Abstract
Vascular endothelial growth factor (VEGF) and fibroblast growthfactor (FGF) play important roles in angiogenesis-related diseases. RC28-E is a soluble fusion protein composed of the human VEGF receptor 1 (VEGFR1) extracellular domain 2 (ECD 2), VEGFR2 ECD 3, FGFR1 ECDs 2 and 3, and the Fc regions of human immunoglobulin G1. By targeting both VEGF and FGF2, RC28-E may represent a useful antiangiogenetic agent, but structural and functional characterizations of this fusion protein are needed. Liquid chromatography–tandem mass spectrometry, size exclusion high-performance liquid chromatography, capillary electrophoresis-sodium dodecyl sulfate, imaged capillary isoelectric focusing, and bio-layer interferometry were used to characterize the properties of RC28-E. The purity of RC28-E was confirmed to be 98% or greater. The glycosylation modification of RC28-E was found to be very complicated, with 11 potential N-linked glycosylation points and 23 types of N-glycans, causing high heterogeneity of the protein. The primary modifications of the amino acid sequence of RC28-E protein included C-terminal K truncation, N-deamidation, and M-oxidation modification. Notably, RC28-E demonstrated a higher affinity for both VEGF and FGF2 than VEGF trap or FGF trap for their respective targets.
Introduction
The pathological growth of blood vessels is an important mechanism in oncogenesis and other angiogenesis-related diseases, such as age-related macular degeneration (AMD) and diabetic retinopathy (DR). AMD is considered as one of the 3 major causes of blindness, 1 and choroidal neovascularization (CNV) is the primary cause of AMD. 2 In the normal ocular vessel, angiogenic stimulators and inhibitors are in equilibrium. However, disturbance in this equilibrium results in the induction of neovascularization, a complex process regulated by at least 20 cytokines, including vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), platelet-derived growth factor (PDGF), and others.
Upon binding to its receptors present on vascular endothelial cells, VEGF plays a critical role in angiogenesis and vascular permeability. 3 VEGF receptors 1 and 2 (VEGFR1 and VEGFR2) have been recognized as high-affinity VEGFRs. 4 On their extracellular regions, both these receptors contain 7 immunoglobulin (Ig)-like domains. Studies have revealed that the second Ig-like domain of VEGFR1 and the third Ig-like domain of VEGFR2 are the key VEGF-binding domains. 5 Additionally, fibroblast growth factor 2 (FGF2) is considered a powerful growth factor for angiogenesis. It stimulates physiological and pathological neovascularization by binding to extracellular regions of the FGF receptor (FGFR). 6 Acting synergistically, FGF2 and VEGF stimulate vascularization, but with distinct effects on vessel functionality and the permeability of vascular endothelial cells. 7 Blockade of either of these growth factors leads to a decrease in the density of blood vessels. 7 Therefore, dual-target drugs that block both VEGF and FGF2 exhibit potential therapeutic effects in angiogenic diseases.
Recently, several VEGF antagonists have been developed for the treatment of wet AMD and tumors, including bevacizumab (Avastin), VEGF Trap, and conbercept. However, no FGF antagonist has yet been approved. Bevacizumab is a humanized monoclonal antibody against VEGF. It acts by preventing the binding of VEGF to VEGFR1 and VEGFR2 on the surface of endothelial cells, thus deactivating the biological activity of endogenous VEGF. Bevacizumab, the first VEGF inhibitor to be approved globally, has received market authorization from the US Food and Drug Administration (FDA) for the treatment of metastatic colon and rectal cancers. 8 VEGF Trap is an Fc fusion protein containing the human VEGFR1 extracellular domain 2 (ECD 2) and VEGFR2 ECD 3. 9 Compared to bevacizumab, VEGF Trap can bind to all exogenous VEGF and placenta growth factor with a stronger affinity. Additionally, VEGF Trap is capable of inhibiting tumor growth and the CNV more effectively than bevacizumab.9,10 Therefore, the US FDA approved VEGF Trap as a curative agent for patients with either metastatic colorectal cancer or wet AMD.11,12 Similarly, in China, conbercept, a VEGF ligand trap, 13 was approved for the treatment of wet AMD. FP-1039, an FGF ligand trap consisting of the ECD of the human FGFR1 α-IIIc isoform, is in the process of clinical evaluation in patients with advanced solid tumors.14-16
In this study, RC28-E (also known as eflimrufusp alfa), a recombinant fusion protein of VEGFR and FGFR, was developed and structurally characterized. RC28-E was expressed in the Chinese hamster ovary (CHO) cells by gene recombination technology. It is composed of human VEGFR1 ECD 2, VEGFR2 ECD 3, FGFR1 ECD 2 and 3, and the Fc portion of human IgG1 (Figure 1). The complex structure of RC28-E has a crucial effect on its biological activity. However, the dual-target design of RC28-E also brings challenges to the development of analytical techniques needed for its structural characterization and functional assessment. In this study, extensive experiments were carried out to characterize the structure of RC28-E using different analytical techniques.
