Abstract
Background:
Juncus decipiens, a medicinal plant with a long history of traditional use, has been reported to possess diverse biological activities; however, its potential in regulating melanogenesis and protecting against ultraviolet (UV)-induced damage has not yet been explored.
Methods:
A bio-guided fractionation strategy targeting melanin inhibition was employed. Among the solvent-partitioned fractions, the ethyl acetate (EtOAc) fraction exhibited the most potent anti-melanogenic activity with minimal cytotoxicity in B16 melanoma cells. Further fractionation and purification led to the isolation of two compounds, structurally characterized as isovitexin and chlorogenic acid using high-resolution mass spectrometry (HR-MS) and nuclear magnetic resonance (NMR) analyses.
Results:
Isovitexin significantly reduced melanin content in a dose-dependent manner and inhibited intracellular tyrosinase activity, indicating its direct role in suppressing melanogenesis. Additionally, isovitexin enhanced cell viability under UVB-induced stress in normal human dermal fibroblasts, suggesting photoprotective potential.
Conclusion:
These findings represent the first report of the anti-melanogenic and photoprotective activities of compounds isolated from J. decipiens, supporting its value as a source of natural, multifunctional agents for skin health applications.
Introduction
Melanogenesis is a complex biological process that leads to the production of melanin, a pigment essential for protecting the skin against harmful ultraviolet (UV) radiation. 1 While melanin plays a vital photoprotective role, its overproduction can result in hyperpigmentation disorders such as melasma, age spots, and post-inflammatory hyperpigmentation, which are major cosmetic and dermatological concerns. 2 Tyrosinase, the key rate-limiting enzyme in the melanin biosynthesis pathway, catalyzes the initial steps of melanin formation. 3 Therefore, inhibiting tyrosinase activity is considered a primary strategy for controlling melanin synthesis and managing hyperpigmentation conditions.4,5
Beyond pigmentation disorders, prolonged exposure to UV radiation accelerates skin aging, induces inflammation, and damages dermal cells. 6 Consequently, effective skincare strategies should not only suppress melanogenesis but also offer protection against UV-induced cellular damage. This is particularly critical for maintaining the integrity of normal human dermal fibroblast (NHDF) cells, which play an essential role in skin structure, repair, and resilience. 7
UV radiation remains one of the primary external stressors affecting skin health, triggering reactive oxygen species (ROS) formation that not only cause DNA damage and inflammation, but also upregulate pigmentation through signaling cascades involving mitogen-activated protein kinase (MAPKs) (eg, p38, JNK, ERK), microphthalmia-associated transcription factor (MITF), and tyrosinase.6,8,9 ROS and UV exposure further exacerbate melanocyte activation and melanin synthesis, which, though protective in moderation, can lead to hyperpigmentation, photoaging, and uneven skin tone when uncontrolled. 10 Current therapies such as hydroquinone, kojic acid, and laser treatments can help reduce pigmentation but are frequently associated with drawbacks including skin irritation, long-term toxicity, risk of ochronosis, and inconsistent outcomes especially across different skin types.11,12 Natural flavonoid glycosides like vitexin have drawn recent attention because they can act on multiple pathways: scavenging ROS, suppressing pro-inflammatory cytokines Interleukin (eg, IL-1β, IL-17A), activating antioxidant defenses via nuclear factor erythroid, antioxidant response element (Nrf2/ARE), and modulating MAPK/MITF/tyrosinase axis. For instance, vitexin has been shown to protect melanocytes from hydrogen peroxide-induced oxidative stress by upregulating heme oxygenase (HO-1) and superoxide dismutase (SOD), enhancing cell survival, and reducing ROS levels, all while maintaining low cytotoxicity.13–15 Given these multi-target advantages, investigating vitexin and isovitexin from Juncus decipiens offers promising potential for safer and more effective treatments of hyperpigmentation and photo-damage.
In recent years, natural products especially those derived from plants have attracted growing interest as safe and effective tyrosinase inhibitors.16,17 Among these, flavonoids have emerged as promising candidates due to their wide-ranging biological activities, including antioxidant, anti-inflammatory, and anti-melanogenic effects.18,19 Their multifunctional nature makes them ideal for addressing both pigmentation and photodamage, key targets in modern skin care and therapeutic development.
Juncus decipiens (Buchenau) Nakai, commonly known as common rush (synonym: J. effusus L. var. decipiens), is a perennial herb widely found in wetland areas and traditionally used in herbal medicine. Previous phytochemical studies have identified several bioactive flavonoids in J. decipiens, suggesting its potential for therapeutic applications.20,21 However, despite its traditional use and phytochemical richness, no studies to date have explored the anti-melanogenic or photoprotective properties of this species.
