, medioresinol; MS, medioresinol sulfate; N, narirutin; nd, not detected; PA, prolithospermic acid; RA, rosmarinic acid; RAS, rosmarinic acid sulfate; SAB, salvianolic acid B; SAE, salvianolic acid E; SAH, salvianolic acid H; SAI, salvianolic acid I; 12-HJS, 12-hydroxyjasmonate sulfate; *, oxylipin.
The most abundant phenolic compound in the extracts was rosmarinic acid, found in the range of 4.3%-52.7%. The lowest content was found in M. × piperita cv. “Grenada” extract (No. 4) and highest in M. spicata “Marrocan” (No. 17). As the major compound, it was found in Algeria grown M. spicata, M. pulegium, and M. rotundifolia by Fatiha et al. 48 A distinctly high rosmarinic acid content has also been reported in M. spicata methanolic extracts originating from the USA. 49 In addition to these Mentha spp., rosmarinic acid has also been detected in M. × piperita, 50 M. australis R. Br., 51 M. canadensis L., 52 and M. haplocalyx Briq., 53 respectively, collected from Finland, Australia, USA, and China.
The second most abundant compound was salvianolic acid B, found in the highest content of 15.2% in the extract of M. spicata “Lime” (No. 7). The lowest content, 1.5% salvianolic acid B, was found in the extract of M. aquatica (No. 19). It is noteworthy that from the studied 33 plant extracts, salvianolic acid B was only detected in 8, with a high content (13.3%) highlighted also for M. “Black Beauty” (No. 8).
The third major compound was eriocitrin with the highest content of 13.6% in the extract of M. × piperita “Morrocan” (No. 30), and the lowest (0.6%) in the extract of M. × piperita “Black Beauty” (No. 29).
In high content also were diosmin, 12.3% in the extract of M. × gracilis “Ginger” (No. 15), and salvianolic acid E, 11.2% in the extract of M. × piperita (No. 20). Both, salvianolic acid B and salvianolic acid E have been earlier reported in M. × piperita, 11 M. spicata, 49 and M. pulegium. 54 Other more abundant polyphenolic compounds were salvianolic acid H, danshensu, medioresinol sulfate, and medioresinol. The water extracts also contained oxylipin 12-hydroxyjasmonate sulfate in high content. The highest content of 12-hydroxyjasmonate sulfate (11.9%) was found in M. × piperita L. (No. 20). Organic acids were detected in minor quantities. Acids present were malic and citric acid.
In general, M. × piperita extracts were richer in unidentified polyphenolic compounds than other Mentha spp. analyzed. Mentha arvensis extracts are highlighted due to the lowest content of identified polyphenolic compounds.
Based on the analyses of the polyphenols in the extracts, no species-specific compounds could be proposed. Although the water extracts of certain Mentha spp. indicated a trend based on the content of individual compounds, the use of the polyphenolic composition for species identification would be doubtful.
Preliminary Antibacterial Susceptibility Testing
Preliminary antibacterial susceptibility testing was carried out using a commercial M. × piperita essential oil and water extracts of sample Nos. 4, 18, 19, and 22. Gram-negative bacteria were represented by Y. ruckeri, and Gram-positive bacteria by Bacillus cereus, B. subtilis, B. pumilus, and Micrococcus luteus. These bacteria are commonly used for testing antibacterial susceptibility. Commercial oil, similar in composition to the Mentha spp. oils, was used due to the limited sample amounts of Mentha spp. oils. The tested water extracts were chosen as they presented various Mentha spp. and cultivars. The strongest antibacterial effect was observed for the essential oil (Supplemental Table S3). The oil was bioactive at both tested concentrations against all the Gram-positive bacteria, as well as against the Gram-negative Y. ruckeri. Of the water extracts, M. × piperita cv. “Grenada” (No. 4) extract showed activity at a concentration of 100.0 mg/mL toward all the tested bacteria, besides M. luteus. The extract of M. aquatica (No. 19) inhibited the growth of Y. ruckeri at a concentration of 100.0 mg/mL. Extracts of M. spicata “Kentucky Colonel” (No. 18) and M. arvensis (No. 20) showed no activity. The preliminary experiments encouraged further studies with potentially pathogenic E. coli and S. aureus.
