Abstract
A new benzofurane 7-methoxy-2-(3-hydroxy-4-methoxyphenyl)-benzofuran-5-carboxylate (
Helicteres genus is a large genus of tropical trees and shrubs (family Sterculiaceae) with axillary flowers and fruits consisting of 5 twisted carpels.
1
Helicteres genus consisting of about 60 species distributed in tropical America and Asia. Many of them are used as folk medicines for diabetes, intestinal infection, stomach affection, empyema, uterus pain, etc.
2,3
In Vietnam, there are 9 Helicteres species, among them 6 species being used as traditional medicines.
4,5
The primary chemical constituents of this genus were triterpenoids,
6,7
cucurbitans,
8,9
lignans,
10
flavonoids,
11,12
and coumarins,
13
which possessed cytotoxic, antinociceptive, analgesic, anti-inflammatory, and antibacterial effects and hypoglycemic activities. Helicteres hirsuta is a small tree that is widely distributed in Asia such as Thailand, Indonesia, Cambodia, and Vietnam. Six lignans from the stems of Indonesian H. hirsuta, including (±)-pinoresinol, exposed potent cytotoxic effects against a small panel of cancer cell lines.
10
Twelve known compounds from H. hirsuta. including 5 lupane triterpenoids exhibited weak cytotoxic activity.
14
This paper describes the isolation and structural elucidation of a new benzofuran derivative 7-methoxy-2-(3-hydroxy-4-methoxyphenyl)-benzofuran-5-carboxylate (

Structures of compounds 1 to
Compound
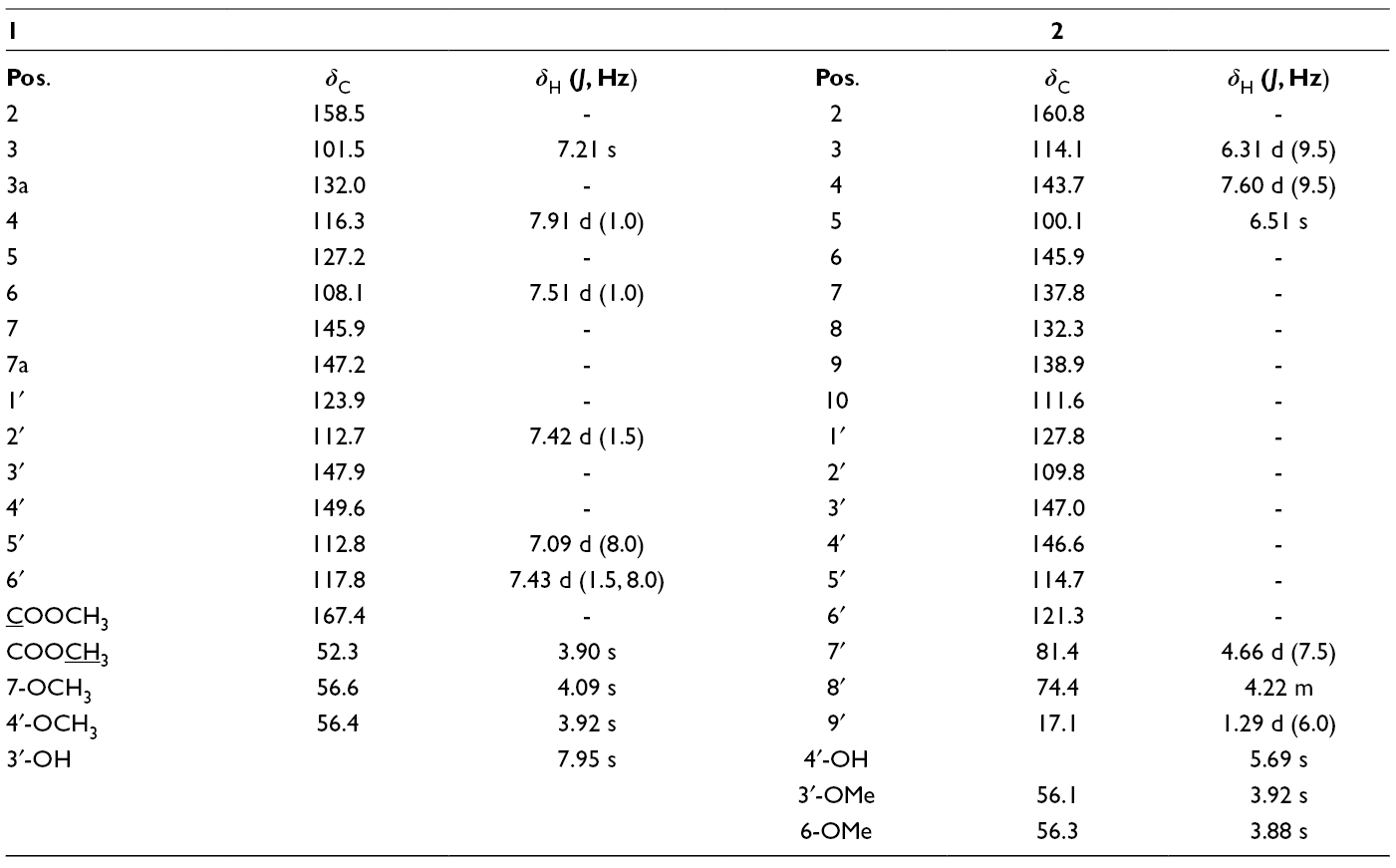
Additionally, HMBC correlations from protons H-4 (δ
H 7.91)/H-6 (δ
H 7.51) to carbonyl carbon (δ
C 167.4) indicated that the carboxylate group joined to the benzofuran ring at C-5. Similarly, HMBC correlations from H-3 (δ
H 7.21)/H-2′ (δ
H 7.42)/H-6′ (δ
H 7.43) to C-2 (δ
C 158.5) and C-1′ (δ
C 123.9), as well as from H-5′ (δ
H 7.09) to C-1′ (δ
C 123.9), supported the linkage between the benzofuran ring and the benzene ring at C-2 (δ
C 158.5). Finally, the HMBC correlations from protons H-2′ (δ
H 7.42) and hydroxyl proton (δ
H 7.95) to carbons C-3′ (δ
C 147.9) and C-4′ (δ
C 149.9), and from the methoxyl proton (δ
H 4.09) to C-4′ (δ
C 149.6) determined the position of the hydroxyl group at C-3′ and methoxyl group at C-4′, which was further confirmed by NOESY correlation from H-5′ (δ
H 7.09) to methoxyl proton (δ
H 3.92) (Figure 2). From the above evidence, compound

