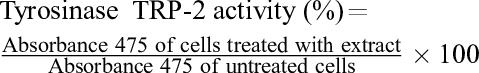Abstract
Phlorotannins isolated from brown algae, such as Eisena bicyclis, have positive physiological effects, including anti-cancer, anti-inflammatory, and anti-Alzheimer’s disease. Although phlorotannins have been shown to inhibit tyrosinase, an enzyme essential for melanogenesis, their effect on melanogenesis remains unexplored. Thus, we isolated phlorotannins from E. bicyclis and examined their effects on α-melanocyte-stimulating hormone (α-MSH)-induced melanogenesis in murine B16 melanoma cells. Both fucofuroeckol-A (FF-A) and phlorofucofuroeckol-A (PFF-A) suppressed α-MSH-induced melanogenesis. Neither inhibited human tyrosinase (TYR) activity, but both inhibited tyrosinase-related protein-2 activity. FF-A downregulated the expression of microphthalmia-associated transcription factor and TYR, which subsequently suppressed melanin production. These results suggest that phlorotannins could be beneficial as melanin control drugs for hyperpigmentation disorders.
The epidermis is composed of multiple layers that serve as protection against external factors such as pathogens and UV radiation (UVR). 1 Melanocytes, located in the basal layer of the epidermis, play a key role in the defense against UVR by producing melanin through a complex process called melanogenesis. 1,2 Although the melanin pigment is crucial in the protection of human skin, excessive melanin accumulation can induce skin disorders, including freckles and melasma. 3,4 Thus, effective approaches to inhibit melanin synthesis are required.
Upon UVR exposure, the melanogenesis signaling pathway is initiated by several melanogenic factors, including α-melanocyte-stimulating hormone (α-MSH) and endothelin-1. 5,6 When α-MSH is cleaved from pro-opiomelanocortin, it increases the levels of intracellular cyclic adenosine monophosphate (cAMP) via the melanocortin 1 receptor. This activates the cAMP-protein kinase A (PKA) pathway and subsequently the downstream molecule, microphthalmia-associated transcription factor (MITF), which regulates the mRNA expression of melanogenesis enzymes such as tyrosinase (TYR), tyrosinase-related protein 1 (TRP-1) and 2 (TRP-2). 7 TYR acts as a rate-limiting enzyme and regulates melanogenesis by catalyzing hydroxylation. 8 Therefore, most skin whitening agents, such as hydroquinone, kojic acid, and ascorbic acid, target TYR. However, allergic and toxic effects on the skin have been reported when these agents are used in high doses and for a long time. 9 -11 Accordingly, many researchers are currently focused on investigating natural skin whitening compounds with fewer side effects.
Eisenia bicyclis is a perennial brown alga that inhabits the coastal areas of Japan and Korea. It contains various compounds such as polysaccharides, tripeptides, and phlorotannins. 12 -14 Phlorotannins are responsible for its anti-cancer, 15 anti-inflammatory, 16 anti-Alzheimer’s disease, 17 and anti-tyrosinase 18 activities. However, the effects of phlorotannins isolated from E. bicyclis on melanogenesis remain unknown. Thus, this study investigated the anti-melanogenesis effects of extracts derived from E. bicyclis.
Results and Discussion
Regardless of age or sex, melanin control drugs are commonly used to prevent skin disorders such as freckles and melasma. 19 However, the long-term use of whitening agents such as hydroquinone, kojic acid, and ascorbic acid can lead to skin diseases. Hence, many researchers are currently focused on searching for safe and effective natural compounds that can serve as substitutes for melanin control drugs. Recently, brown algae have attracted considerable attention of the cosmetic industry because they contain various biologically active compounds. Although the tyrosinase inhibitory effect of phlorotannins isolated from brown algae has been reported, their impact on the regulatory factors involved in melanin synthesis remains unclear. In the present study, we evaluated the inhibitory activity of phlorotannins isolated from E. bicyclis on melanogenesis and investigated the mechanisms underpinning these functions in melanoma cells.
To determine the cytotoxicity of the prepared crude phlorotannins (C-Phlo) and purified compounds, we performed a cell proliferation assay using WST-1 reagents. C-Phlo had no dramatic influence on cell viability in the concentration range of 10‐100 µg/mL (Figure 1(A)). Moreover, among the 6 phlorotannins, only fucofuroeckol-A (FF-A) had an impact on cell viability (79.3%) when used at a concentration of 50 µM compared with the control Figure 1(B). Thus, phlorotannin concentrations of up to 50 µM were chosen for the subsequent experiments.