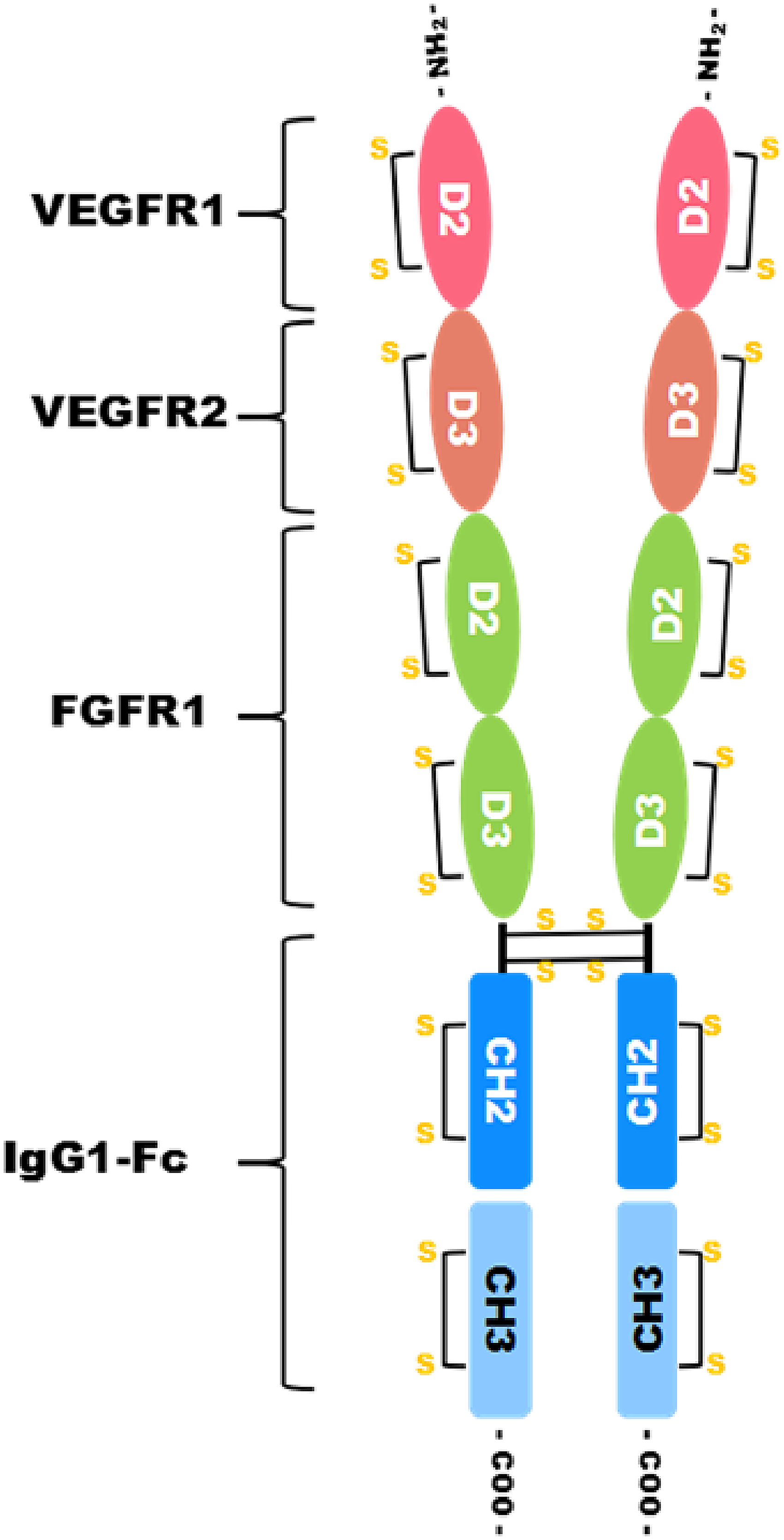
Schematic structure of RC28-E. RC28-E is composed of the human extracellular domain 2 (ECD 2) of VEGFR1, ECD 3 of VEGFR2, ECDs 2 and 3 of FGFR1, and the Fc portion of human immunoglobulin.