Antibacterial Activity of Mentha spp. Essential Oils
The antibacterial activity of the essential oils of 9 Mentha spp. was evaluated at concentrations of 1.0‐4.0 mg/mL (Table 4). The 9 selected oils were diverse in composition and represented various Mentha spp. and cultivars.
MIC90 ± SD (mg/mL) of Mentha spp. Essential Oils (n = 2‐3) and Water Extracts (n = 2).

Abbreviations: MIC90, 90% minimum inhibitory concentration; NA, not active.
aMinimal bactericidal concentration.
Five essential oils had antibacterial activity against E. coli. The 90% minimum inhibitory concentration (MIC90) values for the active samples Nos. 10, 13, 16, and 18 were 3.0 mg/mL and for No. 6, 4.0 mg/mL. The MIC90 value of M. × villosa oil had also a minimal bactericidal concentration (MBC) against E. coli (Table 4).
The oils obtained from M. × villosa (No. 10) and M. “Golden mint” (No. 13) had a high content of carvone. Also, the active M. spicata oil (No. 18) was rich in carvone. This compound was found to possess antimicrobial activity against E. coli by Kapp et al. 55 Similar antibacterial activity has been reported by Hussain et al 15 and Naigre et al. 56 Thus, the antibacterial activity of the oils Nos. 10, 13, and 18 could be related to the content of carvone.
The M. × piperita No. 6 oil contained menthol as the most abundant compound. Menthol was found to be inactive against E. coli. 55 Thus, the higher MIC90 value of oil No. 6 can be due to the high menthol content.
The oil of M. × villosa possessed activity, as shown in the study by Arruda et al. 57 Surprisingly, the oil of M. aquatica (No. 19) was found to be inactive, although it contained a high concentration of menthofuran that in other studies was found to inhibit the growth of E. coli. 55 Furthermore, M. aquatica essential oil, which is rich in pulegone and menthofuran, has been shown to be antimicrobial on E. coli by Dhifi et al 58 and Mimica-Dukić et al. 59 Here it should be noted that, besides the major essential oil components, minor compounds may have a synergistic role and have an important role in the antibacterial effects of Mentha spp.
Most of the essential oils showed ≥90% inhibition against S. aureus at a concentration of 1.0 mg/mL. The oil of M. × piperita “Lavender mint” (No. 32) was active at a concentration of 2.0 mg/mL.
The tested essential oil Nos. 2, 6, and 20 contained menthol and menthone as their most abundant compounds, and both were found to inhibit the growth of S. aureus. 55 The antibacterial activity of oils No. 10 and No. 13 could be due to their high content of carvone, which has been found to possess activity against S. aureus. 55,60 Mentha No. 32 contained linalool and linalyl acetate as its major compounds. These 2 compounds have been found to be active against S. aureus. 55 However, the higher MIC90 value of the essential oil of plant No. 32 can be noted.
Antibacterial Activity of Water Extracts of Mentha spp.
Eight water extracts of Mentha spp. were tested for their antibacterial activity against E. coli and S. aureus at a concentration of 1.0‐4.0 mg/mL (Table 4). The water extracts selected represented various Mentha spp. and cultivars. Also, their composition was rather diverse. Furthermore, the antibacterial effect of M. × villosa, M. × suaveolens, and M. × gracilis water extracts was studied for the first time against E. coli and S. aureus.