The effect of crude phlorotannins and 6 purified compounds on B16-4A5 cell viability. Cells (2.0 × 105 cells/mL) were treated with either (
Although melanin is necessary to protect skin cells from UVR damage, excessive melanin production and accumulation induce hyperpigmentation. Thus, as a next step, we examined whether C-Phlo extracted from E. bicyclis suppresses α-MSH-induced melanogenesis. As shown in Figure 2(A), C-Phlo significantly decreased melanin production in a dose-dependent manner. To confirm whether the phlorotannins contained in C-Phlo decreased melanin content, we repeated the experiment using the purified phlorotannins. All six phlorotannins exhibited anti-melanogenesis activity, and the effect was similar to that of arbutin treatment (Figure 2(B)).
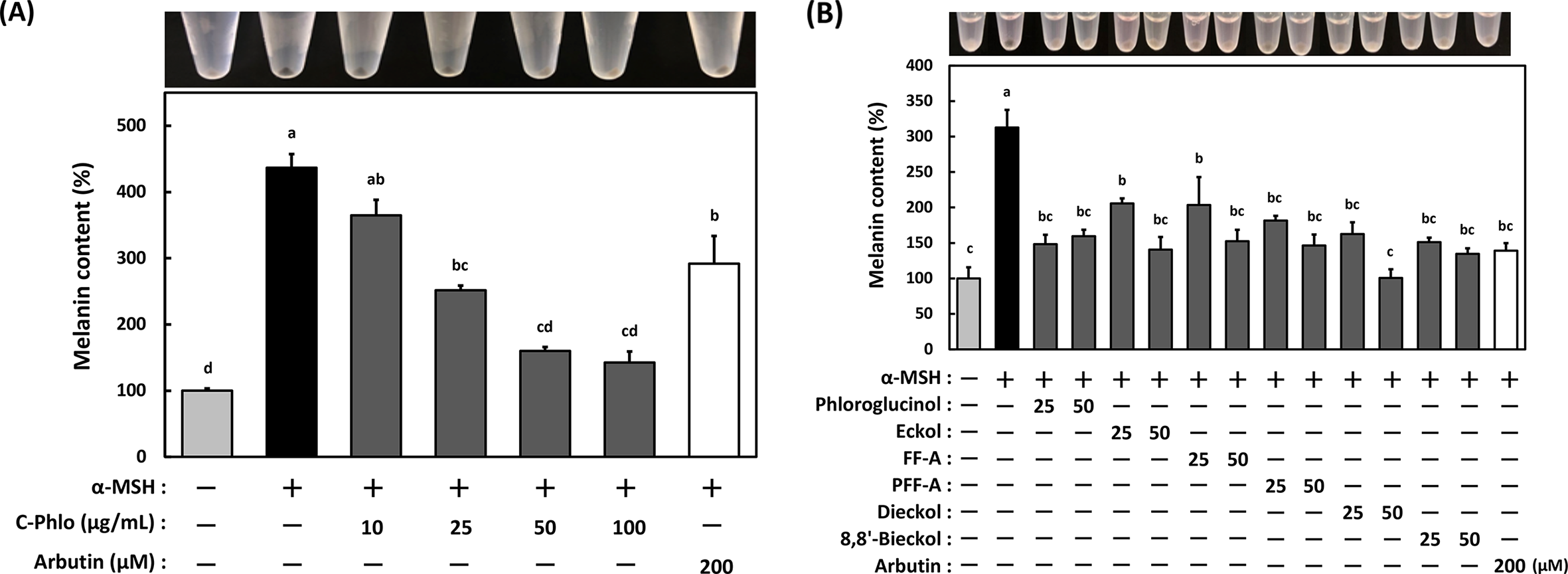
The effect of crude phlorotannins (C-Phlo) and six compounds on melanin content in B16-4A5 cells. Cells (5.0 × 104 cells/mL) were pretreated with either (
Although melanogenesis is a complicated process and is regulated by various factors, targeting TYR, the rate-limiting enzyme, is an easy strategy for inhibiting hyperpigmentation. It has been reported that phlorotannins such as phloroglucinol, eckol, and dieckol inhibit melanin production by targeting TYR. 18,20 In addition, it has been reported that phlorofucofuroeckol-A (PFF-A), which is less effective, also acts as a TYR inhibitor. 18 Therefore, among the six phlorotannins isolated from C-Phlo, we focused on PFF-A (for which the anti-melanogenesis mechanism has not been elucidated) and FF-A (which has a similar structure to PFF-A and has not yet been reported) for subsequent experiments. Although mushroom tyrosinase (mTyr) has been used for TYR activity assays in many reports, we did not consider it to be ideal for our experiments because the structural characteristics of mTyr and TYR are different. 18,21,22 Therefore, we evaluated the inhibitory effects of C-Phlo, FF-A, and PFF-A by using both mTyr and TYR. C-Phlo significantly attenuated mTyr activity in a dose-dependent manner (Figure 3(A)). Similarly, FF-A suppressed mTyr activity by approximately 20%, although the difference was not statistically significant (Figure 3(B)). On the other hand, PFF-A did not suppress either mTyr or TYR activity (Figure 3(B)). Although Shim et al. and Manandhar et al. had previously shown that FF-A and PFF-A notably inhibit mTyr, 23,24 our analysis showed different results. We thus hypothesized that FF-A and PFF-A might exert their anti-melanogenesis activity by targeting TRP-2 activity and we therefore performed inhibition analyses. Compared to the cells stimulated with α-MSH (positive control), TRP-2 activity was significantly downregulated, to a similar extent, by either FF-A or PFF-A (Figure 4).