Materials and Methods
Reagents
Trypsin, chymotrypsin, and Glu-c protease were purchased from Promega, the SDS-MW kit from Beckman, Carrier Ampholytes (CAs) from Serva, Isoelectric Point (pI) Marker and methylcellulose (MC) from ProteinSimple, RapiGest SF and Glycoworks RapiFluor-MS kit from Waters, and trifluoroacetic acid, glacial acetic acid, methanol, and acetonitrile from Merck. VEGF and FGF2 were purchased from R&D Systems, and N-acetylneuraminic acid (Neu5Ac), N-glycolylneuraminic acid (Neu5Gc), 4,5-methylenedioxy-1,2-phenylenediamine dihydrochloride (DMB), iodoacetamide (IAM), β-mercaptoethanol (2-ME), and all the other chemicals were purchased from Sigma Chemical Co. The VEGF trap, FGF trap, and RC28-E used in this work were prepared by RemeGen Co., Ltd. Ultrapure water was obtained using a Millipore Milli-Q Gradient Water Purification System.
Size-Exclusion Chromatography High-Performance Liquid Chromatography (SEC-HPLC)
A SEC-HPLC system (Agilent) consisting of an Agilent 1260 separation module equipped with DAD detector was used in this study. RC28-E proteins were separated on a gel column (TOSOH TSK gel G3000SWXL, 7.8 × 300 mm, particle size 5 μm), for which the flow rate was set at 0.7 mL/min, and the detection absorbance was monitored at 280 nm. RC28-E proteins (20 μL, 100 μg) were loaded and then separated with buffer containing 25 mM phosphate and 0.3 M sodium chloride (pH 6.8) over a period of 20 min.
Capillary Electrophoresis-Sodium Dodecyl Sulfate (CE-SDS) Gel Analysis
Fifty µL of either a reductive (50 mM Tris-Hcl, 50 mM β-mercaptoethanol, 1.0% SDS; pH 9.0) or nonreductive buffer (5 mM citric acid, 20 mM disodium hydrogen phosphate, 1.0% SDS, and pH 6.5) was added to 40 μL of RC28-E sample (about 200 μg), followed by addition of 5 μL of alkylation reagent. Subsequently, the RC28-E protein solution was heated using a 65 °C water bath for 5 min, then cooled to room temperature, and centrifuged at 1000 rpm for 3 min. Finally, 70 μL of a processed RC28-E sample was used for analysis. A PA800 Plus CE system (Beckman Coulter) was used to analyze the purity of the protein. An uncoated capillary (50 µm inner diameter and 30.2 cm length) was used with an SDS-Gel MW Analysis Kit (Beckman Coulter). The capillary temperature was set to 25 °C, and the detection wavelength was 220 nm. The injection and separation conditions were as follows: 5 kV, positive polarity, and electric injection for 20 s, and 5 kV, positive polarity, and voltage separation for 30 min, respectively.
Imaged Capillary Isoelectric Focusing (iCIEF)
RC28-E samples (25 μL, 4 mg/mL), urea solution (99 μL, 8 M), CAs 5 to 9 (4 μL), pI 5.12 marker (1.5 μL), pI 8.18 marker (0.5 μL), and 1% MC (70 μL) were mixed evenly and centrifuged at 12000 rpm for 5 min. The supernatant (170 μL) was transferred into a vial for testing. The iCE3 system (ProteinSimple) was used to perform free solution IEF in a capillary column, and the focused protein zones were detected at 280 nm.17-19 The parameters of the iCE3 system were set at 1500 V, prefocusing for 1 min, and 3000 V, focusing for 6 min, and the temperatures of the capillary and sample trays were set at 4 to 15 °C.
N-Glycan Analysis by Ultra-High Performance Liquid Chromatography (UPLC)
The contents and types of N-glycan and sialic acid were analyzed by UPLC. 20 The UPLC system (Waters Corp) consisted of an H-Class separation module equipped with a column heating compartment, and a fluorescence (FLR) detector.
