Abstract
Radix Bupleuri is a traditional medicine widely used in China and other Asian countries. Phytochemistry and pharmacology study reveal that saikosaponins(SSs) are the main bioactive compounds in Radix Bupleuri. SSs are complex compounds composed of triterpene aglycone and carbohydrate part containing 1-13 monosaccharides, which can be divided into seven types based on their structural characteristics. Many different kinds of SSs have been isolated from plants of Bupleurum L. SSs show a variety of biological activities, such as central nervous system protection, liver protection, antivirus, anti-tumor, anti-inflammation, hormone-like effects, and immune regulation functions. Due to their broad activity and favorable safety profile, SSs attract an increasing amount of attention in recent years. In this review, the structures of 86 SSs are summarized based on the different aglycones due to the diverse structures of saikosaponin(SS). The pharmacological effects and related mechanism of SSs are thoroughly reviewed, and perspectives for future research are further discussed.
Introduction
Radix Bupleuri is the root of Bupleurum chinense DC. or Bupleurum scorzonerifolium Willd., which has been used as a traditional Chinese medicine for more than 2000 years. These Bupleurum medicinal plants are widely distributed in the northern hemisphere, which are perennial herbs with compound umbels, bisexual flowers pale yellow or rarely purple, 5 stamens, fruiting, and single leaves long and slender.1,2 Radix Bupleuri is widely used as herbal medicines in China, Japan, South Korea, and other Asian countries.1–5 Radix Bupleuri was first recorded in Shennong's herbal classic, which is the first medical skill in China. Since then, Radix Bupleuri has been widely used in traditional Chinese medicine to treat colds, fever, influenza and hepatitis in monographs such as “Jinkui Yao Lue”, “Kai Bao Ben Cao”, “Compendium of Materia Medica” and “Xin Bian Ben Cao”.1,3 Clinically, several traditional Chinese medicine prescriptions made of Radix Bupleuri, including Xiaoyao pill, Xiaochaihu decoction, Dachaihu decoction, Chaihu Shugan powder, Chaihu Guizhi decoction and Buzhong Yiqi decoction, are all famous prescriptions with therapeutic effects.4,5 The different pharmacological activities should be attributed to the rich chemical components in Bupleurum species. 5 Essential oils, triterpenoid saponins, polyacetylenes, flavonoids, lignans, fatty acids, and sterols have been previously reported from plants of genus Bupleurum. 6 Among them, triterpenoid saikosaponins are important bioactive compounds. 7 SSs have a variety of biological activities, such as central nervous system and liver protection, anti-virus, anti-tumor, anti-inflammation, and immune regulation functions. SSs are complex compounds composed of triterpene aglycone (the carbon-skeleton of the aglycone consists of six isoprene units), and the carbohydrate part contains 1-13 monosaccharides to form a sugar chain that can be bonded to one or more hydroxyl groups of the aglycone. 8 In this review, SSs were classified and summarized based on the different aglycones (Figures 1-8 and Tables 1–8). The pharmacological activities and mechanisms of naturally occurring SSs were further reviewed.

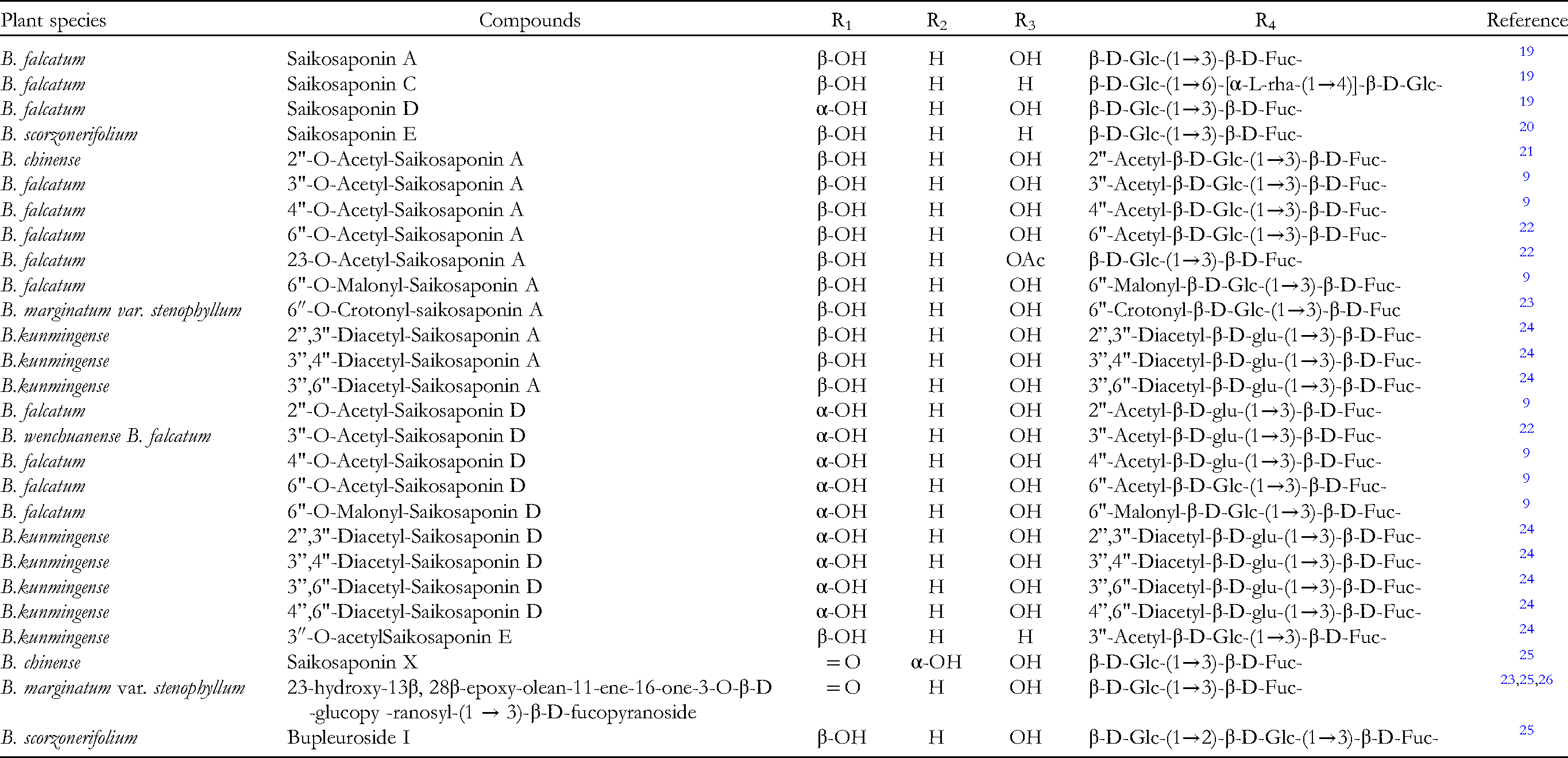
Structure of type I.
Type I Saikosaponins.
In this paper, related literatures such as articles collected from Web of Science, Elsevier, ScienceDirect, PubMed, Scopus, and SciFinder, Springer, Google scholar and China national knowledge internet (CNKI) are searched by computer. This study was carried out from the aspects of structure and pharmacology. The key words “Bupleurum”, “saikosaponin”, “structure” and “pharmacology” were searched individually or in combination.