None of the extracts had antibacterial activity against E. coli. Similar antibacterial activity results have been reported for alcoholic extracts of M. longifolia, 61 M. spicata, 62 M. × piperita, and water extracts of M. × piperita. 63 The most dominant compound in the water extracts of Mentha spp. was rosmarinic acid, which has been shown to lack antibacterial activity against E. coli. 64 Eriodictyol and luteolin, the 2 other compounds in higher content, have been found to exhibit an antibacterial effect against E. coli. 65 Nevertheless, it can be assumed that the total content of eriodictyol and luteolin glycosides in the Mentha spp. extracts was too low to inhibit the growth of E. coli in the present study.
Six of the water extracts inhibited S. aureus. An MIC90 value of 2.0 mg/mL was possessed by extracts of M. × piperita “Chocolate” (No. 2), M. suaveolens “Apfel” (No. 14), and M. × gracilis “Ginger ” (No. 15). The water extract of M. × piperita sample No. 2 was also found to be bactericidal against S. aureus at a concentration of 2.0 mg/mL. Also Sofia et al 66 have reported antimicrobial activity of M. × piperita water extract against S. aureus. M. × villosa (No. 10), M. spicata “Kentucky Colonel” (No. 18), and M. longifolia (No. 24) had MIC90 values of 3.0 mg/mL, whereas M. aquatica (No. 19) and M. arvensis (No. 22) extracts showed no inhibition.
Extract No. 22 had the lowest content of identified compounds. Eriodictyol, 65 luteolin, and apigenin, 67,68 have been found to possess antibacterial activity against S. aureus strains. Thus, the inactivity of the water extract of M. arvensis L. No. 22 could be due to the low content of luteolin derivatives and the absence of eriodictyol and apigenin glycosides. Extract No. 22 also had a low content of danshensu that has been reported to possess antimicrobial activity against S. aureus. 69 This could also explain the inactivity of M. aquatica (No. 19) extract, which had the lowest content of danshensu of the studied Mentha spp. plants.
Conclusions
Mentha spp. are common herbs used for their medicinal and aromatherapeutic properties since written history. Mentha spp. show a large phenotypic plasticity and most species are capable of hybridization with each other. The problematic taxonomy was faced in the present study. Some of the Mentha spp. samples turned out to be difficult to classify or their exact identity remained open. Also, the composition of many mints was different or unique compared with previous reports. Thus, the study raised the need for more extensive studies, especially focusing on cultivars and less cultivated Mentha spp.
Escherichia coli and S. aureus are among the most prevalent potentially pathogenic bacteria with increasing rates of antimicrobial resistance. Mentha spp. essential oils and water extracts showed antibacterial activity against either E. coli or S. aureus or both. Some of the samples were found to be bactericidal. Thus, the use of mint products in the diet may prevent diseases caused by E. coli and S. aureus.
Materials and Methods
Chemicals and Instruments
LiChrosolv n-hexane and SeccoSolv dimethyl sulfoxide (DMSO) were from Merck Millipore, Darmstadt, Germany; methanol (MeOH), diosmin, jasmonic acid, and salvianolic acid B from Sigma-Aldrich (Steinheim, Germany); luteolin, apigenin, narirutin, eriodictyol-7-O-gluronide, and rosmarinic acid from Extrasynthese (Genay, France); and acetonitrile (ACN) for liquid chromatography-mass spectrometry (LC-MS) of ultragradient grade from Romil (Cambridge, UK). Water used was prepared by an EASYpure RF compact system (Barnstead, USA). Cuplaton antifoam agent was produced by Orion Pharma, Espoo, Finland. Mueller Hinton II agar (MHA) and Mueller Hinton II broth (MHB) were obtained from Becton Dickinson, Franklin Lakes, NJ, USA. Ciprofloxacin hydrochloride was purchased from ICN Biomedicals Inc., Ohio, USA; Nunclon Delta Surface 96-well microplates from Thermo Fisher Scientific, Roskilde, Denmark; and Petri dishes from Heger Plastics, Rjukan, Norway.