In the present research, we highlight the isolation of the flavonoid isovitexin first time from J. decipiens. We also investigated its effects on production of melanin and cellular tyrosinase activity, making this the first report on the cellular mechanism of isovitexin in melanogenesis. This study not only exposes J. decipiens’ skin-lightening and photoprotective properties for the first time, but it also provides unique mechanistic insights into isovitexin's bioactivity.
Material and Methods
Plant Material
The J. decipiens stems and roots were provided in July and August 2022 by Kumamoto Prefectural Agricultural Research Center Agricultural System Research Institute, Yatsushiro, Japan. Samples were received at the Systematic Forest and Forest Products Sciences Laboratory, Faculty of Agriculture, Kyushu University, Japan.
Extraction and Fractionation
To investigate the anti-melanogenesis activity, stem, and root parts of J. decipiens were collected, dried at room temperature for two weeks, and refined into a fine powder with a mixer. A total of 3.0 kg of both stem and root parts powdered material was extracted three times with 100% methanol, each lasting five days at room temperature. The stem and root extracts were then concentrated under decreased pressure, yielding a crude methanol extract (323 g and 270 g) respectively.
For preliminary activity testing, the crude extract (200 g) was partitioned sequentially with n-hexane(n-hex), dichloromethane (DCM), ethyl acetate (EtOAc), and water, providing 87.1 g, 15.3 g, 15.1 g, and 78.5 g of the corresponding fractions.
Each fraction was tested for melanogenesis inhibiting activity in cell-based assays. The EtOAc fraction had the highest activity, followed by the DCM fraction, with the aqueous and n-hexane fractions being less active. Further comparative testing of the stem and root methanol extracts revealed that the stem extract was more active, thus it was prioritized for chemical isolation.
The EtOAc fraction was subjected to medium-pressure liquid chromatography (MPLC) using a pure C-850 silica gel column (Buchi, Switzerland) coupled with a flash chromatography system (FP ID HP Si 24 g column, GL Sciences, Tokyo, Japan). MPLC was operated in preparative mode with dual detectors: UV (at 254, 265, 280, and 320 nm, threshold 0.05 AU) and evaporative light scattering detector ELSD (threshold 20 mV). A gradient elution was applied using DCM (solvent A) and methanol (solvent B) with the following gradient: 0% B (10 min), 5% B (10 min), 10% B (10 min), 15% B (10 min), 20% B (10 min), 25% B (10 min), 30% B (10 min), 35% B (8 min), 40% B (8 min), 50% B (2 min), and 100% B (10 min), yielding 29 subfractions pooled together to make 7 subfractions. Bioactivity screening of EtOAc subfractions showed that fractions 13-18 exhibited the most potent anti-melanogenesis activity. Subsequent purification using recycle HPLC led to the isolation of Isovitexin (28.4 mg) from these active subfractions. Building up on our previous LC-QTOF-MS study, we identified chlorogenic acid as one of the major constituents of J. decipiens, showing strong anti-melanogenic activity. The DCM fraction although not subjected to full-scale bioactivity-guided sub fractionation was separated into 21 peaks and pooled into 6 subfractions (fr1-4, fr5-10, fr11-13, fr14-16, fr17-18, fr19-21). From fr14-16, chlorogenic acid (4.8 mg) was isolated via chromatographic purification, supporting its role as a key bioactive compound contributing to the observed melanogenesis inhibition.
Cell Viability
The cell viability was analyzed using a well-known dye [3,4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)]. In 24-well plates, 1 × 105 B16 melanoma4A5 cells (RIKEN BRC, RCB 0557, Ibaraki, Japan) were cultured for 24 h at 37 °C with 5% CO2 in 95% humidified air. After aspirating the supernatant, the cells were given two phosphate-buffered saline (PBS) washes. After that, 1.0 mL of new medium was combined with 50 μL of MTT reagent in PBS (5 mg/mL) and applied to each well. After that, the plates were incubated for four hours at 37 °C in a humidified atmosphere with 5% CO2. Following the removal of the supernatant, 1.0 mL of isopropyl alcohol containing 40 mM HCl was added to each well. For four hours, the plates were kept in the dark and at room temperature. Lastly, the microplate reader (Biotek, Wiooski,VT, USA) was used to measure the absorbance at 570 nm. 22
Melanin Assay
B16 melanoma cells were incubated in 24 well plate at a density of 0.5 × 105 cells/well. After 24 h, samples or solvent were added (water or DMSO) to the cells. Arbutin was used as a positive control. The cells were incubated for 48 h. After incubation, the medium was replaced to trypsin ethylenediaminetetraacetic acid (EDTA) solution to collect the cells into the tubes. The cells were washed with medium and PBS. After that, dissolved with 1N NaOH and incubated at 100 °C for 10 min and measured the absorbance at 450 nm wavelength with the microplate reader (Biotek, Wiooski,VT, USA).