Structures of Naturally Occurring SSs
SSs are important bioactive compounds in plants of genus Bupleurum. So far, several techniques have been employed to identify and analyze different saikosaponins, including TLC, HPLC, HPLC-ELSD, HPLC-MS, UPLC-MS and capillary electrochromatography. 3 Among the above methods, HPLC-ELSD is the most commonly used analytical method. 3 At present, a variety of triterpenoid saponins can be isolated from more than 10 species of genus Bupleurum. 2 For example, SSC can be isolated from B. chinense, SSA and SSC can be isolated from B. falcatum and B. marginatum, SSI can be isolated from B. kaoi, and 3'-O-acetylsaikosaponin a and 3'-O-acetylsaikosaponin d can be isolated from B. wenchuanense.
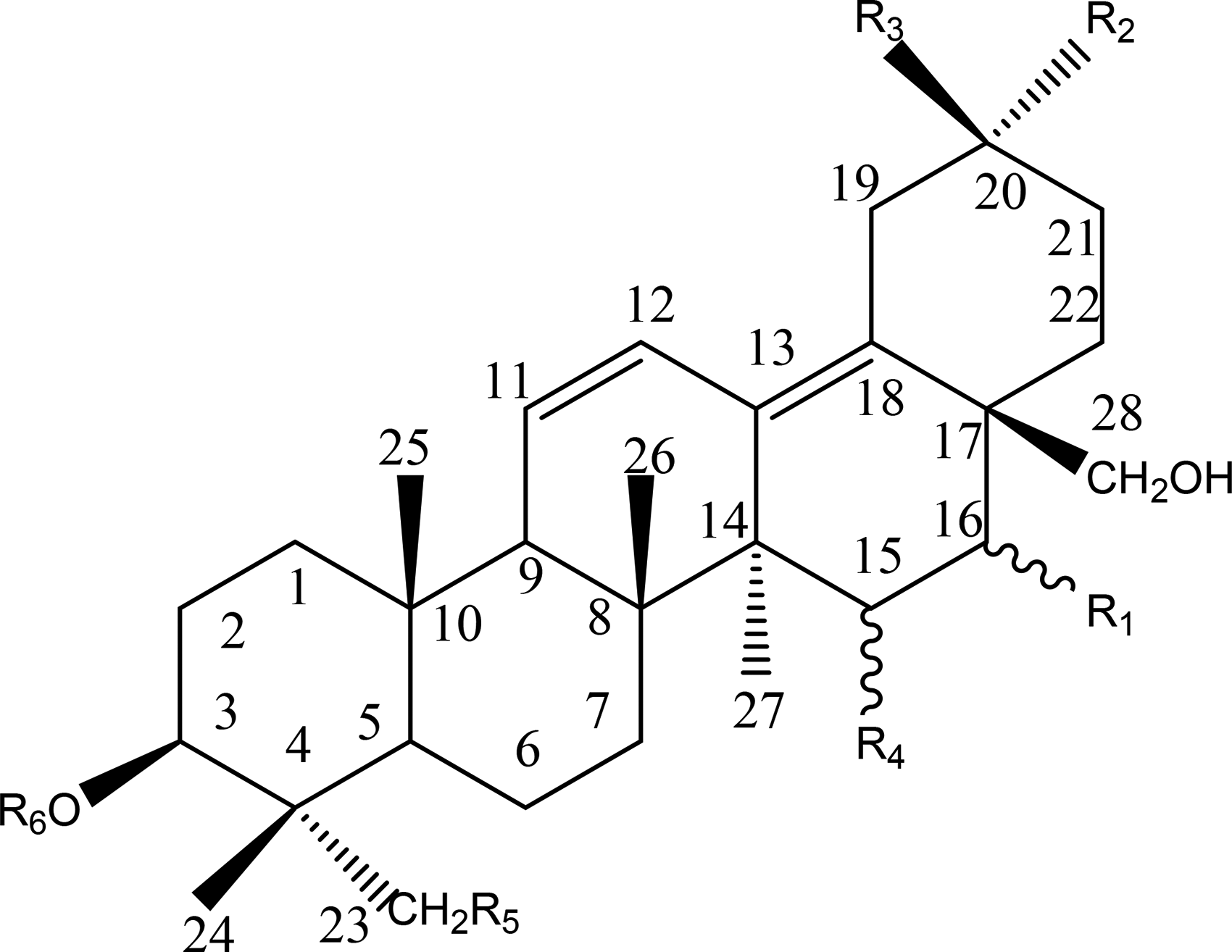
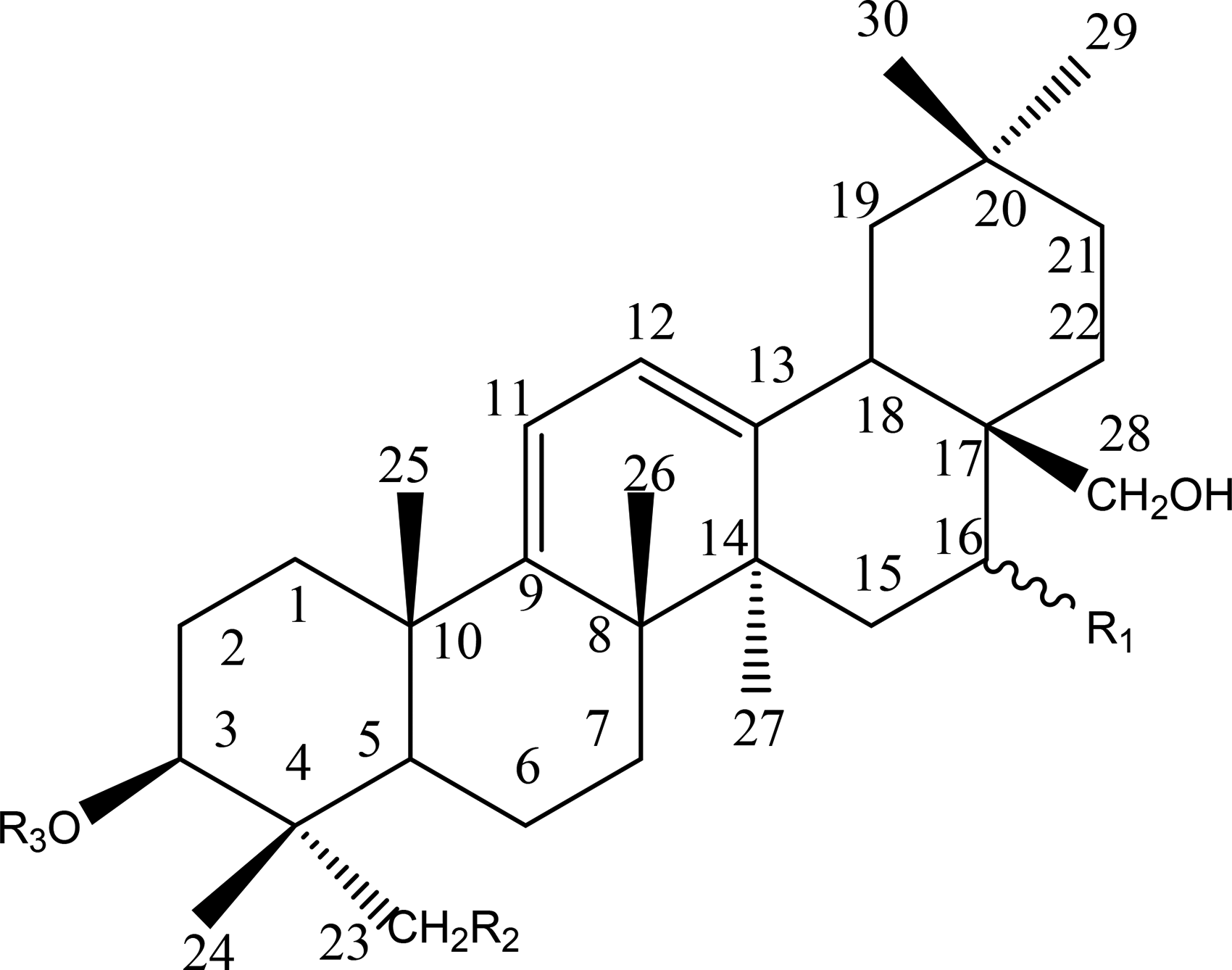
SSs are oleanane-type pentacyclic triterpenoid saponins composed of aglycones and sugar units, and their aglycones can be divided into seven different types: epoxy ether (I); isocyclodiene (II); 12-ene (III); homocyclic diene (IV); 12-ene-28-carboxylic acid (V); isocyclodiene-30-carboxylic acid (VI); and 18-ene (VII). SSs are generally the main components of secondary metabolites of the genus Bupleurum, accounting for almost 7% of the total dry weight of roots. The aglycones of these SSs are oxygen-containing pentacyclic triterpenes, which can only be distinguished by the position and number of double bonds on the C and D rings and the oxygenation patterns at positions of C16, C23, C28, and C30.9–11 The corresponding sugar chain is composed of fucose, rhamnose, xylose, galactose, and glucose. 2
The structure of type I saponins contains 13β, 28-epoxy ether bond and 11-alkene bond. Type II saponins contain two double bonds in different C rings, namely 11, 13(18)-diene or 12, 16(17)-diene. While the two double bonds are in the same C ring of type IV saponins, namely 9(11), 12-diene. There is only one double bond between C12 and C13 in type III saponins, while type V saponins show a similar 12-ene structure but contain a C28 carboxylic group. Type VI saponins contain 11, 13(18)-diene structure and C30 carboxylic group. Most of the type II, III and VI saponins are substituted by α-OCH3 at the C11 position. 12 Type VII saponins show 18-alkene structure. Generally, type I saikosaponins are the most abundant triterpenoid saponins found in plants of genus Bupleurum, and SSA, SSC and SSD are the most common saponins. 13
Conventionally, SSs are extract from Radix Bupleuri using traditional extraction processes such as reflux extraction, ultrasonic assisted extraction and solvent partitioning extraction.14–16 However, these methods have disadvantages such as time-consuming or waste of organic solvents. Recently, some advanced extraction methods have been employed for extraction of SSs. For example, accelerated solvent extraction (ASE)-based method was used to extract SSs from Bupleurum falcatum and it was reported that ASE method was more effective and faster under specific conditions, compared with previous traditional methods. 17 Supercritical fluid extraction with CO2 as solvent was also employed to extract SSs from Radix Bupleuri at lower temperature and lower organic solvent pollution. 18 Separation of SSs was principally performed by solvent partition coupled with different chromatography methods including reversed-phase and normal-phase silica gel column, macroporous resin column and preparative liquid chromatography, which are time consuming and with low recovery rate.
Epoxy Ether (I)
Isocyclodiene (II)
11, 13(18)-Diene
Structure of type II (1).
Type II (1) Saikosaponins.

12, 16(17)-Diene

Structure of type II (2).
Type II (2) Saikosaponins.

12-Ene (III)

Structure of type III.
Type III Saikosaponins.