The effect of (
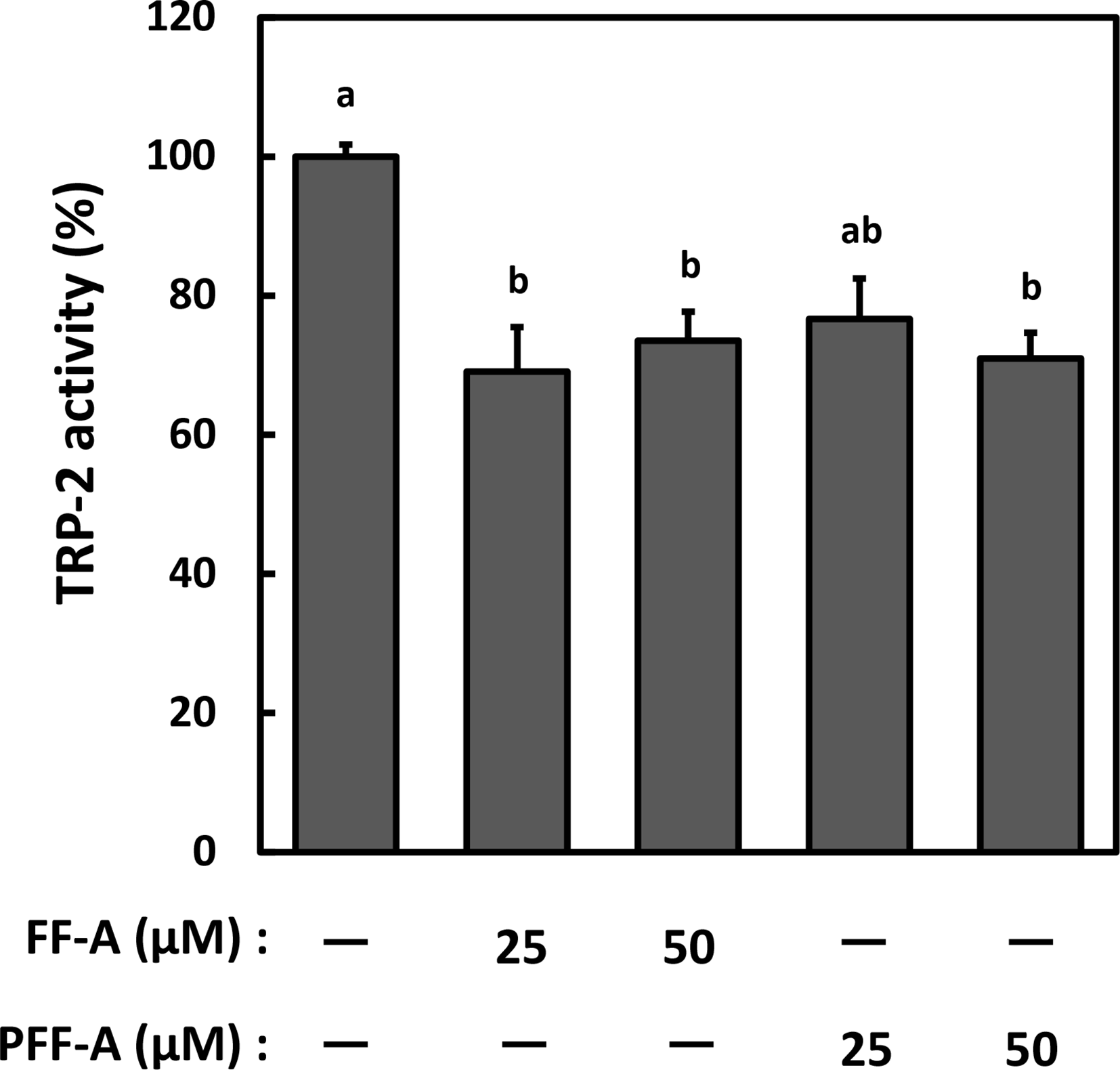
The effect of FF-A and PFF-A on tyrosinase-related protein-2 (TRP-2) activity. Dopachrome, which was prepared by oxidizing L-DOPA with Ag2O in the presence of 0.1 mM urea, was incubated with either FF-A or PFF-A (10, 50 µM) at 37 ℃ for 10 minutes. After incubation, the crude enzyme solution derived from HMV-Ⅱ cells was added at 37 ℃ for 15 minutes. Data represent the mean ± SE and statistical analysis was conducted using the Tukey-Kramer test (n = 12, P < 0.01).