Cellular Tyrosinase Assay
B16 cells were seeded in a 24 well plate at a density of 1 × 105 cells/well for 24 h before being pretreated with isovitexin like the melanin assay. The B16 cells were treated with the compounds for 48 h, then trypsin was used to extract the cells followed by two PBS rinses and a 10-min centrifugation at 1000 g at 4 °C. The cells were ultrasonically sonicated for one minute in ice water after being lysed with 0.5% Triton® X-100 and 1 mM phenylmethanesulfonyl fluoride (PMSF) in PBS. They were then placed in a freezer set at −80 °C for 10 min. The lysates were then centrifuged at 1000 g (4 °C) for 15 min then, 90 μL of the supernatant was combined with 10 μL of L-DOPA (10 mM in PBS) in a 96-well plate and incubated for 90 min at 37 °C. A microplate reader (Biotek) was used to measure optical density at 475 nm to assess dopachrome production.
UVB-Induced NHDF Cells Damaged Assay
NHDF cells were seeded into a 96-well plate at a density of 1 × 105 cells/mL and incubated for 24 h at 37 °C in a humidified atmosphere with 5% CO2. Following incubation, fresh serum-free media was supplemented with the isovitexin at different concentrations (50, 100, and 200 μM) and dimethyl sulfoxide (DMSO) (as a negative control) and incubated for an additional 24 h. The cells were washed twice with PBS. 100 μL of PBS was then added to every well. After that, NHDF cells were exposed to UVB radiation at a dose of 30 mJ/cm2 using a fluorescent lamp (Bio-Link Crosslinker, Vilber Loubert BiolinkTM BLX UVB, Cedex, France) that generated its maximum energy output at 312 nm as previously described method. 7 After then, the supernatant was switched out for new serum-free media and incubated for a further 24 h. Cell viability was then assessed using the MTT assay.
Statistical Analysis
GraphPad Prism 10 and Microsoft Excel 2023 were used to analyze all the data. The mean values ± standard deviation (SD) of a minimum of three independent experiments (n = 4) are displayed as the results. One-way ANOVA and Tukey were used to analyze group differences. For P ≤ .05, differences were considered statistically significant.
Results and Discussion
Modulation of Melanogenesis and Cell Viability of J. decipiens different fractions
The total extract (T.E) and fractions of J. decipiens at a concentration of 200 µg/mL was examined for cell viability and melanin content in vitro to find possible depigmenting fractions with minimum cytotoxicity. All examined fractions had reasonably high cell viability (>90%), indicating low cytotoxicity across samples (as shown in Figure 1A) and having no significant effect on cell survival, indicating their safety for further consideration in skin-related applications.

(A) Cell Viability of Total Extract (T.E) and Solvent-Partitioned Fractions ((n-Hexane (n-Hex), Dichloromethane (DCM), Ethyl Acetate (EtOAc), Aqueous Fraction (Aqu.fr)) at 200 µg/mL. Cell Viability Was Assessed Using a Standard Assay and Expressed as a Percentage of the Untreated Control. (B) Melanin Content (%) in Cells Treated with the Same Fractions at 200 µg/mL. The EtOAc and DCM Fractions Showed Significant Melanin Inhibition, While Maintaining Cell Viability Above 90%. Data Are Presented as Mean ± SD (n = 4). Different Letters Above the Bars Indicate Statistically Significant Differences among Groups at P < .05, as Determined by One-Way ANOVA Followed by Tukey's Multiple Comparison Test.
Regarding melanin synthesis, significant variation among the fractions was observed. The EtOAc fraction had the highest anti-melanogenic activity, lowering melanin concentration to around 50% of the control level with high significant differences and maintaining cell viability over 95% as shown in Figure 1A and B. The DCM fraction also reduced melanin formation considerably, indicating the existence of active depigmenting compounds in these semi-polar fractions. On the other hand, the T.E, n-hexane, and aqueous fractions showed no statistically significant reduction in melanin levels, indicating that there was little or no bioactivity in terms of inhibiting melanogenesis.
The EtOAc fraction emerged as the most promising choice because of its potent melanin inhibiting activity and low cytotoxicity. This shows the existence of bioactive secondary metabolites, such as polyphenols and flavonoids, that have been shown to inhibit tyrosinase or melanogenesis. The DCM fraction also requires further investigation due to its significance, and relatively modest activity, as shown in Figure 1B. These findings support the use of polarity-guided fractionation to isolate potential skin-lightening agents and provide the groundwork for future compound purification and mechanistic research.