Homocyclic Diene(IV)
Structures of type IV.
Type IV.

12-ene-28-Carboxylic Acid (V)

Structures of type V.
Type V.
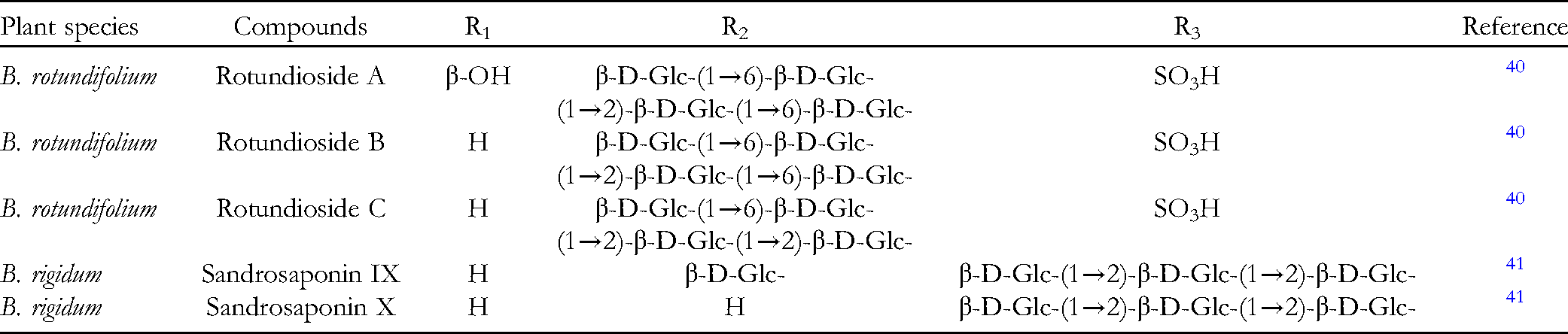
Isocyclodiene-30-Carboxylic Acid (VI)

Structure of type VI.
Type VI Saikosaponins.

Type VII Saikosaponin.

18-Ene(VII)
Pharmacological Effects of SSs
Anticonvulsant and Antiepileptic Activity
SSs exert significant effects on the central nervous system and can be used to treat convulsions, epilepsy, and other nervous system diseases. Previous studies found that spontaneous limbic seizures can lead to the increase in persistent sodium current (INAP), and the decrease in INAP might be related to the anticonvulsant effect of SSA. 45 SSA (0.3 μM∼4 μM) effectively terminates spontaneous recurrent epileptiform discharges in the hippocampal neuron culture (HNC) model of acquired epilepsy and continuous epileptiform high-frequency bursts in HNC of the status epilepticus model in a concentration-dependent manner, SSA inhibited spontaneous recurrent epileptiform discharge (SREDs) with an IC50(the half maximal inhibitory concentration) of 0.42 μM and SSA inhibited status epilepticus (SE) with an IC50 of 0.62 μM. 46 SSA is as effective as phenytoin (50 μM) in the complete inhibition of spontaneous recurrent epileptic discharge at 1 μM. SSA may play an anticonvulsant effect by inhibiting N-methyl-D-aspartic acid receptor current and INAP. 46 The anticonvulsant effects of SSA were observed at doses as low as 0.1 μM and as high as 4 μM. 47 SSA exerts an anticonvulsant effect by inhibiting epileptiform discharges of hippocampal CA1 neurons induced by 4-aminopyridine(4AP). 47 And in the 4AP convulsion model, SSA is similar to other anticonvulsant drugs (such as carbamazepine), which can reduce the seizure amplitude by more than 30 minutes, showing a longer lasting effect. 47
Anti-Alzheimer's Disease
SSA can improve learning and memory impairment induced by a monoclonal antibody, which may be achieved by inhibiting the pro-inflammatory mediators in the hippocampus. Li et al 41 found that the dose groups of 8 and 16 mg/kg/day of SSA can obviously improve the learning and memory impairment caused by amyloid-beta (Aβ) protein, and its mechanism might be related to the inhibition of pro-inflammatory mediators in the hippocampus. Nicotinamide adenine dinucleotide phosphate oxidase might also be involved in this effect. 48 SSC has dual effects on Alzheimer's disease by targeting two key proteins: Aβ and tau. 49 SSC reduces Alzheimer's disease by inhibiting the secretion of the Aβ peptide, inhibiting the microtubule depolymerization mediated by abnormal hyperphosphorylation of tau, and reducing synaptic destruction. 49 3 μM and 10 μM doses of SSC have a beneficial effect on cell tau function; it promotes axonal growth mediated by the nerve growth factor, increases microtubule assembly, and inhibits brain endothelial cell apoptosis induced by antipeptide. 49 SSC also increases synaptic marker proteins, including synaptophysin and postsynaptic dense postsynaptic density-95. 49
Oxidative stress (OS) injury is an important factor in the development and progression of senile neurodegenerative diseases (including Alzheimer's disease and Parkinson's disease). 50 SSD significantly improved the H2O2-induced decrease in the antioxidative ability of PC12 cell at concentrations of 200, 300, and 400 μg/mL. 51 SSD reduces the activation of the mitogen-activated protein kinase (MAPK) signaling pathway in a dose-dependent manner by scavenging reactive oxygen species (ROS), thereby alleviating H2O2-induced apoptosis and oxidative damage of PC12. 51 SSD may be a potential antioxidant for neurooxidative diseases and may relieve Alzheimer's disease-like symptoms.
