Abstract
Using various column chromatographic methods, 5 triterpene tetraglycosides (
Introduction
Within marine organisms, echinoderms are a source of a broad range of secondary metabolites having various biological activities. Of these metabolites, saponins represent the most abundant and diverse products in the phylum Echinodermata. Among these marine invertebrates, saponins are the most known and structurally diverse within sea cucumbers. The majority of them are holostane derivatives possessing an 18(20)-lanostane lactone as aglycon and a carbohydrate chain consisting of from 1 to 6 monosaccharide units. These compounds demonstrate interesting biological effects such as cytotoxic, ichthyotoxic, antifungal, and hemolytic activities, as well as a series of additional effects at subtoxic doses, including immunomodulatory and cancer preventive.1,2
As a part of our ongoing investigations of saponins from Vietnamese sea cucumbers,3–6 this paper deals with the isolation and structural elucidation of 5 triterpene tetraglycosides (
Results and Discussion
Using various column chromatographic separations, 5 triterpene tetraglycosides (
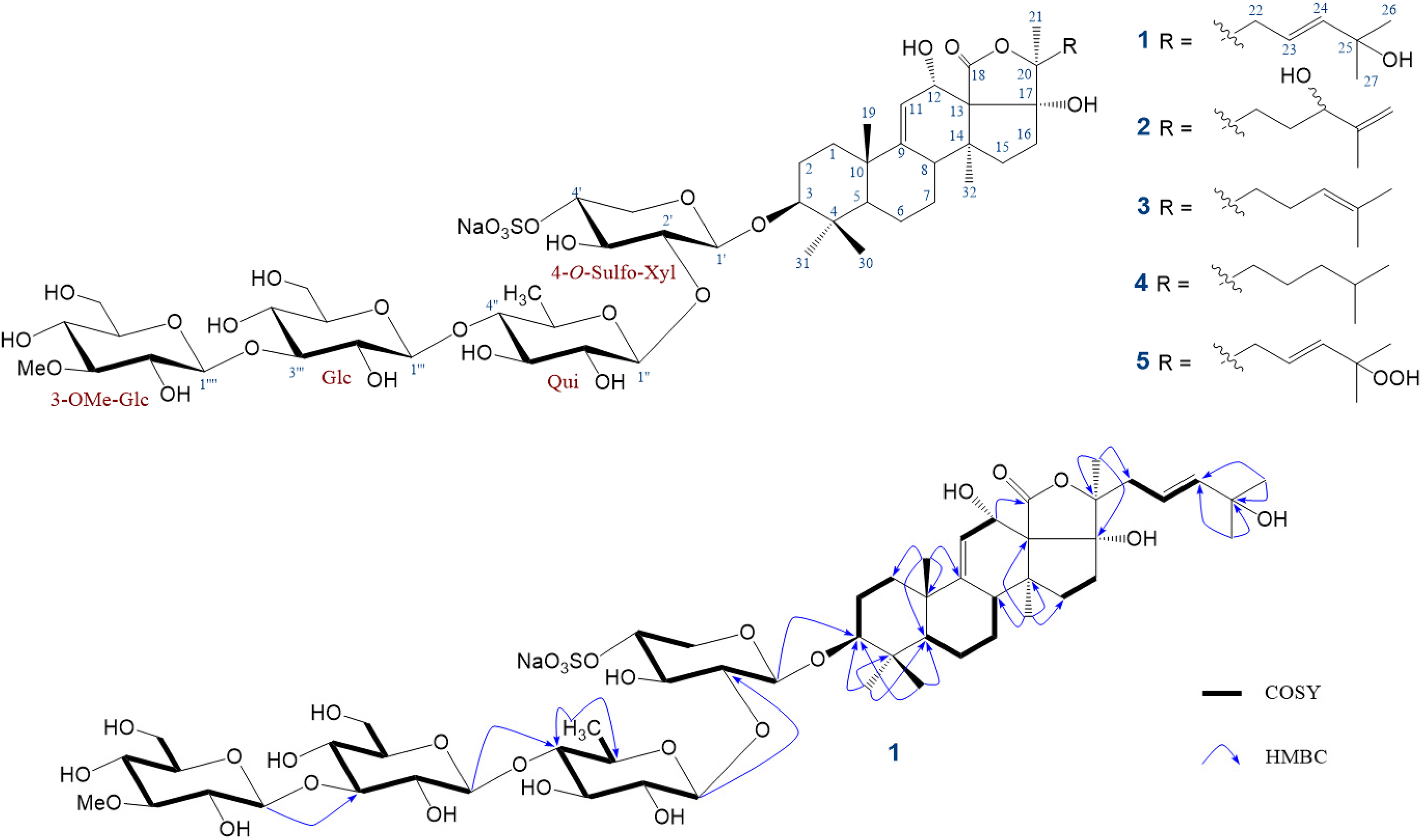
The structures of
Holothurin A6 (
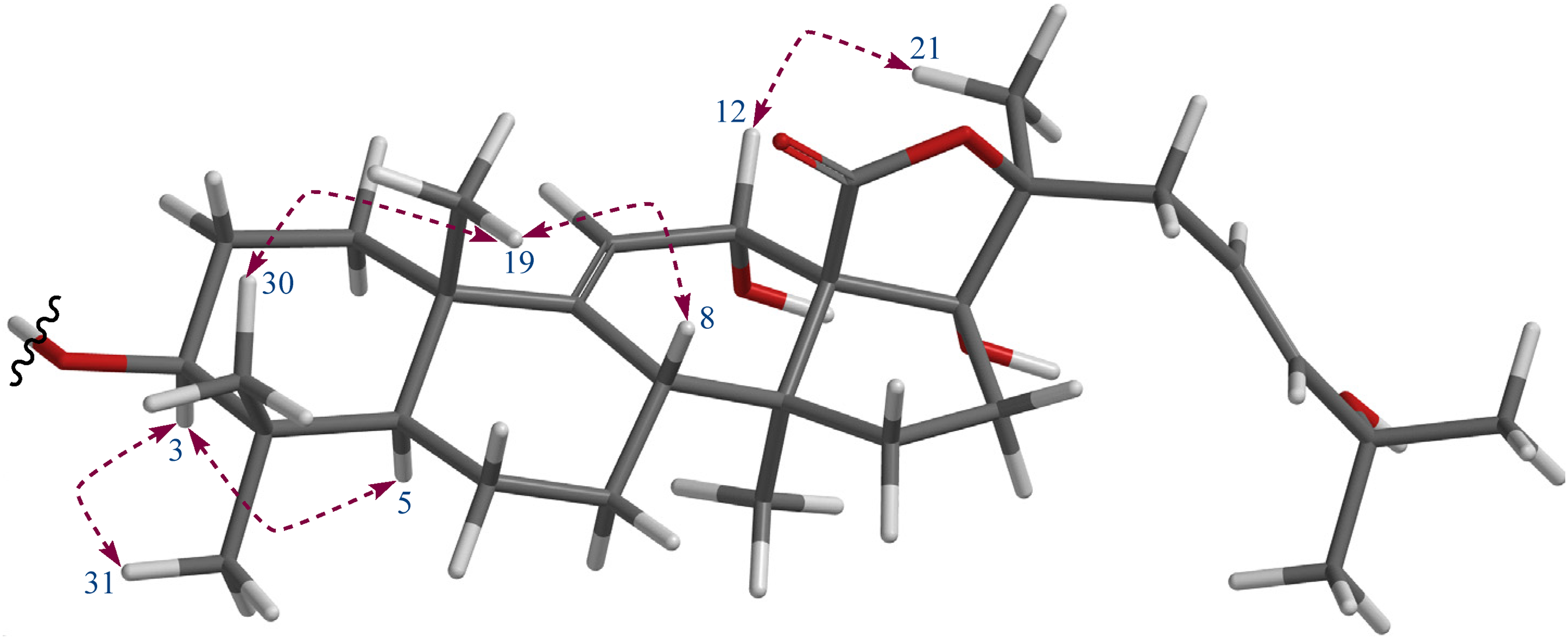
Key ROESY correlations of
1H and 13C NMR Spectroscopic Data of

Overlapped signals. All data were confirmed by HSQC, COSY, HMBC, ROESY, and 1D and 2D TOCSY spectra.
The cytotoxic activity of compounds
The Cytotoxicity of

Positive control. Data are the means ± SD of triplicate experiments.
Experimental
General
Refer to Supplemental Material.
Marine Organism Material