Following the promising anti-melanogenic activity observed in the EtOAc fraction, further fractionation was carried out, yielding 29 subfractions (Fr. 1 to Fr. 29). Based on TLC profiling, several of these were pooled to obtain seven main subfractions: Fr. 1-5, Fr. 6-8, Fr. 9-12, Fr. 13-18, Fr. 19-22, Fr. 23-28, and the final end fraction. These pooled subfractions were evaluated at a concentration of 200 µg/mL for their effects on cell viability and melanin production. Notably, all seven subfractions exhibited minimal cytotoxicity, maintaining cell viability above 90% comparable to the untreated control group (Figure 2A) demonstrating their safety for further biological evaluation.

Effect of Solvent-Partitioned Fractions of Juncus decipiens on (a) Cell Viability and (b) Melanin Content in B16 Melanoma Cells. Cells Were Treated with Different Fractions at a Concentration of 200 μg/mL for 48 h, and Cell Viability and Melanin Content Were Measured. Data Are Presented as Mean ± SD (n = 4). Different Letters Above the Bars Indicate Statistically Significant Differences among Groups at P < .05, as Determined by One-Way ANOVA Followed by Tukey's Multiple Comparison Test.
Melanin inhibition was most significant in sub-fractions fr.9-12 and fr.13-18, which reduced melanin content to approximately 80% and 65% of the control, respectively, with high statistical significance. Moderate inhibition was also observed in Fr.6-8 and Fr.19-22, whereas Fr.1-5 and Fr.23-28 showed no significant activity. The most promising sub-fraction was fr.13-18, which demonstrated strong anti-melanogenic activity while maintaining minimal cytotoxicity (Figure 2B). These findings suggest the presence of concentrated bioactive constituents likely polyphenolic or flavonoid compounds previously enriched in the EtOAc fraction. The combination of melanogenesis inhibition and minimal cytotoxicity highlights these sub-fractions as promising candidates for subsequent purification and structural elucidation of active constituents.
Identification of Compounds
Following fractionation, and purification of sub-fraction fr.13-18, which demonstrated the most potent melanin inhibiting activity with minimal cytotoxicity, was further subjected to identification steps. Compound

Structures of the Identified Isovitexin (1) and Chlorogenic Acid (2).
In parallel with the isolation of isovitexin from the EtOAc fraction, the DCM fraction was also subjected for further fractionation to target major peaks. Compound 2 was obtained from DCM subfraction 14-16 as a brown amorphous solid. The molecular ion peak at m/z 354.0884 [M–H]– observed in ESI-MS (Figure S8) confirmed a molecular formula of C16H18O9 as previously reported. 33 The 1H NMR spectrum (600 MHz, CD3OD) exhibited resonances characteristic of a trans-caffeoyl moiety, including δ 7.54 (1H, d, J = 15.9 Hz, H-7′), 6.24 (1H, d, J = 15.9 Hz, H-8′), 7.03 (1H, d, J = 2.0 Hz, H-2′), 6.93 (1H, dd, J = 2.0, 8.2 Hz, H-6′), and 6.76 (1H, d, J = 8.2 Hz, H-5′). Signals corresponding to quinic acid core were observed at δ 5.31 (1H, td, J = 9.1, 4.4 Hz, H-3), 4.15 (1H, m, H-5), 3.71 (1H, dd, J = 8.4, 3.2 Hz,H-4), 2.18 (2H, m, H-6), and 2.04 (2H, m, H-2). The 13C NMR spectrum (150 MHz, CD3OD) showed 16 carbon signals, including δ 175.7 and 167.3 (C-7 and C-9′, carboxylic and ester carbons), aromatic carbons of the caffeoyl ring at δ 148.3 (C-4′), 145.8 (C-7′), 145.9 (C-3′), 126.5 (C-1′), 121.7 (C-6′), 115.1 (C-5′), 113.9 (C-2′), and 113.8 (C-8′), and oxygenated aliphatic carbons from the quinic acid ring at δ 74.9 (C-1), 72.2 (C-4), 70.6 (C-5), and 69.8 (C-3), with methylene carbons at δ 37.4 (C-2) and 36.8 (C-6) (Table S2, Figures S5 and S6). Based on these data and comparison with literature values, Compound 2 was identified as chlorogenic acid (5-O-caffeoylquinic acid) (Figure 3). 34
Chlorogenic acid has been extensively studied for its antioxidative, 35 anti-inflammatory, 36 antidiabetic, 37 and hepatoprotective 38 properties. In the context of skin biology, it has demonstrated the ability to inhibit tyrosinase activity, 39 reduce melanin production in murine B16 melanoma cells, 40 and suppress pigmentation in zebrafish embryos. 41 These earlier findings suggest that chlorogenic acid contribute to the depigmenting effects observed in the DCM fraction during the initial screening. Mechanistically, its previously documented actions including downregulation of MITF, and reduction of intracellular ROS indicate potential involvement in melanogenesis regulation. 42 Furthermore, chlorogenic acid has been reported to protect against UVB-induced skin damage by scavenging free radicals and enhancing cellular antioxidant defenses. 43 As a well-known polyphenol with enzyme-inhibitory, antioxidant, and photoprotective properties, chlorogenic acid represents a promising candidate for managing hyperpigmentation. Its presence further underscores the chemical and functional diversity of J. decipiens and reinforces the plant's potential as a valuable source of multifunctional skin-active compounds.