Previous studies reported that early postmenopausal use of estrogen can delay or prevent the progression of Alzheimer's disease.52,53 SSD is similar to estradiol in structure and has an estrogenic effect. 54 Zeng et al verified the protective effect of SSs on the Aβ-induced neuronal death AKT (protein kinase B) signaling pathway by using network pharmacology. 55 Du et al 56 elucidated the possible protective effects of SSD on glutamate-induced neurotoxicity in SH-SY5Y cells and the underlying mechanism. They found that SSD(0.5, 1, 5, 10 μM) enhanced cellular antioxidant capacity through not only intrinsic free radical-scavenging activity but also induction of endogenous antioxidant enzyme activities and heme oxygenase-1 (HO-1) expression mediated, at least in part, by activating phosphatidylinositol 3-kinase (PI3K) and subsequently Nrf2 nuclear translocation, thereby protecting the SH-SY5Y cells from glutamate-induced oxidative cytotoxicity. In concert, these data raise the possibility that SSD may be an attractive candidate for prevention and treatment of Alzheimer's disease and other diseases related to oxidation in the future.
Antidepressant Activity
SSA has obvious anti-inflammatory activity; it can improve cytokines and regulate inflammation-related pathways. 57 Moreover, SSA has potential antidepressent activity. In accordance with the inflammation hypothesis of depression, stress stimulation can trigger the inflammatory process, leading to abnormal changes in the normal physiological function of 5-hydroxytryptamine and hypothalamus-pituitary-adrenal (HPA) axis. 58 In addition, high levels of tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) have been found in injured brain tissue, 59 and SSA decreases the levels of interleukin-1 beta (IL-1β), IL-6, and TNF-α in the chronic unpredictable mild stress (CUMS) depression model of peri-menopausal female rats. 60 In addition, SSA improves desperate-like depressive behavior during peri-menopause, which may be achieved by improving neuroendocrine, neuroinflammatory, and neurotrophic systems. 60 The level of brain-derived neurotrophic factor (BDNF) is low in serum of depressed patients. 61 Animals that exhibit symptoms similar to depression also express less BDNF in the serum or brain.62,63 In addition, treatment associated with depression increases BDNF levels.64,65 Chen et al 60 administered SSA to rats by intragastric administration of 25, 50, 100 mg/kg/day SSA, with fluoxetine of 10 mg/kg/day as control. And SSA decreases the expression of BDNF induced by CUMS at 4 weeks after administration of SSA. 60 In addition, SSA can restore the deranged regulation of the HPA axis and neuroinflammatory response induced by CUMS, promote the signal transduction of BDNF–tyrosine kinase receptor B (BDNF-TrkB) in the hippocampus, and inhibit the activation of microglia and the release of pro-inflammatory cytokines in the brain. 60 SSA reverses depression-like behavior by maintaining the normal regulation of the HPA axis and activating the BDNF signal. 60 Proline-rich transmembrane protein-2 (PRRT2; one of 15 differentially expressed proteins in the hippocampus) is mainly located in the presynaptic terminals of neurons and plays a key role in the release of neurotransmitters. 66
In CUMS rats, long-term intragastric administration of SSD (0.75 mg/kg, 1.5 mg/kg) could significantly improve the behavioral defects induced by UCMS, with fluoxetine hydrochloride (10 mg/kg) as the positive control. 67 SSD improves their depression-like behavior by restoring the level of serum cortisol and improving the activity of glucocorticoid receptor (GR), correcting the dysfunction of the HPA axis. 67 In addition, SSA promotes hippocampal neurogenesis by increasing the production of neurons and increasing the level of neurotrophic molecules. 67 Recent evidence has shown an increase in the secretion of high mobility group box-1 (HMGB1), which translocates from the nucleus to the extracellular membrane and binds to transmembrane receptors such as Toll-like receptors on the surface of microglia in chronic stress-induced depressed models.68–70 Subsequently, phosphorylation of IκBα, an inhibitor of nuclear factor kappa-B (NF-κB), inhibits the release of pro-inflammatory factors. 71 The downstream NF-κB signaling pathway is then activated to trigger the pro-inflammatory response.69,70 After administration of the non-selective antagonist of HMGB1, the depressive behavior of mice improves. 68 Su et al 72 observed that lipopolysaccharide (LPS) could induce inflammation-related depressive behavior in mice, SSD (1 mg/kg) for 7 days, and relieved the depression-like behavior of mice. SSD pretreatment down-regulates the protein level of HMGB1 in the cytoplasm of mouse hippocampus and inhibits the translocation of HMGB1 from the nucleus to cytoplasm in primary microglia. 72 SSD pretreatment reverses the activation of Toll-like receptor 4 (TLR4) and the phosphorylation of IκBα in the hippocampus of mice induced by LPS, decreases the protein level of NF-κB in the nucleus of hippocampus, and inhibits the activation of microglia and the release of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α). 72 Targeted lysophosphatidic acid receptor 1 (LPA1) may be a potential treatment for depression, which is related to the regulation of neuronal apoptosis. 73 SSD was dissolved in normal saline and given by intragastric administration of 0.5 and 1 mg/kg/d for 2 weeks, and the two doses of SSD preconditioning could significantly block the prolongation of immobility time induced by LPS stimulation. 74 SSD targeting LPA1 attenuates the activation of the Ras homolog gene family member A/MAPK/NF-κB signaling pathway induced by LPS. 74 Chao et al 75 administered SSD to rats by intragastric administration of 0.75 mg/kg/day and 1.50 mg/kg/day with fluoxetine as positive control (10 mg/kg/day), and SSD significantly improved the depression-like behavior induced by CUMS in rats. SSD can down-regulate the expression of fibroblast growth factor-2 mRNA by negatively regulating NF-κB and positively targeting microRNA-155, thereby attenuating CUMS-induced depression-like behavior in rats. 75
Total SS (TSS, 25 μg/mL) purified from B. yinchowense, which mainly contains SSA, SSC, SSD, SSC and SSF, can partially reverse the neuropathy in corticosterone-induced PC12 cells. The mechanism may be related to stabilize Ca2+ homeostasis and regulate the B-cell lymphoma-2 (Bcl-2) family. 76 Furthermore, TSS stabilizes the endoplasmic reticulum and inhibits the mitochondrial apoptosis pathway. 76 Therefore, the protective effect of TSS on cells may contribute to its antidepressant drug-like effect. Guo et al 77 aimed to explore the anti-depression effect of SSA and screen the target proteins regulated by SSA in a rat model of CUMS-induced depression. Results showed that 8-week CUMS combined with separation could successfully produce depressive-like behaviours and cause a decrease of dopamine (DA) in rat hippocampus, and 4-week administration of SSA (50 mg/kg) could relieve CUMS rats’ depressive symptoms and up-regulated DA content. There were 15 kinds of significant differentially expressed proteins that were detected not only between the control and CUMS groups, but also between the CUMS and SSA treatment groups. PRRT2 was down-regulated by CUMS while up-regulated by SSA. These findings reveal that SSA may exert antidepressant effects by up-regulating the expression level of PRRT2 and increasing DA content in hippocampus.