A Glycoworks RapiFluor kit (Waters Corp) was utilized for the analysis of N-glycans. The N-glycans of RC28-E protein were derived following enzymatic cleavage with Rapid PNGase F, and the free glycans were labeled using a RapiFluor-MS fluorescence reagent to produce glycan-containing fluorescence groups. The labeled glycans were then purified and enriched using a solid phase column. The chromatographic parameters included a Waters chromatographic column (ACQUITY UPLC BEH Glycan), dimensions of 2.1 × 150 mm, and particle size of 1.7 μm, a 10 μL injection volume, a column temperature of 60 °C, and a sample tray temperature of 4 °C. The excitation and emission wavelengths were set at 265 and 425 nm, respectively. The mixtures of N-glycans were separated by gradient elution with mobile phase A (100 mM ammonium formate, pH 4.5) and mobile phase B (acetonitrile). Gradient conditions were set at 0 to 46.5 min, 25% to 40% A; 46.5 to 47.0 min, 40% to 50% A; 47 to 49 min, 50% to 50% A; 49 to 50 min, 50% to 25% A; and 50 to 63 min, 25% A. The flow rate was set at 0.4 mL/min. Finally, the labeled N-glycans were detected by the FLR detector.
For analysis of sialic acid, the sialic acid was first released by acid-catalyzed hydrolysis and then reacted with DMB. The chromatographic parameters included a Waters chromatographic column (ACQUITY UPLC HSS C18), dimensions of 3.0 × 100 mm, a particle of size 1.8 μm, a 1 μL injection volume, a column temperature of 25 °C, and a sample tray temperature of 4 °C. The excitation and emission wavelengths were 373 and 448 nm, respectively. The mobile phase (7% methanol, 8% acetonitrile, and 85% ultrapure water) was maintained for 10 min, and the flow rate was set at 0.4 mL/min. Finally, the separated samples were identified by the FLR detector.
Liquid Chromatography Tandem-Mass Spectrometry (LC-MS/MS) Analysis
The N-glycosylation site,21,22 amino acid modification, and disulfide bonds of RC28-E were analyzed by LC-MS/MS (Q-TOF 6530, Agilent) using MassHunter B.06 software.
RC28-E was prepared for the analysis of glycosylation sites and amino acid modifications as follows: 100 μg RC28-E sample in 1% ammonium bicarbonate (ABC, pH7.4) buffer was treated by addition of guanidine hydrochloride (80 μL, 8 M) and DTT solution (3 μL, 1 M), followed by mixing at 56 °C for 40 min. Then, at room temperature, IAM solution (5.0 μL, 1 M) was added and protected from light for 35 min. Then, the pretreated sample and 1% ABC buffer (300 μL, pH7.4) were added to an ultrafiltration centrifuge tube and centrifuged at 12 000 rpm for 15 min, twice. Subsequently, trypsin (80 μL, 1:40) and chymotrypsin (80 μL, 1:40) were added, and the sample was placed in a 37 °C water bath for 4 h. Finally, formic acid (1 μL) was added to terminate the digestion reaction.
RC28-E was prepared for the characterization of disulfide bonds as follows: 100 μg RC28-E was deglycosylated with Rapid PNGase F and then treated with trypsin and Glu-c following the above method without addition of DTT and IAM.
In the experiment, an Agilent 1290 Infinity II separation module equipped with a DAD detector was used, and the chromatographic parameters included a Poroshell 120SB-C18 column, dimensions of 2.1 × 150 mm, a particle size of 2.7 μm, a 10 μL injection volume, a column temperature of 40 °C, and a sample tray temperature of 4 °C. The absorption wavelength was set at 214 nm. The samples were separated by gradient elution with buffer C (0.1% formic acid, 99.9% ultrapure water) and buffer D (0.1% formic acid, 99.9% acetonitrile), and gradient conditions were set at 0 to 60 min, 97% to 55% C, 3% to 45% D; 60.0 to 66.0 min, 55% to 10% C, 45% to 90% D. The flow rate was set to 0.3 mL/min.
The MS parameters were as follows: positive mode ESI, source voltage (Q-TOF) of 3.5 kV, drying gas flow rate of 10 L/min, a gas temperature of 325 °C, a sheath gas flow rate of 12 L/min, a sheath gas temperature of 380 °C, a nebulizer pressure of 35 psi, a Vcap of 3500 V, a nozzle voltage of 0 V, an MS range of m/z 250 to 1700, and an MS/MS range of m/z 50 to 2500. MS was performed in the data-dependent mode with dynamic exclusion enabled. The MS/MS scan was acquired on the 3 most abundant peaks in each full scan spectrum when the signal exceeded a predefined threshold.