Anti-Melanogenesis Activity of Isovitexin
The EtOAc fraction of J. decipiens exhibited the strongest melanin-inhibitory activity with minimal cytotoxicity. Among its subfractions, Fr. 13-18 showed the potent efficacy in suppressing melanogenesis while maintaining cell viability (Figure 4A), leading to the isolation and structural identification of isovitexin, a C-glycosylated flavone. Its structure was confirmed by HR-MS and detailed NMR analysis.

Melanin Content in Cells Treated with Isovitexin and Arbutin. (A) Cell Viability of Isovitexin. Cells Were Treated with Isovitexin (Isovit) and Arbutin (Arb) at Concentrations of 50, 100, and 200 µM. (B) Melanin Content Was Measured and Expressed as a Percentage Relative to the Untreated Control. Data Are Presented as Mean ± SD (n = 4). Different Letters Indicate Statistically Significant Differences Between Groups (P < .05, One-Way ANOVA Followed by Tukey's Post Hoc Test).
Isovitexin significantly inhibited melanin production in a concentration-dependent manner (50-200 µM), reducing melanin content to approximately 70% and 40% of control levels at 100 µM and 200 µM, respectively as shown in Figure 4B. These effects were comparable to those of arbutin, a standard skin-lightening agent, which followed a similar dose-response trend.
These results indicate that isovitexin is a key active compound in J. decipiens with strong anti-melanogenic potential and low cytotoxicity. Its C-glycosidic structure may offer enhanced stability and bioactivity over O-glycosides, 44 making it a promising candidate for further development in cosmetic and dermatological applications targeting hyperpigmentation.
Cellular Tyrosinase Activity
To elucidate the mechanism of isovitexin's anti-melanogenic activity, its effect on cellular tyrosinase activity was evaluated at concentrations of 50, 100, and 200 µM. Isovitexin significantly reduced tyrosinase activity in a dose-dependent manner. While no notable change was observed at 50 µM, a significant reduction was detected at 100 µM, and a marked decrease to approximately 32% of control levels was observed at 200 µM as shown in Figure 5. These findings demonstrate that isovitexin effectively suppresses melanin synthesis by inhibiting tyrosinase activity, a key regulatory enzyme in the melanogenesis pathway. This supports its role as a potential natural agent for managing hyperpigmentation.

Effect of Isovitexin on Cellular Tyrosinase Activity. Cells Were Treated with Isovitexin (Isovit) and Arbutin (Arb) at Concentrations of 50, 100, and 200 µM. Cellular Tyrosinase Activity Was Measured and Expressed as a Percentage Relative to the Untreated Control. Arbutin Was Used as a Positive Control. Data Are Presented as Mean ± SD (n = 4). Different Letters Indicate Statistically Significant Differences Between Groups (P < .05, One-Way ANOVA Followed by Tukey's Post Hoc Test).
Photoprotection Assay
The effect of isovitexin on cell viability was assessed under both normal and UVB-induced stress conditions at concentrations of 50, 100, and 200 µM. Under normal conditions, isovitexin enhanced cell viability in a concentration-dependent manner, with 100 µM and 200 µM showing significant increases above 120% compared to the DMSO control (P < .05). A moderate, non-significant increase was observed at 50 µM. Following UVB irradiation, cell viability dropped to approximately 55% in the UVB control group, confirming UVB-induced cytotoxicity. Isovitexin treatment significantly increased the cell viability at all tested concentrations. The 200 µM dose showed the strongest protective effect, restoring viability to nearly 100%, comparable to the DMSO control and significantly higher than 50 and 100 µM (P < .05). Alphabetical groupings (a–e) indicate statistically significant differences. The UVB control (e) was significantly lower than all others, while the 200 µM post-UVB group (b) was comparable to the DMSO control and non-UVB isovitexin groups (a) (Figure 6), suggesting strong cytoprotective effects at higher doses.