Plant Material
The leaves of either cultivated or wild-grown Mentha spp. (n = 33) were collected during full bloom in August 2012 from different areas in Estonia (Table 5). The plant material was dried at room temperature (20 ± 2 °C), protected from direct sunlight. Each sample was labeled, packed in a paper bag, and stored at room temperature until assayed. Voucher specimens were prepared and identified by Intendant Arto Kurtto, Finnish Museum of Natural History, University of Helsinki. The voucher specimens (Lamiaceae/Mentha No.) have been deposited at the Faculty of Pharmacy, University of Helsinki, Finland. Voucher specimens were not prepared from Mentha spp. collected from Tartu University Botanical Garden (Nos. 24-32) or when the plants did not survive the winter following the plant material collection (Nos. 6, 8, 9, 13-15, 23). Samples without voucher specimens are marked with an asterisk in Table 5. Mentha spp. without original taxonomic data and identification by voucher specimen are given with the genus and cultivar name.
Mentha spp. Studied.

aNo voucher specimen.
bWild-grown plant.
Isolation of Essential Oils
The essential oils of Mentha spp. were isolated using a Marcusson-type microdistillation apparatus 70 with 300 µL n-hexane as a trap. The sample (0.85-5.0 g) with 100 mL of distilled water was subjected to distillation for 2 hours. The essential oil amount was determined gravimetrically after evaporating the n-hexane in a nitrogen flow. All the essential oils were stored in sealed vials under refrigeration (−20 °C) prior to analysis.
Extraction of Polyphenols
Water extracts were prepared from the plant material previously subjected to distillation. Two-hour decoctions of the Mentha spp. were filtered, concentrated, and the residues from a rotary evaporator frozen at −20 °C and thereafter lyophilized in a Heto LyoPro 3000, Allroed, Denmark) for 7 days. Water extracts were prepared by dissolving 10.0 mg of lyophilized extract in 1.0 ml of water.
Gas Chromatography-MS Analyses
The essential oils were analyzed using an Agilent 7890A combined with an Agilent 5975C TAD Mass Selective Detector (gas chromatography [GC]/MSD) with a Triple-Axis High Energy Diode (HED) Electron Multiplier (EM) detector on a DB-5 capillary column [(5%-phenyl) methylpolysiloxane, 30 m × 0.25 mm, film thickness 0.25 μm] (Agilent Technologies, Palo Alto, CA, USA) (n = 1). The oven temperature was programmed from 50 °C to 240 °C at 2 °C/min with the injector temperature of 300 °C. The carrier gas was helium, with a split ratio of 1:30 and a flow rate of 1.3 mL/min. The identification of the oil components was accomplished from spectra using commercial spectral libraries NIST 11, Scientific Instrument Services Inc., and FFNS 2 Wiley Library. Identification of compounds was confirmed by retention indices of reference standards and library data. The composition of the oils was calculated as the percentage from peak areas using a normalization method without correction factors. The relative SD of percentages of oil components in 3 repeated GC analyses of a single oil sample did not exceed 5%.
HPLC-UV-MS/MS Analyses
For the identification and quantitation of polyphenols in the water extracts, an HPLC combined with UV-vis diode array and ion trap mass spectrometric detection (LC-DAD-ESI-MS/MS) method was used (n = 1). A 1100 Series LC/MSD Trap-XCT equipped with an electrospray interface (ESI) (Agilent Technologies, Palo Alto, CA, USA) working in negative ionization was used. The conditions of the MS2 detection were m/z interval, 50-1000 amu; target mass, 400 amu; number of fragmented ions, 2; maximal accumulation time, 100 ms; compound stability, 100%; drying gas, nitrogen from generator; and collision gas, helium. The ion trap was connected to the HPLC instrument consisting of an autosampler, solvent membrane degasser, binary pump, column thermostat, and UV-vis diode array detector. HPLC 2D ChemStation software with a ChemStation Spectral SW module was used for the process guidance as well as processing the results. The compounds were separated on a reversed-phase column Zorbax 300SB-C18 (150 × 2.1 mm i.d.; 5 μm particle size; Agilent Technologies, Palo Alto, CA, USA) in the following gradient of 0.1% formic acid (solvent A) in water (v/v) and acetonitrile (solvent B): 0-5 minutes 1% B, 5-60 minutes a linear gradient of B 1%-35%, and 60-70 minutes 35%-95% B. The column temperature was 35 °C, eluent rate 0.3 mL/min, and injection volume 5 μL.