Analysis of Binding Affinity
The affinities and binding kinetics of RC28-E, VEGF trap, and FGF trap were detected using bio-layer interferometry (BLI) (ForteBio Octet, PALL). The individual samples (50 nM RC28-E, VEGF trap or FGF trap) in phosphate-buffered saline (PBS, pH 7.4) were loaded on the SA sensors (PALL) for 900 s. After washing, the loaded samples on the sensors were reacted with different concentrations of either VEGF (500, 250, 125, or 62.5 nM) or FGF2 (400, 200, 100, or 50 nM) in PBS for 900 s. Finally, PBS was added as a dissociation solution, and the program was set at 3600 s for dissociation. During the reaction period, the kinetic association (ka), disassociation (kd), and affinities (KD, KD = kd/ka) were measured longitudinally using ForteBio Octet 9.0 software.
Results
Analysis of RC28-E Purity
The purity analysis of RC28-E was performed by SEC-HPLC, and the test results are depicted in Figure 2A and D. After the RC28-E samples were loaded, the polymer and monomer in the samples were eluted consecutively from the resins, and the impurity peak of the polymer and the main peak of the monomer were collected and analyzed. The monomer percentage in 3 batches of RC28-E samples was more than 98.0%, and the polymer percentage was less than 2.0%.

Purity analysis of RC28-E. (A) RC28-E protein was analyzed by size-exclusion high-performance liquid chromatography. (B) Nonreduced RC28-E protein was analyzed using a PA800 plus capillary electrophoresis system. (C) Reduced RC28-E protein was analyzed using a PA800 plus capillary electrophoresis system. (D) The purity of 3 batches of RC28-E.
The CE-SDS method was further adopted to determine the purity of RC28-E. With low viscosity, linear polymer gel solution as the sieving medium, CE-SDS could separate the target protein from other impurities in the sample. The CE-SDS analysis showed that the purity of RC28-E in 3 batches number was more than 99.0% under the nonreduced (Figure 2B and D) and the reduced condition (Figure 2C and D).
Charge Heterogeneity (Isoelectric Point) of RC28-E
Due to its high resolution and linearity over a wide pH range, ICIEF was used to measure the isoelectric point (pI) of RC28-E proteins. The isoforms with different charges were separated at their different pIs, and the focused bands were identified. The pI standards (pI 5.12, 8.18) were used to fit a calibration curve by linear regression to calculate the pI and the area percentage of each peak. As shown in Figure 3, the RC28-E protein exhibited a heterogeneous charge distribution with a main peak pI and pI range of 6.15 and 5.74 to 7.39, respectively. Seventeen protein variants were divided into 3 regions: main peak region, basic region (with pI greater than the main peak region), and acidic region (with pI less than the main peak region). The percentages of acidic, main, and basic peaks in the sample were 20.6%, 44.4%, and 35.0%, respectively.

Charge heterogeneity of RC28-E. The charge heterogeneity of RC28-E was analyzed by imaged capillary isoelectric focusing, and multiple peaks were observed for 3 batches of RC28-E with a pI range of 5.74 to 7.39. The pI of Marker 1 and Marker 2 were 5.12 and 8.18, respectively.
N-Glycosylation Analysis of RC28-E
Qualitative analysis of N-glycans in RC28-E was performed using UPLC. The distribution of N-glycans included enzyme digestion, labeling, derivatization, purification, and detection. Finally, the labeled N-glycan contents were accurately identified and quantified. As shown in Figure 4A and Table 1, more than 23 types of N-glycans were identified, accounting for 90% of all detected glycans. The main N-glycans in 3 batches of RC28-E consisted of 2-antennae sialic acid glycoforms (the relative content percentages of G2SA1, G2FSA1, G2SA2, G2FSA2 were 12.17 ± 0.61, 19.40 ± 0.30, 6.50 ± 0.20, 13.73 ± 0.15, respectively).