Hepatoprotective Activity
The liver protective effects against D-galactosamine hydrochloratemine (D-GalN) induced liver injury were investigated after treatment of rats with each kind of SSs (SSA, SSD, SSB1, SSB2 and SSC) (5 mg/kg /day, i.p.) for 4 days. 78 The results showed that SSA and SSD have significant inhibitory effects on liver injury induced by D-galactosamine hydrochloratemine (D-GalN). They can reduce the activity of drug metabolism in the whole body of rats, resulting in a significant decrease in microsomal enzyme activity and P450 content, and SSD shows much stronger effect than SSA. 78 However, SSB1, SSB2 and SSC did not cause any change in enzyme activities. 78 And long-term intraperitoneal injection of different doses (1.0, 1.5 and 2.0 mg/kg/d) of SSD has a certain therapeutic effect on hepatic fibrosis and down-regulates the expression of TNF-α, IL-6, and NF-κBp65 in rat liver tissue. 79 In the study of Lin et al, different doses of SSD (0.5, 1 or 2 μM) could attenuate acute hepatocyte injury induced by CCl4 in HL-7702 cells. 80 The results showed that SSD played an antioxidant role by inhibiting the production of malondialdehyde (MDA) and increasing the level of total superoxide dismutase to alleviate the acute hepatocyte injury of HL-7702 cells induced by CCl4. 80 And SSA (5 μM) can regulate the level of bone morphogenetic protein-4 (BMP4) growth factor BMP4 by inhibiting the expression of alpha-smooth muscle actin (α-SMA) and preventing the activation of hepatic stellate cells (LX-2 cells). 81 Therefore, SSA and SSD can be used to treat liver diseases with increased expression of BMP4 in a dose-dependent manner. 81 In addition SSA(5,10 and 20 mg/kg, i.p) could attenuate liver injury induced by LPS (60 mg/kg) and D-GalN (800 mg/kg) in mice. 82 SSA might inhibit the NF-κB signaling pathway and inflammation by increasing the expression of liver X receptor alpha (LxRα) and then prevents LPS/D-GalN-induced liver injury in a dose-dependent manner. 82
Only estrogen receptor beta (ERβ) is expressed in primary cultured rat stellate cells but not estrogen receptor alpha (ERα). 83 SSD (5 µM) prevents the activation of hepatic stellate cells induced by OS depend on ERβ activity, and may be at partially attributed to inhibition of the ROS/MAPK signaling pathway. 84
Li et al 85 found that both SSA(5,10, and 20 mg/kg) and SSD (5, 10, and 20 mg/kg) improved diet-induced Nonalcoholic Fatty Liver Disease (NAFLD). Integrative lipidomic and transcriptomic analysis revealed that SSA and SSD modulated glycerolipid metabolism by regulating related genes, like Lipe and Lipg. SSD profoundly suppressed the fatty acid biosynthesis by downregulating Fasn and Acaca expression and promoted fatty acid degradation by inducing Acoxl and Cpt1a expression. Bioinformatic analysis further predicted the implication of master transcription factors, including peroxisome proliferator-activated receptor alpha (PPARα), in the protective effects of SSA and SSD.
Chang et al 86 also showed that SSD has a hepatoprotective effect in liver injury by suppressing inflammatory responses and acting as an antioxidant. The SSD (2 mg/kg) group showed significantly higher food intake, body weight, and hepatic antioxidative enzymes such as catalase (CAT), glutathione peroxidase (GPx) and superoxide dismutase (SOD), lower hepatic cyclooxygenase-2 (COX-2), serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), IL-1β, TNF-α, and fibroblast growth factor-21 (FGF21) compared with controls. SSD also reduced the mRNA expression of inflammation-related genes such as nuclear factor kappa B (NFκB) and inducible nitric oxide synthase (iNOS). In NAFLD mice, SSD reduced serum ALT, AST, triglycerides, fatty acid-binding protein 4 (FABP4) and sterol regulatory element-binding protein 1 (SREBP1) mRNA, and endoplasmic reticulum (ER)-stress-related proteins, including phosphorylated eukaryotic initiation factor 2αsubunit (p-eIF2α), activating transcription factor 4 (ATF4), and C/EBP homologous protein (CHOP).
Antivirus Activity
SSA, SSB2, SSC, and SSD have activities against human immunodeficiency virus (HIV), measles, influenza virus, herpes simplex virus, and Streptococcus hydrophila virus.
