Abstract
Introduction
Vitiligo is a long-term skin condition characterized by the loss of pigment due to either death or dysfunction of melanocytesthat affects approximately 1–2% of the population worldwide. Autoimmune processes, environmental and genetic factors, and oxidative stress, are the main factors related to its development. Although many studies have attempted to explain vitiligo's pathogenesis, these studies did not fully explain the clinical and experimental observations of the disease.
Melanin is produced by melanocytes through a process called melanogenesis, catalyzed by tyrosinase, tyrosinase-related protein 1 (TRP-1), and tyrosinase-related protein 2 (TRP-2). 1 These enzymes catalyze tyrosine hydroxylase to form 3,4-dihydroxyphenylalanine (DOPA), followed by DOPA oxidation to produce DOPA-quinone. TRP-1 and TRP-2 function in the biosynthesis of melanin downstream of TYR. TRP-2 catalyzes dopachrome rearrangement to form 5,6-dihydroxyindole-2-carboxylic acid (DHICA), 2 while TRP-1 oxidizes DHICA to produce carboxylate indolequinon. 3 The tyrosinase family genes (TYR, TRP-1, and TRP-2) are tightly regulated by a microphthalmia-associated transcription factor (MITF), an important transcription factor regulating TYR gene expression. 4
The regulation of tyrosinase activity has been shown to be crucial for treating hypermeter conditions, such as vitiligo. At present, many tyrosinase inhibitors have been applied in cosmetics. Vernonia anthelmintica (Linn.) Willd. (Compositae) is a plant that grows in the high altitude regions of southern Xinjiang and Pakistan, as well as in the plateaus of Pakistan and India. Itsfruit extract is commonly used for the treatment of vitiligo. It was originally used in Yao yong zong library, with about 300 companies.7,8 Chalcone is an essential natural product found in fruits, vegetables, spices, tea, and soybean that possess anti-tumor, anti-bacterial, anti-oxidant, anti-inflammatory, and anti-HIV activities. Many chalcones and their derivatives have been described as effective inhibitors of tyrosinase. 9 Our research group has been extensively studying tyrosinase-activated chalcone compounds, 12 and our results revealed that the chalcone derivatives with substituents on benzene ring may activated tyrosinase (Table 2). 15 In contrast, tyrosinase was slightly inhibited when the B-ring was introduced into the same group. 17 In this study, we further examined the effects of4-dimethylamino-4'-methoxy chalcone (DMC) (Table 1) on the expression of tyrosinase (TYR), tyrosinase-related protein-1 (TRP-1), tyrosinase-related protein-2 (TRP-2), and microphthalmia-associated transcription factor (MITF) in murine B16 cells.
Structure of Chalcones.

The Activator Effect of Substituted Chalcones on Tyrosinase. 17 .