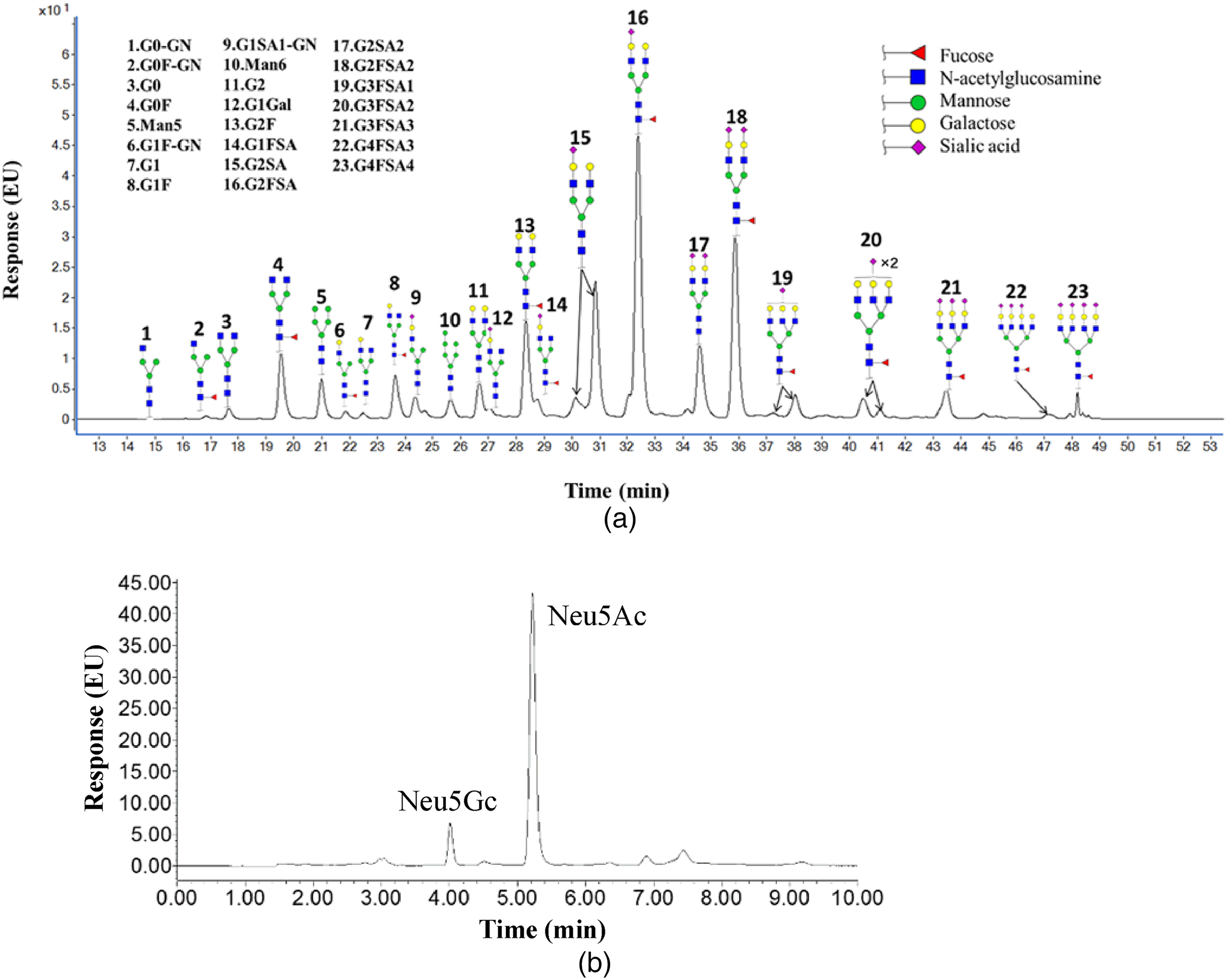
Glycosylation characterization of RC28-E. RC28-E protein was digested with Rapid PNGase F, and free glycans were labeled with RapiFluor-MS fluorescence reagent. The reactants were further analyzed by ultra-high-performance liquid chromatography. (A) The main types of N-glycans in RC28-E. (B) Types and contents of sialic acid in RC28-E.
N-Glycan Contents of 3 Batches of RC28-E.

N-glycosylation of proteins is well known to occur at asparagine residues that fall within the canonical sequence motif N-X-S/T (X≠P). By analyzing the theoretical amino acid sequence of RC28-E, it was concluded that the single-chain of RC28-E maybe contains 11 potential N-glycosylation sites. The glycosylation sites and corresponding glycoforms were determined by analyzing the fragmentation of the glycopeptides. Among the fragmentations in MS/MS, the CID-MS2 spectrum of glycopeptides was generated and consisted of not only B-type oxonium ions (m/z 204.1 for HexNAc + , m/z 366.1 for Hex-HexNAc + , etc) but also Y1 ions (the innermost GlcNAc + peptide backbone). With this information, the glycoforms of each N-glycosylated site could be identified. The glycoform percentage could be calculated from the peak intensities of the modified glycopeptides. All 11 potential N-glycosylation sites were found to be modified by N-glycans, and the main glycoforms of each N-glycosylated site are presented in Table 2.