Effect of Isovitexin on Cell Viability in the Presence and Absence of UVB Exposure. Cells Were Treated with Isovitexin at Concentrations of 50, 100, and 200 µM for 24 Hours, with or Without Subsequent UVB Irradiation. Cell Viability Was Assessed Using a Standard Assay and Expressed as a Percentage Relative to the DMSO Control. Data Are Presented as Mean ± SD (n = 4). Different Letters Indicate Statistically Significant Differences Between Groups (P < .05, One-Way ANOVA Followed by Tukey's Post Hoc Test).
These results demonstrate that isovitexin enhances baseline cell viability and effectively protects against UVB-induced cytotoxicity, likely through antioxidant or anti-inflammatory mechanisms, warranting further investigation.
The EtOAc fraction of Juncus decipiens exhibited the strongest anti-melanogenic activity in our fraction screening, reducing melanin content to nearly 50% of control levels while maintaining high cell viability. Bio-guided fractionation of this fraction led to the isolation of isovitexin as the major active compound, while chlorogenic acid was isolated from the DCM fraction. Both compounds have been previously reported to possess antioxidant and enzyme-modulating properties, and their isolation in this study provides new insight into the bioactive constituents responsible for the observed activities.25,35
Melanogenesis is tightly regulated at the transcriptional and enzymatic levels, and our findings align with this regulatory framework. MITF functions as the master regulator of melanocyte differentiation and pigmentation by binding to the promoters of key melanogenic genes, including tyrosinase, tyrosinase-related protein 1 (TRP-1), and tyrosinase-related protein 2 (TRP-2). Among these, tyrosinase is the rate-limiting enzyme that catalyzes the hydroxylation of tyrosine to DOPA and its subsequent oxidation to dopaquinone, thereby initiating melanin biosynthesis. 1 Thus, measuring tyrosinase activity alongside melanin content provides a mechanistic rationale for interpreting how isovitexin and chlorogenic acid exert their anti-melanogenic effects. By reducing tyrosinase activity and melanin production while maintaining cell viability, both compounds appear to target upstream MITF-regulated signaling as shown in Figure 7, consistent with previously reported mechanisms of flavonoid-mediated pigmentation control. 42
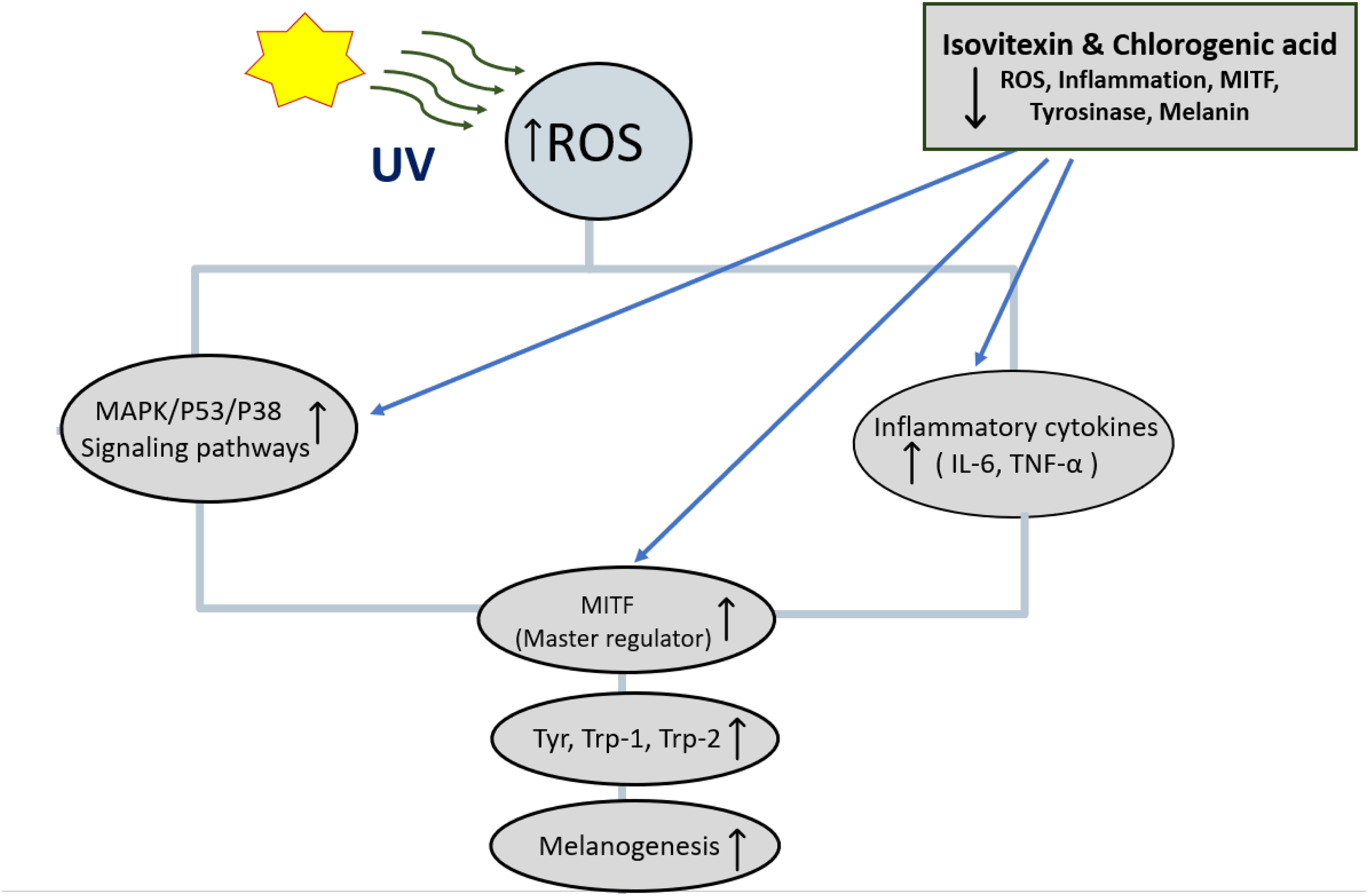
Proposed Mechanism of Isovitexin and Chlorogenic Acid in Inhibiting Melanogenesis. Ultraviolet (UV) Radiation Induces Reactive Oxygen Species (ROS), Activating Mitogen-Activated Protein Kinase (MAPK)/p53/p38 Signaling and Increasing Inflammatory Cytokines Such as Interleukin-6 (IL-6) and Tumor Necrosis Factor-α (TNF-α). These Pathways Upregulate Microphthalmia-Associated Transcription Factor (MITF), Which Stimulates Tyrosinase (Tyr), Tyrosinase-Related Protein 1 (TRP-1), and Tyrosinase-Related Protein 2 (TRP-2), Leading to Melanin Synthesis. Isovitexin and Chlorogenic Acid Suppress ROS, Inflammation, MITF, and Tyrosinase Activity, Thereby Reducing Melanogenesis.
