Abstract
In an aging society, the prevalence of Alzheimer disease (AD) and type 2 diabetes (T2D) has increased. It is currently hypothesized that these diseases are caused by the aggregation of amyloid β (Aβ) in the brain and human islet amyloid polypeptide (hIAPP) in the islets of Langerhans, respectively. Therefore, the disaggregation of these existing amyloid aggregates is a promising approach to the prevention and treatment of both diseases. In our previous studies, we found a remarkable Aβ and hIAPP aggregation inhibitory activity of polyphenolic compounds containing catechol moieties. Compared to previous reports on their aggregation inhibitory activity, there are few on the disaggregation activity of polyphenolic compounds. Additionally, there are few findings on the disaggregation activity of polyphenolic compounds on hIAPP. In this study, we investigated the Aβ and hIAPP disaggregation activity of scirpusin B, a polyphenolic compound found in passion fruit seeds, and related compounds. Thioflavin T (Th-T) assays and transmission electron microscopy (TEM) were performed on these compounds to evaluate their Aβ42 and hIAPP disaggregation activities. The results showed that scirpusin B and its related compounds showed remarkable disaggregation activity. The structure–activity relationship of these compounds revealed that the presence of catechol moieties is important for this activity. This study also showed that polyphenols from passion fruit seeds have significant disaggregation activity against amyloid polypeptide aggregation.
Keywords
In an aging society, the increasing number of patients suffering from dementia has become a serious concern. Alzheimer disease (AD), an intractable neurodegenerative complaint, accounts for more than half of all dementia cases. Neurodegenerative diseases, such as AD, interfere with the daily activities of patients, who are in need of long-term care and become an increasing burden on their families. Although research is underway to develop a cure, no effective treatment nor drug has been found to fundamentally treat AD itself. Similar to AD, type 2 diabetes mellitus (T2D), which is on a rise across the globe, is also a major problem. T2D is characterized by decreased insulin function, which leads to high blood glucose levels and various complications. Previous studies have shown a relationship between AD and T2D. 1 AD and T2D share many common pathophysiological characteristics, such as increased oxidative stress and the aggregation of amyloid proteins with intermolecular β-sheet structures.2–4 Amyloid proteins include amyloid β (Aβ) and human islet amyloid polypeptide (hIAPP).5–7 Aβ, which consists of 36 to 43 amino acids, is produced from amyloid precursor protein in the brain, and hIAPP, which consists of 37 amino acids, is secreted from pancreatic β-cells. 8 These aggregates attack cells in various ways. 9 For example, they stimulate the production of cytotoxic molecules, such as nitric oxide, reactive oxygen species (ROS), and pro-inflammatory cytokines in glial cells, contributing significantly to neuronal damage and death.10,11 It has also been proposed that these aggregates load unfolded-protein response pathways, 12 which leads to cerebral and hippocampal atrophy in the brain and insulin deficiency in the pancreas. Furthermore, recent studies have shown that hIAPP is mixed with senile plaques, which are Aβ aggregates found specifically in the brains of patients with AD. 13 Additionally, Aβ has been found to aggregate in the pancreas of transgenic mice expressing both Aβ and hIAPP. 14 Therefore, the disaggregation of these toxic oligomeric and fibrillar species may prove important for the treatment of AD and T2D. However, there is no effective therapy that can reverse the formation of these aggregates. Finding a compound that can disaggregate both the amyloid proteins would therefore be an effective agent for the prevention and treatment of both diseases.
Recently, plant extracts, 15 and especially flavonoids, 16 have been reported to exhibit disaggregating activity. In addition, resveratrol and related compounds have been reported to inhibit the aggregation of Aβ protein and hIAPP, which has been attracting significant attention.17,18 However, there are no reports on resveratrol analogs. Nonetheless, it is important to investigate the disaggregation activities of both hIAPP and Aβ amyloid polypeptides.
In our previous study, we found that polyphenols derived from various natural products inhibit amyloid polypeptide aggregation.19–27 In this study, we evaluated the disaggregation activities of scirpusin B and its related compounds from passion fruit seeds against amyloid polypeptide aggregation and expounded a structure–activity relationship for these compounds.
Materials and Methods
Tested Compounds 1 –7
Trans-scirpusin B (
Thioflavin T (Th-T) Assay
The disaggregation abilities of Aβ42 and hIAPP were evaluated using the Th-T method developed by Naiki et al,
12
the procedure for which has been described previously.
28
Herein, Aβ42 was dissolved in 0.1% NH4OH or hIAPP (KareBay Biochem Inc) dissolved in a 250 mM solution of 1,1,1,3,3,3-hexafluoro-2-propanol (0.5% acetic acid aqueous solution). The amyloid polypeptide solution was then diluted 10-fold with 50 mM PBS (pH = 7.4) and incubated with or without compounds