N-Glycosylation Modification of RC28-E.

Sialic acid is a general term for derivatives of neuraminic acid. N-acetylneuraminic acid (Neu5Ac) and N-hydroxyacetylneuraminic acid (Neu5Gc) are the main derivatives. In this study, acid hydrolysis was used to release sialic acid from RC28-E protein. After derivatization with MDB, Neu5Ac and Neu5Gc were finally separated and quantified by using UPLC with a fluorescence detector (Figure 4B). The sialic acid content of RC28-E was relatively high, and the contents of Neu5Ac and Neu5Gc in 3 batches of RC28-E were found to be 18.35 to 20.53 and 0.38 to 0.50 mol/mol protein, respectively.
Amino Acid Modification Analysis of RC28-E
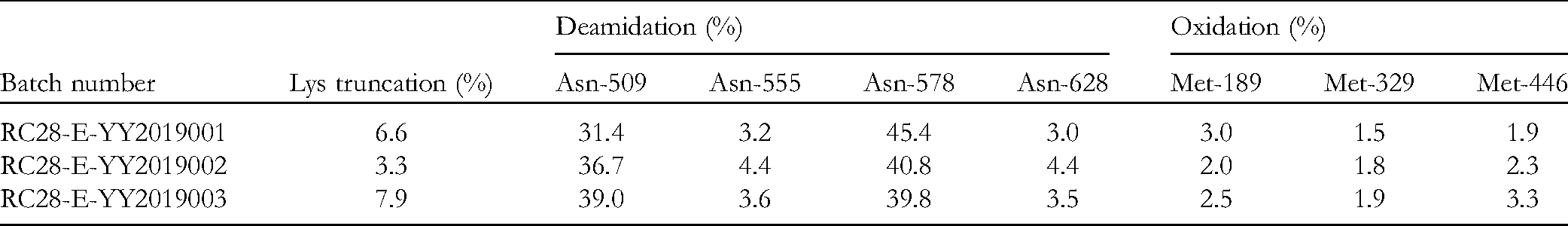
In IgG antibodies, lysine (Lys, K) is the main site causing C-terminal heterogeneity. As shown in Table 3, the C-terminal K truncation of different batches of RC28-E was less than 10%. In proteins, Asn and glutamine (Gln, Q) are common deamidation modification sites. For Asn, the major AA sequences that may cause deamidation are Asn-Gly, Asn-Ser, and Asn-Thr. The Asn was deamidated to generate Asp. Trypsin was used to digest the RC28-E protein, after which the deamidation sites of peptides were analyzed and identified by LC-MS. The characteristic peptides were studied using Mass Hunter B.06 software. With each deamidation of peptide, the molecular weight of the peptide increased by 1 Da. The findings revealed 4 deamidation sites from Asn at positions 509, 555, 578, and 628 of the RC28-E protein chain (Table 3). Methionine (Met) residues in proteins are common oxidation modification sites. With each oxidation of the peptide, the molecular weight of the peptide increases by 16 Da. Our findings revealed 3 oxidation sites from Met at positions 189, 329, and 446 of the RC28-E protein chain (Table 3).
Lysine Truncation at C-Terminus, Deamidation, and Oxidation of RC28-E.
Disulfide Bond Analysis of RC28-E
RC28-E contains 2 pairs of the same protein chains, which are connected by 12 pairs (6 types) of intrachain disulfide bonds and 2 pairs of interchain disulfide bonds. The primary structure of RC28-E with the theoretical disulfide bonds is shown in Figure 1.
After deglycosylated RC28-E was digested by trypsin and Glu-c, the disulfide-linked peptides were analyzed with LC-MS/MS and determined using Mass Hunter B.06 software. The tested peptide masses in the LC-MS chromatogram were matched with theoretical peptide masses. The matched peptide masses (with <5 ppm mass accuracy) were further confirmed by the corresponding CID-MS2 fragmentation. Thus, 6 types of intrachain disulfide bonds (C27-C76, C121-C182, C231-C283, C330-394, C455-C515, C561-C619) and 2 pairs of interchain disulfide bonds (C420-C420, C423-C423) were identified (Table 4).
Disulfide Bond Assignment of RC28-E.