S herrmanni Semper, 1868 samples were collected at Bai Giau island (coordinates: 12°40′44′′−109°20′33′′) and Khai Luong island (coordinates: 12°35′06′′−109°24′44′′) in Van Phong, Khanhhoa, Vietnam, during July 2020. Its scientific name was identified by one of our authors, Prof Do Cong Thung. Voucher specimens (No. DLB-2020-DG06) were deposited at the IMBC and the IMER, VAST.
Extraction and Isolation
Refer to Supplemental Material.
Holothurin A6 (1 )
White amorphous powder.
[α]D25 −21 (c 0.1, MeOH);
1H (pyridine-d5, 600 MHz) and 13C NMR (pyridine-d5, 150 MHz) data given in Table 1;
HR ESI QTOF MS m/z 1243.4767 [M + Na]+ (calcd. for C54H85Na2O27S+, 1243.4783).
Cytotoxic Assays
Refer to Supplemental Material.
Conclusions
Five triterpene tetraglycosides, with one new compound holothurin A6 (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221105369 - Supplemental material for Triterpene Tetraglycosides From Stichopus Herrmanni Semper, 1868
Supplemental material, sj-docx-1-npx-10.1177_1934578X221105369 for Triterpene Tetraglycosides From Stichopus Herrmanni Semper, 1868 by Le Thi Vien, Tran Thi Hong Hanh, Tran Hong Quang and Nguyen Van Thanh, Do Thi Thao, Nguyen Xuan Cuong, Nguyen Hoai Nam, Do Cong Thung, Phan Van Kiem in Natural Product Communications
Footnotes
Acknowledgments
This study was supported by Vietnam Academy of Science and Technology (grant number TĐDLB0.02/20-22). The authors are thankful to the Institute of Chemistry, VAST for the NMR spectral measurements and Dr Bui Huu Tai, Institute of Marine Biochemistry, VAST for the HR-QTOF mass spectrum measurement.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vietnam Academy of Science and Technology, (grant number TĐDLB0.02/20-22).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