Isovitexin showed a clear, concentration-dependent inhibition of melanin accumulation and cellular tyrosinase activity in B16 melanoma cells. At the same time, it protected dermal fibroblasts against UVB-induced cytotoxicity, suggesting that its effects extend beyond depigmentation to broader cellular protection. These observations align with reports that flavone C-glycosides act at multiple levels of melanogenesis regulation, combining direct tyrosinase inhibition with modulation of upstream signaling cascades. Importantly, these dual activities highlight its potential as a multifunctional agent that both suppresses pigmentation and maintains skin cell viability.45–48
Beyond enzyme inhibition, flavonoids are increasingly recognized as modulators of oxidative and inflammatory pathways that influence pigmentation. Excessive ROS generated by UV radiation activate MAPK and p53 signaling, upregulate MITF, and subsequently increase the expression of tyrosinase, TRP-1, and TRP-2.49,50 Literature evidence indicates that isovitexin can attenuate this process by activating the MAPK–Nrf2/ARE pathway, leading to the upregulation of antioxidant enzymes such as HO-1 and SOD, which in turn suppress oxidative stress and prevent ROS-driven melanogenic signaling.51–53 In addition, isovitexin has been shown to downregulate pro-inflammatory cytokines including Interleukin-6 (IL-6) and Tumor Necrosis Factor-α (TNF)-α, which are often elevated under UVB-induced stress and contribute to melanocyte dysfunction. Together, these activities provide a mechanistic explanation for the dual anti-melanogenic and cytoprotective effects we observed in cell-based assays.51,52,54
Recent advances in skin biology have highlighted that small bioactive peptides and natural compounds can exert profound effects on skin health by modulating oxidative stress and inflammatory signaling pathways. These molecules often act through receptors such as Toll-like receptors (TLRs) and downstream transcription factors like NF-κB, thereby reducing pro-inflammatory cytokine expression and enhancing tissue repair. In addition, some peptides influence stem cell activation and Wnt/β-catenin signaling, contributing to improved skin regeneration and barrier function. 55 Although these works do not directly measure melanogenesis, they underscore that skin homeostasis and protection involve coordinated regulation of oxidative stress, inflammatory pathways, and cell signaling. Consistently, in our study, isovitexin not only reduced melanin content and cellular tyrosinase activity but also protected cell viability under UVB challenge, suggesting that its mechanism likely includes suppression of ROS and inflammatory mediators, in line with literature showing flavonoids modulate NF-κB, MAPK, and Nrf2/ARE pathways. Incorporating measurements of ROS levels and cytokines (eg, IL-6, TNF-α) would therefore validate this hypothesis and position isovitexin and chlorogenic acid as multifunctional agents capable of promoting skin photoprotection beyond pigment inhibition.