Binding Affinity of RC28-E to VEGF/FGF2
The binding affinities of RC28-E to VEGF and FGF2 were evaluated dynamically with BLI, and the affinity constants are listed in Table 5. The affinity constants for VEGF trap binding to VEGF and for FGF trap binding to FGF2 were found to be 8.75 × 10−12 mol/L and 5.12 × 10−12 mol/L, respectively. Unlike VEGF trap and FGF trap, RC28-E could bind VEGF and FGF2 simultaneously, and the KD values were found to be less than 0.4 × 10−12 mol/L and 1 × 10−12 mol/L, respectively.
KD Values for Binding of Different Proteins to VEGF and FGF2.

Discussion
RC28-E is a recombinant Fc fusion protein with dual action against VEGF and FGF2. Previous studies showed that compared to blockage of VEGF or FGF2 alone, treatment with RC28-E could significantly suppress the formation of neovascular tufts. 23 Thus, RC28-E may represent a novel agent for the treatment of ocular neovascularization-related diseases. 23
The variety and complexity of recombinant proteins impose a great difficulty in protein quality control. 24 Additionally, an Fc fusion protein with a high molecular weight may result in a large amount of variation during production and storage processes and lead to heterogeneity in molecular sizes, electric charges, and glycosylation modifications. 25 Therefore, characterization of recombinant Fc fusion proteins plays a crucial role in supporting and guaranteeing the safety and efficacy of the product.
The molecular size variants of recombinant proteins include polymers, broken fragments, and disulfide bond isomers. 26 According to previous studies, the polymers may result in safety risks and affect the activity of protein products, while the fragments may affect the efficacy of protein products. Thus, both substance types should be controlled as critical quality attributes of protein products.27,28 In this study, a combination of SEC-HPLC and CE-SDS 29 was adopted to analyze systematically the purity of RC28-E protein. Under the reduced conditions, a certain amount of 2-ME as a reductant was added to RC28-E, which could break the intermolecular disulfide bond and convert the RC28-E protein to its single-strand form. Therefore, the reduced RC28-E had a shorter retention time than nonreduced RC28-E during CE-SDS analysis.
During the production of an Fc fusion protein using engineered cells, various posttranslational modifications, such as glycosylation, oxidation, deamidation, isomerization, and C-terminal K deletion, can easily happen, leading to the further heterogeneity of the therapeutic protein. During the production of monoclonal antibody (mAb) and Fc fusion proteins, glycosylation is one of the most common and complex modifications. Multiple studies have shown that the N-linked glycosylation modifications of proteins have certain impacts on their stability, antibody-dependent cellular cytotoxicity (ADCC), complement-dependent cytotoxicity (CDC), and immunogenicity.30-32 In this study, 11 potential N-linked glycosylation sites and more than 23 types of N-glycans were identified and quantitatively characterized within RC28-E. However, the effect of the different glycoforms on the biological function of RC28-E still needs to be verified in future studies, for example, the effect of mannose glycoforms for ADCC activity, and the effect of sialic acid modification for CDC activity.
The primary modifications of the AA sequence of RC28-E protein included C-terminal K truncation, N-deamidation, and M-oxidation. Theoretically, the C-terminal sequence of an Fc fusion protein is Pro-Gly-Lys. During the expression and production of an Fc fusion protein in CHO cells, incomplete excision of the terminal K of protein by endogenous carboxypeptidase can happen. According to previous research, C-terminal K truncation of mAb usually does not affect the function and pharmacokinetic properties of the protein. 33 Moreover, it is known that N-deamidated proteins tend to carry less positive charges with lower pI values, thus changing the protein properties. 34 Therefore, N-deamidation is the main source of acidic variants of proteins. In this study, the ratio of C-terminal K truncation, N-deamidation, and M-oxidation remained consistent for different batches of RC28-E, which ensured the consistency and stability of the product.
The affinity of RC28-E for VEGF and FGF2 affects the activity and efficacy of the drug. In this study, the reactions of RC28-E with VEGF and FGF2 were found to be slow dissociation reactions. RC28-E could simultaneously bind both VEGF and FGF2 and showed much greater affinity than either VEGF trap or FGF trap. Thus, RC28-E maybe a highly potent agent for the treatment of angiogenesis-related diseases.
Footnotes
Acknowledgments
The authors thank Wei Song and Yuhuan Zhong at RemeGen Co., Ltd, for input regarding analytical techniques.
Authors’ Note
Xuejing Yao and Guiping Qi authors contribute equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science and Technology Major Project of China (grant number 2013ZX09102040).