50% effective concentration, result were the means ± SD from three independent experiment.
NA means slightly activated and EC50 > 200 m mol/L.
8-MOP as positive control.
Materials and Methods
L-3,4-dihydroxyphenylalanine (L-Dopa), dimethyl sulfoxide (DMSO), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazole ammonium bromide (MTT) were acquired from SIGMA (St. Louis, MO, USA), β-actin antibodies from Cell Signaling Technology (Danvers, MA, USA), horseradish peroxidase-conjugated secondary antibodies from GE Healthcare (Piscataway, NJ, USA), TYR, MITF, TRP-1, and TRP-2 antibodiesfrom Santa Cruz Biotechnology (Santa Cruz, CA, USA), an enhanced Bradford protein assay kit from Beijing Biomed Co. Ltd (Beijing, China), and phenylmethylsulfonyl fluoride (PMSF) and the components of the whole-cell lysis buffer for Western blotting analysis from SIGMA (St. Louis, MO, USA).
DMC Stock Solution
A stock solution of DMC (5 mg/mL) was prepared in DMSO for further application.
Cell Culture
The mouse B16 melanoma cell line was obtained from the Chinese Academy of Sciences. Cells were cultured in DMEM (Gibco, Life Technologies, USA) supplemented with 10% heat-inactivated fetal bovine serum (FBS) (Gibco), 100 U/mL penicillin, and 100 μg/mL streptomycin (Hyclone, USA) in a humidified atmosphere containing 5%CO2/95% air at 37 °C.
Cell Viability
MTT assay was used to detect cell viability. B16 cells were incubated in a 96-well culture dish at a density of 5 × 103 cells per well. After 24 hours, cells were exposed to different concentrations of DMC for 48 h. Subsequently, sterile MTT (5 mg/mL in PBS) was added to each well and incubated at 37 °C for 4 h. After removal of the medium, 150 μL of DMSO was added to each well, and the plate was gently shaken for 10 minutes. The absorbance at 570 nm was determined using a microplate reader. The absorption of untreated cells was considered as 100% cell survival rate. Each process consisted of five parts, and each experiment was repeated three times.
Western Blotting
B16 cells were inoculated in a 60 mm culture dish at a density of 1 × 106 cell per dish and treated with 20 μg/mL DMC for five time periods (0, 12, 24, 48, and 72 hours). The plates were washed twice with cold PBS in buffer solution [1 mM PMSF, 50 mM KCl, 1% NP-40, 25 mM HEPES (pH 7.8), 100 μg/mL leupeptin, 20 μg/mL aprotinin, 125 μM DTT, 1 mM Na3VO4]. A total of 30 μg of each protein sample was added to the sodium dodecyl sulfate (SDS) sample buffer. The protein was then separated by 10% polyacrylamide gel electrophoresis, and transferred to a polyvinylidene fluoride (PVDF) membrane; the membrane was sealed in 0.01 M Tris-HCl buffered saline (TBS) containing 2% BSA and 0.1% Tween 20 for 1 h. After washing three times with TBS containing 0.1% Tween 20 (TBST), the membrane was incubated overnight at 4 °C with either TYR, MITF, TRP-1, TRP-2 (dilution 1:200) or β-actin (dilution 1:1000) antibody in TBST containing 5% BSA. After washing three times with TBST and three times with TBS, samples were incubated with secondary antibody bound to horseradish peroxidase (dilution 1:10000) at room temperature for 1 h. After washing with TBST and TBS, the protein was visualized by ECL Western blotting detection reagent (GE Healthcare). Densitometric analysis was performed using Quantity One (Bio-Rad, Hercules, CA, USA). Western blotting assay results shown are representative of at least three independent experiments.
Statistical Analysis
All data were expressed as mean ± SEM. Statistical analysis was based on a single factor analysis of variance, followed by numerous comparative random tests. A p < 0.05 was considered to be statistically significant
Results and Discussion
Our previous results suggested that 0–40 μg/mL of DMCupregulates tyrosinase activity and enhances cellular melanin synthesis in B16 cells in a dose-dependent manner. 24 Thus, in this study, 20 μg/mLDMC was chosen to examine furtherthe effect of DMC on melanogenic protein expression. The results showed that 20 μg/mLDMCsignificantly increased TYR and MITF levels after 24 h of treatment (Figure 1). MITF has a vital role in melanogenesis as the central transcription regulator of TYR. 5 Moreover, DMC significantly increased the expression of TRP-1 and TRP-2 in B16 cells at 48 h. TRP-1 and TRP-2 are transmembrane proteins spanning melanosomal membranes (Figure 2). These proteins may synergistically act to modulate TYR activity. TRP-1 has been reported to influence TYR activity by forming a complex with it and/or stabilizing it. 18 On the other hand, TRP-2 functions as a dopachrome tautomerase downstream of TYR in the melanogenic pathway and affects the quantity and quality of melanin produced during melanin biosynthesis. 19

Effect of DMC on MITF and TYR protein levels in B16 cells.

Effect of DMC on TRP-1 and TRP-2 protein levels in B16 cells.
MITF gene locus encodes a transcription factor of the basic-helix-loop-helix-leucine zipper (bHLH-LZ) type, which belongs to a large family of bHLH factors. Together with TFE3, TFEB, and TFEC, MITF-M constitutes a small bHLH subfamily with high sequence similarity. CREB is another transcription factor that activates MITF-M expression. The αMSH hormone increases the cAMP level and activates protein kinase A (PKA), which results in increased MITF-M expression and, in turn, increased pigmentation. TGF-β can repress PKA activity and therefore reduce the CREB-dependent transcription of the MITF-M promoter. 20 CREB requires the coactivators p300/CBP. These proteins, which are histone acetyltransferases, interact with MITF-M and are believed to co-activate MITF-M target genes.21,22 Furthermore, the phosphorylation of serine 73 is required for MITF-M-p300 interaction, at least after Kit signaling. 23 In contrast, a MITF-M construct with mutation of this serine and the N-terminus truncated (including S73) was fully capable of activating promoter-reporter and the endogenous target tyrosinase. 24 In this study, we examined the expression of p-CREB after treatment with 20 μg/mLDMC for 0–6 h (Figure 3). We found that DMC significantly increased p-CREB levels 2 h after treatment (p < 0.01). The result showed that the regulation of MITF mediated by DMCwas positively correlated with the expression of p-CREB.