Key HMBC and NOESY correlations of compounds 1 and 2.
The 1H-NMR data of
NMR Data of Compounds 1 and 2 (1H: 500 MHz and 13C: 125 MHz).
The known compounds were identified as hisbiscolatone A (
Compounds
Inhibition Capacity of 1 to

The other remaining compounds had inhibition less than 50% at 128 µg/mL on the cell lines. As the results shown in Table 3, compound
Cytotoxicity of Compounds 1 to

Experimental
General Procedure
Melting points were measured in melting point meter Buchi B545. HR-ESI-MS were measured on a FT-ICR 910 MS TQFTMS-7T mass spectrometer. NMR spectra were recorded on a Bruker 500.13 MHz spectrometer, operating at 125.76 MHz for 13C NMR and at 500.13 MHz for 1H-NMR. 1H chemical shifts were referenced to CDCl3 and CD3OD at δ 7.27 and 3.31 ppm, respectively, while the 13C chemical shifts were referenced to the central peak at δ 77.0 (CDCl3) and 49.0 (CD3OD). For HMBC experiments, the delay (1/2J) was 70 ms, and for the NOESY experiments, the mixing time was 150 ms.
Plant Material
H elicteres hirsuta L. was collected in 2018 at Thua Thien-Hue, Vietnam and identified by Dr Tran The Bach. A specimen (Tra292018) was deposited at Institute of Ecology and Natural Resources, Vietnam Academy of Science and Technology.
Extraction and Isolation
Dried stems of H. hirsuta L. (3.2 kg) were extracted in alcohol at room temperature (1.0 L × 5) overnight. After evaporating under reduce pressure, the crude extract was obtained (106 g). This extract was dissolved in water (1.0 L) and successively extracted in hexane and EtOAc. The hexane, EtOAc, and water solutions were concentrated under diminished pressure to afford 20 g, 32 g, and 50 g dry extracts, respectively. The EtOAc extract (32 g) was subjected to a silica gel column chromatography (CC) with a gradient solvent of hexane/acetone to furnish 9 fractions. Fraction 1 was separated on a silica gel CC (gradient hexane/acetone) to yield 3 subfractions (1-3). Subfraction 2 was purified by preparative TLC (hexane/acetone: 85/15) and then purified on Sephadex LH-20 column (MeOH/CH2Cl2: 9/1, v/v) to give compound
7-Methoxy-2-(3-Hydroxy-4-Methoxyphenyl)-Benzofuran-5-Carboxylate (1)
Colorless power.
MP: 165°C-168°C.
R f: 0.5 (hexane-acetone, 8:2, v/v).
1H-NMR and 13C-NMR (CDCl3): see Table 1.
HR-MS-ESI: found m/z 329.1019 [M+H], calcd for C18H17O6: 329.1025.
Cytotoxic Activity Assay
The 3-(4,5-dimethylthiazolyl)-2,5-diphenyl-tetrazolium bromide (MTT) assay detects the reduction of MTT (Sigma) by mitochondrial dehydrogenase to blue formazan product, which reflects the normal function of mitochondria and hence the measurement of cytotoxicity cell and viability. 23,24 Cells were maintained in Dulbecco’s modified Eagle medium, supplemented with 10% fetal bovine serum, l-glutamine (2 mM), penicillin G (100 UI/mL), streptomycin (100 µg/mL), and amphotericine B (10 µg/mL). Stock solution of test compounds were prepared in dimethylsulfoxide (DMSO) and diluted in H2O. The cytotoxicity assays were carried out in 96-well microplates with 1 × 104 viable cells/mL and incubated at 37°C in air/CO2 (95:5) with test compounds. After 72 hours incubation, 20 µL of MTT were added per well. After 4 hours incubation, medium was removed, 100 µL of DMSO were added per well and shaken for 5 to 10 minutes. Viable cells were estimated by optical density at 540 nm with Epocher 2 (BioTek) microplates reader. The IC50 value was determined as the concentration of compound that inhibits 50% cell growth compared to the control. Ellipticine was used as a reference compound.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Vietnam Academy Science and Technology (Grant no. VAST 04.03/1819).