The results of Cheng et al 87 showed that SSs have antiviral activity in the range of 0.25–25 μM, among which SSB2 has the strongest antiviral activity (IC50 = 1.7 ± 0.1 μM). SSB2 is the most selective SS for inhibiting hepatitis C virus (HCV) infection. 88 SSB2 can neutralize virus particles, inhibit virus attachment, and prevent virus invasion. 88 SSB2(10, 100 μM) can inhibit the infectivity of HCV, and SSB2 did not affect cell viability up to 100 µM in Huh-7.5.1 cells. 89 The results show that SSB2 can neutralize the virus or induce conformational changes in the glycoprotein so that the virion is not contagious, and SSB2 can effectively prevent the entry of HCV virus into cells and RNA replication. 89 SSB2 has been found to inhibit the infection of several other viruses (measles virus, dengue virus, herpes simplex virus type I, and unenveloped reovirus). 88 In addition SSC exhibited an anti-HBV activity against secretion of HBeAg (IC50 = 11 μg/ml) and expression of HBV DNA (IC50 = 13.4 μg/ml). 90 SSC (2∼40 mg/mL) stimulates the expression of IL-6, down-regulates the host transcription factors hepatocyte nuclear factor 1α and hepatocyte nuclear factor 4α, and inhibits the synthesis of HBV pgRNA on HepG2 and HepG2.2.15 cells. 91 SSD (5 μM) can inactivate measles and herpesvirus. This effect may be caused by breaking the envelope, which is due to the interaction between SSD and the sialic acid residues of the viral glycoprotein on the envelope. 92
Evidence show that type I SSs not only exert potent anti-influenza virus activity but also strong cytotoxicity. In addition, nepasaikosaponin K, SSN, and SSH are promising antiviral drugs in terms of inhibition and selectivity. 23 Enterovirus A71 (EV-A71), the cause of hand-foot-mouth disease, has been shown to induce autophagy. It is found that SSD (15, 30 μM) potently inhibited EV-A71 RNA replication and subsequent viral protein synthesis, thereby preventing EV-A71-induced cell death. 93 ATG5 knockdown inhibited EVA71 viral protein synthesis, whereas autophagy induction by rapamycin promoted synthesis. These data indicate that SSD and SSA are potent late-stage autophagy inhibitors that can be used to prevent EV-A71 infection. In addition, the emergence of viral pneumonia caused by a novel coronavirus (CoV), known as the 2019 novel coronavirus (2019-nCoV), resulted in a contagious acute respiratory infectious disease. SSD showed antiviral activities against different respiratory viruses in vitro and in vivo. 94
Antitumor Activity
SSs can inhibit the adhesion of some solid tumor cells, interfere with tumor cell proliferation cycle, interfere with protein metabolism, inhibit cell proliferation, and induce cell apoptosis and other anti-tumor effects. 95 SSA, SSD, and SSE can significantly inhibit tumors by reducing the adhesion of solid tumor cells. 95 SSA can activate caspase-2 and caspase-8 to trigger apoptosis of hepatoma cells. 96
SSD has a glucocorticoid-like steroid structure that can inhibit Na+-K+-ATP enzyme activity; it also has a potential anti-tumor effect. 95 And SSD inhibits the activation of NF-κB induced by TNF-α and participates in the expression of target genes involved in cancer cell proliferation, invasion, angiogenesis, and survival. 97 SSD could apparently inhibit the TNF-α induced invasive ability of H1299 human lung cancer cells at nontoxic concentrations(10 μM), suggesting that SSD-mediated inhibitory effect on cancer cell invasion is specific, and it has no direct cytotoxicity of the drug. 97 SSD (10, 20 μM) has been reported to show effective antitumor activity through inhibited the cell growth of human lung cancer cell line A549 in a dose-and time-dependent manner. 98 And the activity of SSD-induced p53 and Fas (TNF receptor superfamily member 6)/Fas ligand (FasL) apoptosis system may be involved in the inhibitory effect of SSD on the proliferation of A549 cells. 98 P53 can regulate various downstream target genes related to apoptosis and cell cycle arrest, such as Bax, Bcl-2, and p21.26,99 As an inhibitor of cell cycle progression, the upregulation of p21 protein will lead to cell cycle arrest in the G1 phase. 100 In addition, cyclin-dependent kinase 2 (CDK2) is a member of the silk cyclin-dependent kinase family, which plays an important regulatory role in the G1/S phase. 101 The treatment with SSD (3μg/mL) can significantly induce apoptosis of SMMC7721 cells under hypoxia and hypoxia conditions, and SSD increases radiosensitivity and induces the apoptosis of SMMC7721 hepatoma cells. 102 In another study, SSD(0, 1, 2.5, 5, 10, 20 or 50 μM) inhibited the proliferation of DU145 cells in a concentration-dependent manner, and mitochondrial dysfunction induced by SSD is the main reason for the apoptosis of human prostate cancer cell DU145 induced by SSD. 103 According to Wang et al, 104 SSD (10, 15 or 20 μM) could lead to tumor cell apoptosis through inhibit Wnt (Wingless/Integrated)/β-catenin signal transduction and the expression of β-catenin and its downstream target genes but does not act on epidermal growth factor receptor and neurotensin receptor 1. SSD (1-5 μg/ml), as a reversal agent of multidrug resistance mediated by P-glycoprotein (P-gp), down-regulates the expression of multidrug resistance gene 1/P-gp and significantly improves the chemosensitivity of MCF-7 (human breast cancer cell line) to Adriamycin. 105 SSA (10 μM) and SSD (5 μM) were cytotoxic to A375.S2 cells, while the concentration of SSC reached 20 μM showed no antiproliferative activity. Therefore, SSD was the most potent compound in terms of anti-melanoma activity. 106 SSD nanoparticles induce apoptosis through the mitochondrial pathway. 106 Their anti-melanoma activity is mediated by the phosphorylation of c-Jun N-terminal kinase, the phosphorylation of p38 and p53, the increase in cytochrome c level, and the activation of caspase-9. 106
SSB2 (64 μM, 128 μM) enhances the efficacy of liver-targeted anticancer drugs by inhibiting multidrug resistance-related drug transporters (Pgp, MRP1/Mrp1 and MRP2/Mrp2). 107 Therefore, SSB2 improves the efficiency of chemotherapeutic drugs and prevents the development of multidrug resistance in hepatocellular carcinoma. However, the combination of platinum-containing anticancer drugs with SSB2 should be avoided. 107 Cancer chemotherapy induced neutropenia (CCIN) is one of the most common toxicity caused by cytotoxic anticancer agents. A study exploring the function of SSD in CCIN therapy found that SSD (6,12 mg/kg) contributed to generate functional mature neutrophils which capable of fighting infection both in vitro and in vivo. 108 Network pharmacology was employed to explore the mechanism, 61 signal pathways might play an important role in CCIN treatment. Western Blot was employed to further confirm the potential pathway involved. They found CBL- ERK1/2 pathway was activated by SSD, followed by upregulating PU.1 and CEBPβ expression and leading to neutrophil differentiation. Their findings suggest a natural regimen SSD which could regenerate microbicidal neutrophils to effectively reduce CCIN-associated infection via activating CBL-ERK1/2, providing a rationale for future therapeutic approaches. In addition,as a highly efficient anticancer agent, Doxorubicin (DOX) is used for various cancers’ treatment, but DOX-induced oxidative damages contribute to a degenerative irreversible cardiac toxicity. Recent study 109 has shown that 1 μM SSD could enhance the proliferation of H9c2 cells, and inhibit DOX-induced apoptosis,and SSD(10 mg/kg) efficiently protected the cardiomyocytes from DOX-induced cardiotoxicity by inhibiting the excessive OS via p38 MAPK signaling pathway. Although SS has the effects of anti-tumor, yet we still do not know the mechanism by total Bupleurum saponin extracts (TBSE) produces this effect on colon cancer. Zhang et al 110 firstly confirmed that TBSE(50ug/ml) significantly up-regulated the expression of pro-apoptotic proteins Bax, Caspase3, Caspase9, cleaved Caspase3 and cleaved Caspase9, and down-regulated the expression of anti-apoptotic protein Bcl-2, significantly down-regulate (P < 0.01) the expressions of PI3 K, Akt, mTOR and phosphorylated proteins P-PI3 K, P-Akt, P-MTOR. Chemosensitivity is also one of the key factors affecting the therapeutic effect on cancer, but the clinical application of corresponding drugs is rare. Zhang et al 111 first found that SSD (6 µM) inhibits the malignant phenotype of HCC cells while increasing their sensitivity to the herpes simplex virus thymidine kinase/ganciclovir (HSVtk/GCV) drug system under hypoxia in vitro and in vivo, and SSD could reverse the effects promoted by hypoxia, specifically active sentrin/small ubiquitin-like modifier (SUMO)-specific protease 5 (SENP5), a SUMO-specific protease, in a time- and dose-dependent manner while inhibiting the expression of SUMO1 and GLI proteins. These findings confirm the important role of SSD in the chemoresistance of liver cancer, provide some data support for further understanding the molecular.