Effect of DMC on CREB and p-CREB protein levels in B16 cells.
The expression and function of MITF regulate several signaling pathways, including the MAPK and PKA signaling pathways. The MAPK family, including p38 MAPK, ERK, and JNK, has an essential role in regulating melanogenesis.25,26 Previous studies have suggested that the activation of the ERK and JNK/SAPK pathways is related to the down-regulation of melanogenesis. 27 Moreover, p38 MAPK in melanogenic differentiation has been reported in numerous studies. 28 Recent investigations suggested that p38 MAPK positively contributes to pigmentation. The activation of the p38 MAPK pathway induces MITF expression29,30 and promotes tyrosinase transcription.MITF is the substrate regulated by phosphorylated ERK1/2, which is involved in the differentiation, proliferation, and survival of melanocytes.31,32 ERK1/2, which are components of the Ras/Raf/Mek/ERK MAPK signaling pathway, are known to regulate cell survival, proliferation, and differentiation in response to extracellular stimuli.In the present study, we examined the expression of p-P38 and p-ERK1/2 after treatment with 20 μg/mLDMC for 0–6 h (Figures 4 and 5). Our results showed that DMC significantly increased p-P38 levels 1 h after treatment (p < 0.01) and increased the p-ERKlevels, which continued to increase 1h after treatment (p < 0.01).

Effect of DMC on P38 and p-P38 protein levels in B16 cells.

Effect of DMC on ERK and p-ERK protein levels in B16 cells.
β-Catenin is a multifunctional protein that is widely distributed and highly conserved during evolution. It is a crucial epithelial adhesion molecule and is a downstream signaling molecule in the Wnt pathway. 33 β-Catenin is involved in cell-cell adhesion, intracellular signaling, and gene transcription regulation, including development and the neural crest. Aftersuccessfully knocking-down β-catenin in neural crest stem cells, Hari 34 found that β-catenin is involved in the differentiation and migration of neural crest cells into melanin and has a role in the transport process of melanin. 35 MITF is an essential helix-loop-helix leucine zipper transcription factor involved in various cells’ development process, including melanocytes, that regulate the expression of β-catenin. It is also involved in the regulation of transcription of downstream genes 36 ; however, the detailed mechanism remains unclear. In this study, we examined the role of DMC on β-catenin protein expression in mouse B16 melanoma cells after 0–72 h treatment (Figure 6). The results showed that 24 h after DMC treatment, β-catenin protein levels were significantly enhanced and peaked at 48 h. Therefore, DMC can be expressed by β-catenin to regulate the expression of MITF and can work together with the regulation of transcription of downstream genes.