Identification of the compounds was made as described previously in Kapp et al. 11 Stock solutions (1.0 mg/mL) of reference standards were prepared by dissolving individual compounds in MeOH. Diosmin was dissolved in DMSO. Working solutions of all the standards were obtained by diluting the stock solutions with methanol. Phenolic compounds were identified by comparing the retention times, order of elution, UV spectra, and MS/MS fragmentation spectra either with reference standards or with literature data. The identification of caffeoylquinic acids was made according to the results shown by Liao et al 71 and Gouveia et al. 72 Salvianolic acid I and H were determined according to Taamalli et al. 54 A compound characterized by m/z 409 was detected as a derivative of elenolic acid according to Leouifoudi et al. 73 Rosmarinic acid sulfate was determined according to Miron et al. 74
Quantification of all polyphenols was achieved using a calibration curve of MS-base peak areas of rosmarinic acid as a reference. For that, rosmarinic acid was dissolved in MeOH to 8 different concentrations (0.003-0.3 mg/mL). The mg per g of herb was converted into percentage concentration by summarizing all the polyphenols and considering the latter sum as a total.
Antimicrobial Susceptibility Testing
Preliminary Antibacterial Susceptibility Testing by Agar Diffusion Method
Preliminary antibacterial susceptibility testing was performed using a modified agar well-diffusion method described by Pikkemaat et al 75 and Raudsepp et al. 76 Bacterial strains were obtained from the reference strains collections of the Estonian Veterinary and Food Laboratory. Gram-negative bacteria were represented by Yersinia ruckeri (NCIM 13282). Gram-positive bacteria were B. cereus (ATCC 11778), B. subtilis (BGA), B. pumilus (CN 607), and M. luteus (ATCC 9341). Cultures from the solid medium were subcultivated into liquid media. Incubated bacterial suspension was mixed with sterilized Iso-Sensitest Agar (B. cereus, M. luteus), plate-count agar (B. subtilis, Y. ruckeri), or DST-agar (B. pumilus) to obtain a final density of 106 colony-forming units (CFU)/mL and then poured into Petri dishes for solidification at room temperature. Wells were made into agar gel (5 mm in diameter) and filled with 30 µL of sample dissolved in either n-hexane or water. After 24 hours of incubation at either 30 °C for B. cereus and Y. ruckeri or at 37 °C for M. luteus, B. subtilis, and B. pumilus, the radius of the inhibition zone was measured. Commercial peppermint oil (Oleum mentha × piperita L., Solnetšnogorsk, Russian Federation) and n-hexane as a negative control were used for the testing. Water extracts (Nos. 4, 18, 19, 22) were dissolved in water.
Determination of MIC90 by Broth Dilution Method
Antimicrobial assays were performed following the guidelines of the Clinical and Laboratory Standards Institute (CLSI) and European Committee on Antimicrobial Susceptibility Testing (EUCAST)
Clinical control strains of S. aureus (Gram-positive, ATCC 25923) and E. coli (Gram-negative, ATCC 25922) were obtained from Microbiologics Inc. (St. Cloud, MN, USA) and used for the antimicrobial screening. Bacterial strains were grown on MHA. Media were prepared in MilliQ water, according to the manufacturer’s instructions, and autoclaved at 121 °C for 15 minutes. Prior to the assay, bacterial suspensions were prepared in MHB from fresh slant cultures and incubated at 37 °C for 16-20 hours at 100 rpm.