Anti-Inflammatory Activity
SSs exhibit anti-inflammatory activity, 112 and SSD has the strongest anti-inflammatory effect among all SSs. SSD can inhibit a variety of inflammatory processes, including inflammatory exudation, increased capillary permeability, release of inflammatory mediators, leukocyte migration, and connective tissue proliferation.57,113 The production of inflammatory cytokines is regulated by the NF-κB signaling pathway. 114 The results of Lu et al 57 showed that both SSA (3.125 μΜ∼12.5 μΜ) and SSD (3.125 μΜ∼50 μΜ) can inhibit the activation of NF-κB, thereby inhibiting the production of inducible nitric oxide synthase, COX-2, and proinflammatory cytokines in LPS-induced RAW264.7 macrophages. 57
LPS can also induce the activation of the NF-κB signaling pathway. 115 SSA (12.5∼100 μM) could significantly reduce the survival rate of RAW264.7 cells. 116 SSA can inhibit pro-inflammatory cytokines in LPS-stimulated macrophages and promote the production of anti-inflammatory cytokines, and its mechanism is related to the regulation of MAPK and NF-κB signals. 116 And SSA (50 µM) can inhibit the signal transduction of PI3 K/AKT/NF-κB/NOD (nucleotide-binding oligomerization domain)-like receptor family 3 and the expression of inflammatory cytokines in human acute monocytic leukemia cell line THP-1 cell. 117 In addition SSA(5∼20 mg/kg/day, i.p) can significantly inhibit the production of nitric oxide (NO), TNF-α, and IL-1β induced by cigarette smoke (CS). 118 SSA inhibits the contents of myeloperoxidase (MPO) and MDA in lung tissue induced by CS and significantly inhibits NF-κB induced by CS. 118 A study in which the mice received SSA (5, 10, and 20 mg/kg) intraperitoneally 1 h before LPS treatment revealed that SSA up-regulates the expression of nuclear factor E2-related factor 2 (Nrf2) (Nrf2 can regulate the inflammatory response 119 ) and HO-1 in a dose-dependent manner. 118 SSA may inhibit NF-κBp65 and IκBα phosphorylation induced by LPS and inhibit the NF-κB signaling pathway by activating Nrf2, thereby producing anti-inflammatory effect. 120 LxRα is involved in the regulation of inflammation. 121 Activation of LxRα attenuates the activation of NF-κB. 122 According to report of Zhou et al, the mice were received SSA (5, 10, 20 mg/kg/d, i.g.) for 14 days and the results shown that SSA can dose-dependently inhibit TNF-α and NF-κB activation induced by dextran sulfate sodium (DSS), up-regulate the expression of LxRα, and significantly inhibit the level of IL-1β. 123 SSA(2 mg/kg /day and 10 mg/kg/day) inhibits IL-6 signal transduction, signal transducer and transcriptional activator 3 (STAT3)/retinoid-related orphan nuclear receptor γt (ROR-γt), and the NF-κB pathway by regulating T helper cell 2/T helper cell 17 (Th2/Th17) cytokines. 124 In the study of Ali et al, SSA (1,5,10 mg / kg / day) was administered one hour before 5-fluorouracil (5-FU) injection for 7 days. 125 The results shown that SSA significantly inhibits pro-inflammatory mediators such as TNF-α, COX-2, IL-1β, and IL-6, and SSA significantly inhibits apoptosis markers such as phosphorylated c-Jun N-terminal kinase (p-JNK) and caspase-3. 125
SSD can also significantly decrease the level of ROS, inactivate P38 and JNK, and inhibit the NF-κB signaling pathway. 126 SSD was intragastrically perfused with 8 mg/kg/day to improve intestinal inflammation induced by DSS. 127 And SSD can increase the expression of anti-inflammatory cytokine interleukin-10 mRNA. 127 Thus, SSD decreases the release of pro-inflammatory cytokines (TNF-α, IL-1β, and IL-6). Total SS purified from Radix Bupleuri, which mainly contains SSA, SSB2, SSC and SSD, shows obvious anti-inflammatory effect on formalin-induced mice. 128 In terms of plasma metabonomics, SS plays an anti-inflammatory role by regulating the metabolism of nicotinic acid, nicotinamide, and arachidonic acid. The study indicating that the development of inflammation was inhibited in all dose groups with SS-treated mice, especially in HD-SS-treated (4.68 g/kg) mice. 128 And the inhibited effect of SS on paw edema was similar to that of aspirin. 128
Wu et al 129 evaluated the potential therapeutic properties of SSD in LPS-induced inflammatory bone loss mouse model and found that SSD suppressed LPS-induced inflammatory bone loss in vivo. Further mechanism study revealed that SSD (2 μM) inhibited the formation and bone resorption of osteoclasts induced by Receptor Activator of Nuclear Factor-κ B Ligand (RANKL) in vitro.
Hormone-Like Activity
Radix Bupleuri is believed to be able to treat some gynecological diseases, such as menopausal symptoms. 130 The chemical structure of SSD is similar to that of estradiol. It plays an estrogenic role by binding to the estrogen receptor and is a weak phytoestrogen. 54 SSD (0.01 μM∼10 μM) stimulated the growth of MCF-7 cells and significantly affected the cell cycle changes of MCF-7 cells. 54 The proliferation-promoting effect of SSD can be reversed by the combination of anti-estrogen ICI-182780. 54 Therefore, SSD has potential estrogenic activity in MCF-7 cells. 54 As an agonist of estrogen receptor, SSD could activate estrogen response element (ERE)-luciferase activity via the ERα-mediated pathway in a dose-dependent manner (10 nM to 10 µM); and the activation of ERβ-mediated ERE-luciferase activity by SSD only occurred at a high concentration (10 µM). 54 SSD can increase the expression of ERα protein and mRNA and activate ERα preferentially. 54
SSA can increase the levels of plasma adrenocorticotropin (ACTH) and corticosterone in vivo. 131 In addition, SSD (0.2, 2.0 and 20 µg/kg, i.c.v.) has a special effect on corticotropin-releasing factor (CRF) neurons. 132 SSD may stimulate the expression of the CRF gene and the release of CRF, which in turn stimulates the ACTH secretion of anterior pituitary and the expression of the proopiomelanocortin gene in a dose-dependent manner. 132
Immunomodulatory Activity
Previous studies have shown that SSA, SSF, and 23-O-acetylsaikosaponin a have moderate immunomodulatory activity, whereas SSD has the strongest immunomodulatory activity among all SSs. 133
SSA (2, 10 mg/kg/day, i.g.) can reduce the level of ovalbumin-specific immunoglobulin IgE/IgG1 in BALB/c male mice. 124 Sun et al 134 indicated that SSA (5 and 10 µM) selectively promotes the apoptosis of activated T lymphocytes, thereby avoiding non-specific immunosuppression. SSA destroys the mitochondrial membrane potential in a dose-dependent manner, resulting in the release of cytochrome c from the mitochondria to the cytoplasm. 134 SSA can also block the G0/G1 phase and induce T lymphocyte apoptosis through the mitochondrial pathway. 134
SSD (10 and 20 µM) can significantly inhibit the expression of early (CD69) and late (CD71) T lymphocytes stimulated by concanavalin A or phorbol 12-myristate 13-acetate (PMA). 135 CD69 may be involved in the pathogenesis of some autoimmune and inflammatory diseases. 136 SSD interferes with the transport of protein kinase Cθ (PKCθ) from the cytoplasm to membrane and inhibits the phosphorylation of IκBα and JNK in PMA-activated mouse T lymphocytes, but it does not inhibit the phosphorylation of extracellular signal-regulated kinase. 135 SSD regulates protein kinase C pathway through PKCθ, JNK, and NFκB. 135
Anti-Asthmatic Activity