To elucidate the molecular mechanism underlying the anti-melanogenesis effect of FF-A and PFF-A, we examined the mRNA expression of crucial melanogenic genes in B16 cells treated with FF-A and PFF-A, including Mitf, Tyr, Trp-1, and Trp-2, using real-time qPCR. The results showed that FF-A significantly downregulated the mRNA levels of Mitf and Tyr in the α-MSH-induced B16 cells (Figure 5). In addition, the protein expression levels of Mitf and Tyr in cells treated with FF-A also decreased (Figure 6). On the other hand, PFF-A-treated cells did not show any change in mRNA and protein expression compared to the untreated cells (Figures 5 and 6).

The effect of FF-A and PFF-A on the expression of melanogenesis-related genes. B16-4A5 cells were stimulated with α-MSH in either the presence or absence of either FF-A or PFF-A (50 µM). The mRNA levels of Mitf, Tyr, Trp-1, and Trp-2 were determined using RT-qPCR. Gapdh was used as an internal standard. Data represent the mean ± SE and statistical analysis was conducted using the Tukey-Kramer test (n = 9, P < 0.01).

The effect of FF-A and PFF-A on melanogenic protein expression. B16-4A5 cells were stimulated with α-MSH in either the presence or absence of either FF-A or PFF-A (50 µM). Cell lysates were separated by SDS-PAGE and expression levels were determined using Western blotting. β-Actin was used as a loading control. Representative Western blots from 3 independent experiments are shown. Statistical analysis was performed using the Tukey-Kramer test; p-values < 0.05 indicated significant differences.
During melanogenesis, α-MSH stimulation results in the activation of the cAMP/PKA/cAMP response element-binding protein (CREB) signaling pathway. CREB binds to and activates the Mitf promoter via the cAMP response element. Seo et al. have previously reported that Leathesia difformis extracts exert their anti-melanogenesis activity via the cAMP/PKA/CREB signaling pathway located upstream of Mitf. 25 Therefore, we examined the levels of CREB to gain further insights into the mechanism underlying the suppression of melanogenesis in cells treated with FF-A. However, FF-A did not attenuate CREB phosphorylation after α-MSH stimulation (Figure 7). Since the translocation of p-CREB to the nucleus is observed in melanogenesis in B16 cells treated with forskolin, 26 we next further analyzed the levels of p-CREB translocation to the nucleus. However, FFA did not inhibit the nuclear translocation of p-CREB (Supplemental Figure 1). Hence, we speculate that FF-A may inhibit either the transcriptional activity or other signaling pathways (such as PAK4, MAP kinases, and etc.) leading to MITF expression. With regard to the inhibitory mechanisms of melanogenesis by FFA, we need further study and would like to show our results in a later report.

The effect of FF-A on CREB protein activation (phosphorylation). B16-4A5 cells were treated with α-MSH in either the presence or absence of FF-A (50 µM) for the indicated times. Cell lysates were separated by SDS-PAGE and expression levels were determined using western blotting. β-Actin was used as a loading control. Representative western blots from 3 independent experiments are shown.