Molecular structures of compounds
Transmission Electron Microscope (TEM) Observations
The procedure was performed as previously reported 28 but with slight modifications. First, after the Th-T assay, 5 µL of the amyloid polypeptide sample was spotted onto a glow-discharge carbon-coated Formvar grid, incubated for 2 min, then washed twice with 5 µL of distilled water. The resulting grid was negatively stained twice for 1 min each with 5 µL of 0.4% silicotungstic acid. After air-drying for 10 min, the samples were analyzed by transmission electron microscopy (TEM) (JEOL JEM-1400).
Results
Aβ42 Disaggregation Activity of Compounds 1 –7
Th-T fluorescence assays were performed on compounds
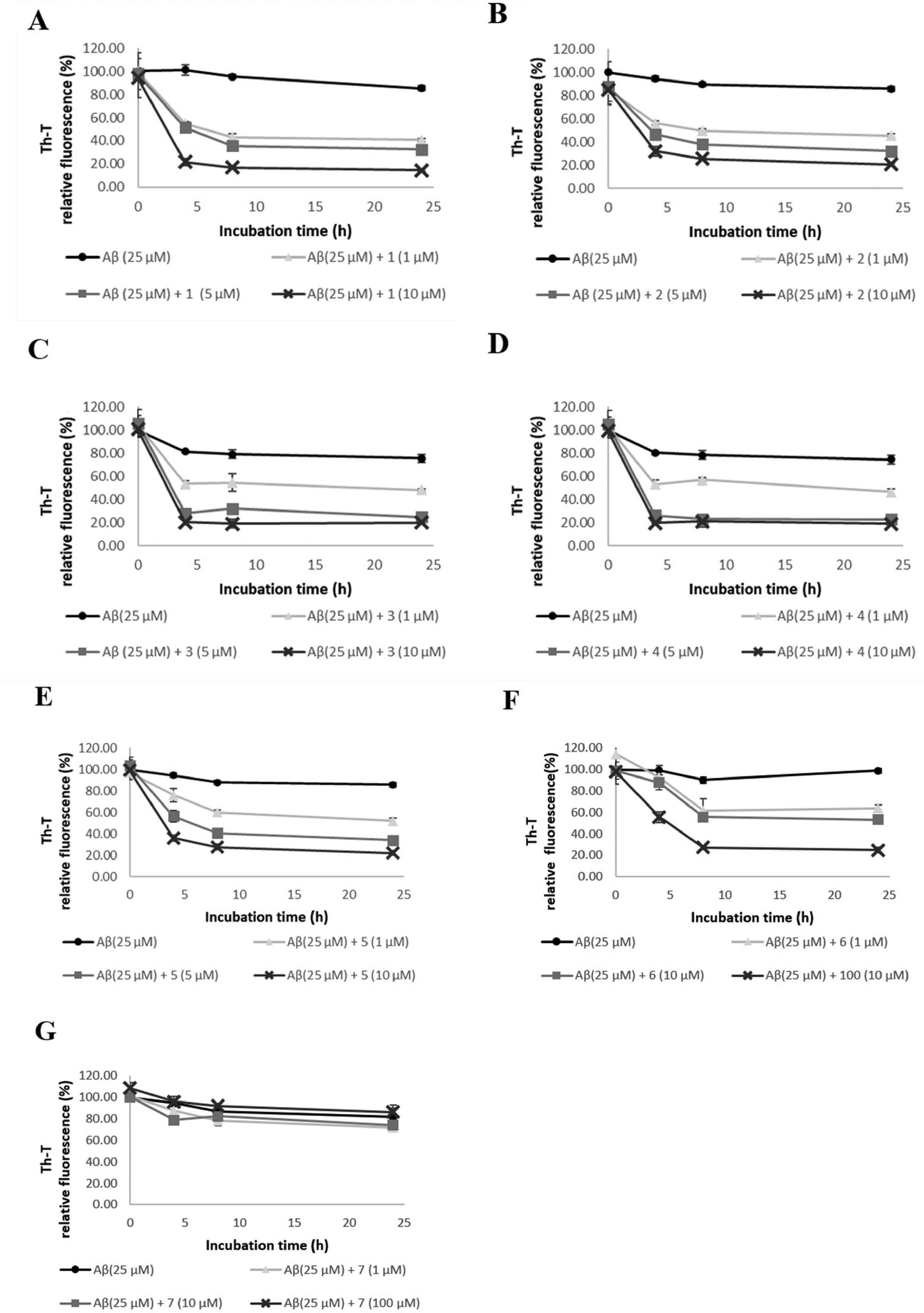
Effect of compounds
Evaluation of Aβ42 and hIAPP Disaggregation Activity of Compounds