Effect of DMC on β-catenin protein levels in B16 cells.
During pigment cell development, both SOX-10 and MITF act autonomously and both proteins are normally expressed in pigment cell precursors. 37 This study showed that SOX-10 and MITF could rescue the respective mutant pigment cells after gene transfer and were functionally linked to a common pathway (Figure 7). Combined with other factors, SOX-10 regulates the expression of MITF through binding sites in the melanocyte-specific M promoter of MITF in vitro. 44 Interestingly, in zebrafish, SOX-10 is rapidly down-regulated in pigment cell precursors when they start migrating from the neural crest.39,48 In contrast, in mice, stable SOX-10 expression in developing melanocytes persists during the migration and homing of cells into the skin.49,50 In this study, the expression of SOX-10 after treatment with 20 μg/mL DMC for 0–72 h was examined. DMC could decrease SOX-10 level after treatment, indicating that SOX-10 did not participate in the DMC mediated enhancement of melanogenesis in B16 cells.
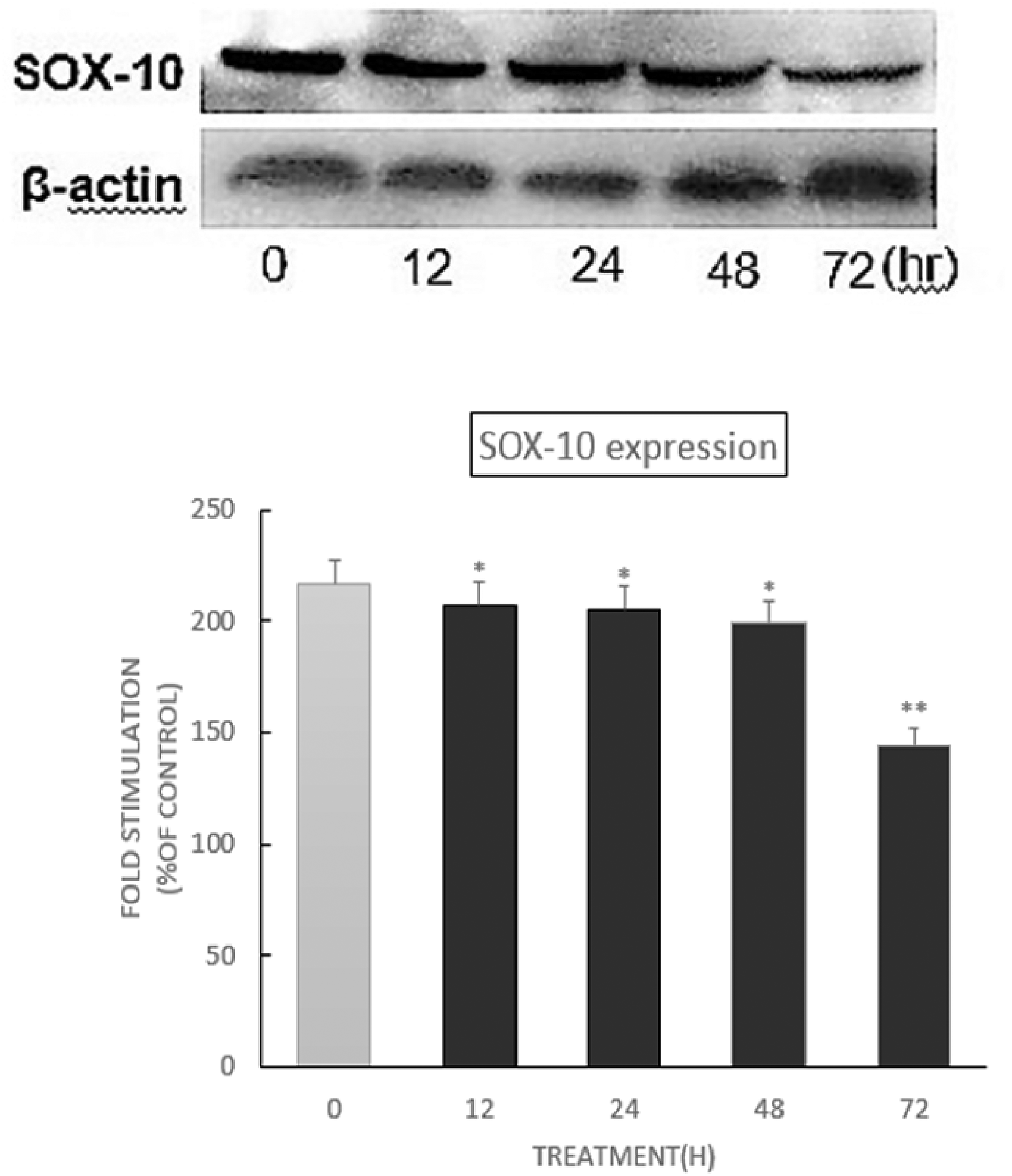
Effect of DMC on SOX-10 protein levels in B16 cells.
Conclusions
Tyrosinase is a research hotspot due to its important role in melanogenesis, which is involved in repigmentation in vitiligo (Figure 8). 51 In our study, DMC was synthesized and shown to be a potent agonist of tyrosinase, which may be further developed as a leading compound for treating vitiligo. DMC significantly enhanced tyrosinase activity and melanin amounts in B16 cells. 24 In addition to DMC’s regulatory effects on TYR, TRP-1, TRP-2 and MITF, we also investigated the signaling pathways involved in melanogenesis, examining the effects of DMC on CREB and p-CREB (AC/cAMP/PKA/CREB pathway), p38 MAPK, p-p38 MAPK, ERK and p-ERK (MAPK pathway), β-catenin (Wnt/β-catenin pathway) and SOX-10 (SWI/SNF pathway) protein expression levels by Western blot. As shown above, DMC can significantly enhance tyrosinase activity and promote melanin biosynthesis by up-regulating p-CREB, p-p38, p-ERK and β-catenin protein expression in B16 melanoma cells. Thus, this study suggests that DMC is a potent agonist of tyrosinase, which may be used for vitiligo treatment.

Melanin synthesis via multiple pathways. 51 .
Footnotes
Acknowledgments
This study was funded by the doctor's start-up Fund of Xinjiang Normal University [grant number XJNUBS1507]; Scientific Research Program in college universities of Xinjiang [grant number XJEDU2017S029]; Youth Innovation on Promotion Association, Chinese Academy ofScience (2019425); Tianshan Youth Program of XinjiangAutonomous Region in 2019 (2019Q028). Special Training Program of Natural Science Foundation of Xinjiang Autonomous Region in 2022.
Author Contributions
Adila Tuerxuntayi and Tajiguli Abulikeum performed experiments. Chao Niu designed the study and revised the paper. All authors read and reviewed the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The Youth Innovation on Promotion Association, Chinese Academy of Science, Special Training Program of Natural Science Foundation of Xinjiang Autonomous Region in 2022 (Chao Niu), West Light Foundation of the Chinese Academy of Sciences, Doctor's start-up Fund of Xinjiang Normal University, Tianshan Youth Program of Xinjiang Autonomous Region in 2019, Scientific Research Program in colleges universities of Xinjiang, (grant number 2019425, XJNUBS1507, 2019Q028, 2021-JCTD-001, XJEDU2017S029).
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