In the study of Park et al, SSA (1 mg/kg∼10 mg/kg, i.v.) has anti-asthmatic effects in rats. SSA exerts partial anti-allergic or anti-asthmatic effects and significantly inhibits bronchoconstriction by directly antagonizing the histamine effect and reducing the release of histamine from mast cells. 137 SSB is a direct TAS2R14 (TAS2R14, as a bitter receptor, has the function of bronchiectasis and anti-inflammation) agonist, which can inhibit IgE-induced mast cell degranulation. 138 Further studies are needed to verify their anti-asthmatic activity. Chloroquine, as a positive control, significantly inhibited IgE-induced hexosidase release from mast cells at 1000.0, 500.0 and 250.0 μM. In addition, SSB at 10.0 μM and 5.0 μM inhibited IgE-induced mast cell degranulation. 138
Anti-Adipogenic Effects
Obesity is a lipid metabolism disorder caused by genetic, medicinal, nutritional, and other environmental factors. It is characterized by a complex condition of excess lipid accumulation in adipocytes. Adipogenesis is a differentiation process that converts preadipocytes into mature adipocytes and contributes to excessive fat deposition. Lim et al 139 investigated the anti-obesity effects of SSA and SSD in mouse 3T3-L1 adipocytes. They showed that SSA and SSD significantly inhibited lipid accumulation without affecting cell viability within the range of the tested concentrations (0.938-15 µM). SSA and SSD also dose-dependently suppressed the expression of peroxisome proliferator-activated receptor gamma (PPARγ), CCAAT/enhancer binding protein alpha (C/EBPα), sterol regulatory element binding protein-1c (SREBP-1c), and adiponectin. Furthermore, the decrease of these transcriptional factors resulted in the repressed expression of several lipogenic genes including fatty acid binding protein (FABP4), fatty acid synthase (FAS), and lipoprotein lipase (LPL). In addition, SSA and SSD enhanced the phosphorylation of adenosine monophosphate-activated protein kinase (AMPK) and its substrate, acetyl-CoA carboxylase (ACC), and inhibited the phosphorylation of extracellular-regulated kinase 1/2 (ERK1/2) and p38, but not c-Jun-N-terminal kinase (JNK). These results suggest that SSA and SSD inhibit adipogenesis through the AMPK or MAPK pathways in the early stages of adipocyte differentiation. This is the first study on the anti-adipogenic effects of SSA and SSD, and further research in animals and humans is necessary to confirm the potential of SSs as therapeutic agents for obesity.

Structure of type VII.

Pharmacological activities of Saikosaponins.
Pharmacological Effects of SS with Detailed Information.

Structure-Activity Relationship
As mentioned above, SSA and SSD have obvious protective effect on liver injury caused by D-galactosamine hydrochloride (D-GalN), which can significantly reduce microsomal enzyme activity and P450 content, and the effect of SSD is obvious stronger than SSA. 78 However, SSB1, SSB2 and SSC did not alter microsomal enzymatic activity. 78 In addition, SSA and SSD can induce apoptosis of tumor cells and are cytotoxic to tumor cells,104,106 but SSC has no anti-proliferative activity at high concentration, which may be due to the different types of sugar chains on C-3. And the anti-proliferative activity of SSD is stronger than that of SSA.104,106 SSA can increase ACTH levels in plasma. 132 The role of SSA in the stimulation of CRF neurons appears to be small. 132 The specific effect of SSD on CRF neurons can stimulate the expression of CRF gene and the release of CRF, thereby promoting the secretion of ACTH in the anterior pituitary, and SSD has the strongest anti-inflammatory activity. 132 In addition, the 13,28-epoxy group, as the unique structure of type I saponins, show good anti-influenza activity and strong cytotoxicity. 23 SSs with different ethylenic structures show different anti-influenza activities, which indicates that different ethylenic structures may affect the pharmacological activity of SSs. 23 However, some SSs with same aglycone structure have different anti-influenza activities, which may also be due to the effect of different types of sugar chains on C-3 on the activity. 23 In addition, SSN and SSH showed similar EC50 (50% effective concentration) values, indicating that the hydroxyl groups on C-23 show almost no effect on antiviral activity. 23 And the direction of 16-OH had a slight effect on the antiviral activity. 23 Nepasaikosaponin K showed more effective antiviral activity than 11-α-methoxy-saikosaponin F, which indicated that the methoxy group of C-11 might weaken the antiviral activity. 23
It can be seen from the above that 13,28-epoxy group, as a unique structure of type I saponins, can enhance the protective effect of liver injury, enhance the anti-tumor effect, increase the level of ACTH by stimulating the expression of CRF gene and the release of CRF, enhance anti-influenza parade and have greater cytotoxicity.23,104,106,132 The hepatoprotective activity, anti-tumor activity, anti-inflammatory activity and immunomodulatory ability of SSD were stronger than those of SSA.23,104,106,132 It is suggested that 16α-OH has stronger pharmacological activity than 16β-OH. And the methoxy group of C-11 and the different alkene bonds on different types of saponins also affect the anti-influenza virus activity.
Conclusion
Studies in vivo and in vitro have shown that SSs are a group of widely distributed triterpenoids. SSs play an important role in anticonvulsant, antiepileptic, anti-Alzheimer disease, antidepressant, liver protection, antiviral, anti-tumor, anti-inflammatory, hormone-like activity and immunomodulatory function. The structures of SSs are closely related to their pharmacological activity. At present, research on the structure-activity relationship of SSs is still lacking. Future work is necessary to better understand the structure-activity relationship of SSs for clinical application.
By reviewing pharmacological activities of SSs, this work provides a basis for further studying the mechanism of action of SSs and developing better therapeutic drugs with SSs in the future. However, the detailed mechanism of action and the metabolism process of SSs in vivo was still insufficient, and it needs to be strengthened in the future. There are many different kinds of SSs, and the contents of SSs in several of Bupleurum species are also different. Among them, SSA, SSC and SSD are the most common saponins, which have many related studies. But studies on other saponins with low content are still lacking.
Taken together, this review summarizes the structure and pharmacological action of SSs as the main active component of Bupleurum L. plants, which reflects the importance of SSs and provides a necessary direction for future research. In addition, this review also provides a reference for the research and development of new drugs with SSs.
Footnotes
Funding
This research was supported by the National Natural Science Foundation of China (No. 82160759, 82060792, 81660645, 81660673); Key R&D Program of Ningxia (2018BFG02005, 2021BEG03100); Chunhui Project of Ministry of Education (Z2016057); Natural Science Foundation of Ningxia (2020AAC03133, 2021AAC03143) and the Fourth Batch of Ningxia Youth Talents Supporting Program (grant numbers TJGC2019091, TJGC2019100).
Author Contributions
Y.Y. and J.L. conceived and designed the manuscript. A.J. and X.Y. wrote, revised the manuscript, and prepared Figure 1–9 and Table 1-9. A.J., X.Y., B.Z., J.L., Y.W. and R.M. collected and analyzed the literatures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