IC50 values were calculated from the inhibitory rate (%) of each concentration of derivatives for amyloid polypeptide aggregation estimated using the Th-T assay after 24 h.
Th-T fluorescence intensity was suppressed in a concentration-dependent manner after treatment with each compound. Therefore, these compounds likely disaggregate the formation of the β-sheet structures associated with Aβ aggregation in a concentration-dependent manner. Compounds
To confirm this result, we used TEM to observe directly the aggregated form of Aβ42 (Figure 3). Conducting the experiment with only Aβ42 fibers showed that the Aβ42 fibers spread in a mesh pattern. A direct TEM observation revealed that the formation of Aβ fibers was most significantly disaggregated by compounds

Effects of compounds
hIAPP Disaggregation Activity of Compounds 1 –7
Th-T fluorescence assays were performed on compounds

Effect of compounds
To verify the effects of compounds

Effects of compounds
Discussion
In the present study, we investigated the effects of trans-scirpusin B (
In conclusion, the compounds with catechol moieties exhibited better disaggregation activity. The degree of activity was related to the number of catechol moieties present in the compounds, which suggests that catechol moieties affect Aβ and hIAPP structure. These results (Table 1) are consistent with our previous studies on aggregation inhibition.19–27 The following mechanisms were proposed based on the experimental results. First, the catechol moiety is autoxidized to o-benzoquinone. 29 The autoxidation of the catechol moiety to o-benzoquinone is thought to result in a Michael addition of basic amino acid residues to Aβ, in particular to the Lys16 and Lys28, resulting in structural changes of the protein.30,31 Furthermore, the benzene ring of the compounds employed in this experiment may cause protein conformational changes by inducing π-π stacking, according to the orientation of the amino acid residues of Aβ. 32 This π-π stacking may destabilize the fibrils by disrupting their β-sheet structure, which is thought to be necessary for aggregation. According to literature, Aβ42 and hIAPP are structurally similar 33 and may destabilize the fibril structure through a similar mechanism.
Second, small molecules such as brazilin 34 and polyphenols34–38 have also been reported to disaggregate amyloid fibrils into non-toxic amyloid aggregates. The exact mechanism is unclear, but it has been suggested that direct interaction of natural compounds with the fibril β-sheet is important for understanding the dissociation mechanism.32,39 Molecular dynamics simulations show that hydrogen bonds are formed with Asp23 within the fibrils and there is binding to the intermolecular Asp23-Lys28 salt bridge, which is important for stabilizing amyloid fibrils.40–42 This destabilizes the hydrogen bonds in the amino acid backbone that supports the fibrils, resulting in fibril remodeling and disaggregation. Amentoflavone-type bioflavonoids have been shown to bind preferentially to the N-terminus of fibrils via π-π interactions, resulting in the disaggregation of Aβ fibrils.43,44 The aromatic ring of the compound binds to the aromatic residues of the fibrils and fits into the N-terminus pocket, thereby stabilizing the biflavonoid-fibril complex. Hydrogen bonds are subsequently formed between the hydroxyl groups of the compound and the peptide backbone, significantly reducing the β-sheet content of the fibrils and altering the Aβ fibril conformation, leading to the fibril disaggregation. 42 Since the compound used in this experiment is a small molecule with a catechol moiety, a structure similar to that of brazilin, it may disaggregate via the mechanism described above.
We also compared the disaggregation and anti-aggregation activities of Aβ42 and hIAPP and found that Aβ42 and hIAPP showed similar behavior. These results support the results obtained in our previous studies,19–27 that is, polyphenols show comparable results in both Aβ42 and hIAPP disaggregation activity.
In the present study, Aβ42 and hIAPP disaggregation activity tests using trans-scirpusin B (
Footnotes
Acknowledgments
The authors thank Professor Kazuhiro Irie, Associate Professor Kazuma Murakami, Dr Mizuho Hanaki, and Dr Yumi Irie, Graduate School of Agriculture, Kyoto University for preparing Aβ42. The TEM observation method was used with a JEOL JEM-1400 at the Center for Medical Electron Microscopy, University of Tsukuba. The authors would like to thank Editage (![]() ) for English language editing.
) for English language editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (grant number JP20H05581).