For the assays, bacterial suspensions were diluted with MHB to obtain a final inoculum of 5 × 105 CFU/mL in the assay (determined on the basis of absorbance values at 620 nm previously calibrated against plate counts). Assays were carried out by the tube dilution method combined with absorbance measurement at 620 nm in 96-well microtiter plates.
MHB (245 µL) and 250 µL of bacterial suspension were added to the tube, followed by test samples dissolved in 5 µL of DMSO (Mentha essential oils) or water (Mentha water extracts). Ciprofloxacin at a concentration of 0.5 µg/mL was used as a positive control. The tubes were incubated at 37 °C for 24 hours at 100 rpm and observed for turbidity at 4, 8, and 24 hours. Absorbance at the 24-hour time-point was measured at 620 nm with a Multiskan GO microplate spectrophotometer (Thermo Fisher Scientific, Vantaa, Finland) by transferring the incubated samples to 96-well microplates. The antibacterial activity of the samples was calculated from the absorbance values by comparing with untreated controls and expressed as the percentage inhibition of growth. The MIC90 was defined as the lowest concentration that showed ≥90% inhibition of growth.
Determination of Minimal Bactericidal Concentration (MBC)
Samples with ≥90% inhibition of growth were further tested for minimal bactericidal concentration (MBC). Samples of 50 µL from the MIC assay were plated on fresh MHA plates and incubated for 24 hours at 37 °C. Concentration at which 99.9% of the initial bacterial inoculum had been killed was considered as MBC.
Supplemental Material
Table S1 - Supplemental material for Chemical Composition and Antibacterial Effect of Mentha spp. Grown in Estonia
Supplemental material, Table S1, for Chemical Composition and Antibacterial Effect of Mentha spp. Grown in Estonia by Karmen Kapp, Tõnu Püssa, Anne Orav, Mati Roasto, Ain Raal, Pia Vuorela, Heikki Vuorela and Päivi Tammela in Natural Product Communications
Supplemental Material
Table S2 - Supplemental material for Chemical Composition and Antibacterial Effect of Mentha spp. Grown in Estonia
Supplemental material, Table S2, for Chemical Composition and Antibacterial Effect of Mentha spp. Grown in Estonia by Karmen Kapp, Tõnu Püssa, Anne Orav, Mati Roasto, Ain Raal, Pia Vuorela, Heikki Vuorela and Päivi Tammela in Natural Product Communications
Supplemental Material
Table S3 - Supplemental material for Chemical Composition and Antibacterial Effect of Mentha spp. Grown in Estonia
Supplemental material, Table S3, for Chemical Composition and Antibacterial Effect of Mentha spp. Grown in Estonia by Karmen Kapp, Tõnu Püssa, Anne Orav, Mati Roasto, Ain Raal, Pia Vuorela, Heikki Vuorela and Päivi Tammela in Natural Product Communications
Footnotes
Acknowledgments
The authors thank Intendant Arto Kurtto from the Finnish Museum of Natural History, University of Helsinki for identification of the Mentha spp. plants and Dr Ülle Reier from the Department of Botany, University of Tartu for consultation. Estonian University of Life Sciences, Chair of Food Hygiene and Veterinary Public Health, and Tallinn University of Technology, Institute of Chemistry are thanked for providing access and equipment for chromatographic analyses. Estonian University of Life Sciences, Chair of Food Hygiene, and Veterinary Public Health and Estonian Veterinary and Food Laboratory are thanked for the support in preliminary antimicrobial susceptibility testing.
Authors’ Note
Pia Vuorela: Deceased 1.10.2017.
In Honor of Professor Dr K. Hüsnü Can Başer on his 70th birthday and in recognition of his outstanding contribution to natural product research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by The Academy of Finland, grant number 277001 and 272266. Karmen Kapp acknowledges FinPharma Doctoral Programme (FPDP) for financial support.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