Chlorogenic acid, while structurally distinct, has also been documented as a tyrosinase modulator and antioxidant. Its ability to scavenge ROS and to influence melanogenesis-related transcription factors suggests that it may contribute additively to the activity of the crude fractions. Given that isovitexin and chlorogenic acid target overlapping but complementary pathways, their combined presence in J. decipiens extracts likely enhances the overall anti-melanogenic activity. This interplay between flavonoid C-glycosides and phenolic acids provides a coherent explanation for the strong biological activity of the fractions prior to isolation and highlights the value of bio-guided fractionation in uncovering synergistic natural products.
The relevance of these findings extends beyond simple enzyme inhibition assays. Previous work on vitexin and isovitexin has largely relied on mushroom tyrosinase as a model, which, while informative, does not reflect the complexity of mammalian melanocyte regulation. By demonstrating activity in B16 cells, our study provides more physiologically relevant evidence of anti-melanogenic effects. Moreover, the observed UVB protection in fibroblasts suggests that these compounds can act on both melanocytes and non-pigmented skin cells, a feature that enhances their potential as multifunctional cosmetic or therapeutic agents.
Taken together, the results highlight the novelty and significance of our work. Isovitexin and chlorogenic acid are reported here for the first time from J. decipiens, and their activity against cellular melanogenesis and UVB-induced stress establishes new biological roles for this species. The combined enzyme-inhibitory, antioxidant, and anti-inflammatory activities of these compounds support a multi-target mechanism of action that is particularly valuable in addressing the limitations of current depigmenting agents, which often suffer from cytotoxicity or limited efficacy. Further studies measuring intracellular ROS levels, cytokine expression, and melanogenesis-related transcription factors will provide additional mechanistic clarity and strengthen the translational relevance of these findings.
Conclusion
This study highlights J. decipiens as a promising source of multifunctional phytochemicals with potential applications in dermatology and cosmetic science. Through a systematic bioassay-guided fractionation strategy, we successfully isolated and identified isovitexin as the principal bioactive compound in the EtOAc fraction. Isovitexin significantly inhibited melanin synthesis and intracellular tyrosinase activity in B16 melanoma cells without inducing cytotoxicity, indicating its safety and effectiveness as a depigmenting agent. Importantly, this is the first report to isolate isovitexin from J. decipiens and to experimentally demonstrate its role in melanogenesis inhibition. In addition to its anti-melanogenic properties, isovitexin exhibited significant photoprotective activity by preserving cell viability under UVB-induced oxidative stress, suggesting its dual role as both a melanin-inhibiting and skin-protective compound an ideal candidate for cosmetic and dermatological formulations.
Furthermore, chlorogenic acid was identified in the DCM fraction. The literature reports support its role in pigmentation modulation, indicating possible synergistic effects within the plant extract. Collectively, these findings establish J. decipiens as a novel source of bioactive compounds with dual-action potential for skin depigmentation and photoprotection. Future research involving in vivo models, mechanistic elucidation, and formulation studies is warranted to fully explore its therapeutic and commercial potential.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251388346 - Supplemental material for Isovitexin and Chlorogenic Acid from Juncus decipiens (Buchenau) Nakai Target Melanogenesis and Enhance Photoprotection
Supplemental material, sj-docx-1-npx-10.1177_1934578X251388346 for Isovitexin and Chlorogenic Acid from Juncus decipiens (Buchenau) Nakai Target Melanogenesis and Enhance Photoprotection by Sana Ullah, Ahmed Othman, Moustafa M. Zohair, Fahd M. Abdelkarem, Masako Matsumoto, Maki Nagata, Yhiya Amen and Kuniyoshi Shimizu in Natural Product Communications
Footnotes
Acknowledgment
The authors express their sincere gratitude to the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) of Japan for its financial assistance through the MEXT Scholarship Program. We also want to express our gratitude to the Kumamoto Prefectural Research Center's Agricultural System Research Institute for providing lab space and technical support during the study. Their assistance and input were crucial to this study's effective conclusion.
Ethical Approval
Ethical approval is not applicable for this work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