In conclusion, melanogenesis is attenuated by FF-A through the suppression of Mitf and Tyr expression in α-MSH-induced B16 cells. However, since Mitf expression is regulated by the CREB transcription factor, further studies are needed to determine the mechanisms of this suppression. In contrast to FF-A, PFF-A did not influence either mRNA or protein expression; however, it reduced melanin production by targeting TRP-2 activity (Figure 8). Therefore, FF-A and PFF-A are useful botanical sources of anti-melanogenesis agents.

A scheme of the inhibitory mechanisms whereby FF-A and PFF-A exert their anti-melanogenesis activities.
Materials and Methods
Antibodies
Antibodies against MITF (#MAB3747) were purchased from Millipore (Burlington, MA, USA), those against TYR (ab52493) and TRP-2 (ab74073) from Abcam (Cambridge, UK), those against TRP-1 sc-166857) from Santa Cruz Biotechnology (Santa Cruz, CA, USA), those against CREB (#9197) and phosphorylated-CREB (Ser 133, #9198) from Cell Signaling Technology (Beverly, MA, USA), and those against β-actin from Sigma-Aldrich (St. Louis, MO, USA). ECL anti-rabbit IgG horseradish peroxidase and ECL anti-mouse IgG horseradish peroxidase were purchased from GE Healthcare (Chicago, IL, USA).
Extraction and Purification
The brown alga Eisenia bicyclis (Kjellman) Setchell was obtained from the coasts of the Itoshima Peninsula (33°63’N, 130°18’E) in Fukuoka, Japan. The air-dried alga was pulverized using an absolute mill (Osaka Chemical, Osaka, Japan) and stored at −30 °C until further use. The preparation of crude phlorotannins (C-Phlo) from algal powder was performed according to the protocol described by Nagayama et al. 27 Each phlorotannin was fractionated on a Wakogel C-300HG column (1.5 cm i.d. ×150 cm, Wako Pure Chemical Ind., Osaka, Japan) with chloroform-methanol-water (80:20:2, v/v) as the eluent. 28 Further purification of the phlorotannins was conducted using a preparative HPLC system equipped with an Inertsil ODS-3 column (10 mm I.D. ×250 mm, GL Science, Tokyo, Japan), according to a previously described method. 29 Identification of the isolated phlorotannins was carried out using an LC/MS system (6120 Quadrupole LC/MS with 1260 Series HPLC System, Agilent, CA, USA). Elution was performed at a flow rate of 1.0 mL/min using a linear gradient from 20% to 100% methanol for 20 minutes, and maintained for 20 minutes. Each phlorotannin was monitored by electrospray ionization mass spectrometric analysis in the negative mode. The purity of each phlorotannin at 98% or higher was confirmed by 3 dimensional HPLC (SPD-M10AV, Shimadzu, Kyoto, Japan) with an Inertsil ODS-3 column (4.6 mm I.D. × 250 mm, GL Science). The structural formulae of the isolated phlorotannins are shown in Figure 9.

Chemical structures of the phlorotannins isolated from the brown alga Eisenia bicyclis.
Cell Culture
The mouse melanoma cell line B16 and the human malignant melanoma cell line HMV-II were purchased from the Riken BioResource Research Center (Ibaraki, Japan). The B16 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; GE Healthcare, Chicago, IL, USA), and the HMV-II cells in Ham’s F-12 nutrient medium (Sigma-Aldrich, St. Louis, MO, USA), both supplemented with 10% heat-inactivated fetal bovine serum (Life Technologies, Carlsbad, CA, USA) and 50 U/mL penicillin-50 µg/mL streptomycin (Thermo Scientific, MA, USA). The cells were cultured at 37 °C in a humidified atmosphere of 5% CO2.
Cell Viability
Cell viability was assessed using a WST-1 cell proliferation assay. B16 cells were seeded in a 96-well plate at a density of 2.0 × 105 cells/mL and pre-cultured for 24 hours. The cells were treated with 10, 25, 50, and 100 µg/mL of C-Phlo, or 25 and 50 µM of each purified phlorotannin, at 37 °C for 24 hours. After incubation, 100 µL of WST-1 (1/10 dilution in DMEM, Takara Bio, Ohtsu, Shiga, Japan) was added to each well for an additional 1 hours. The absorbance was measured at a wavelength of 470 nm using a microplate reader (Varioscan LuxTM, Thermo Scientific, MA, USA).
Measurement of Melanin Content
B16 cells were seeded in a 12-well culture plate at a density of 5.0 × 104 cells/mL and cultured for 24 hours. Cells were then treated with 10, 25, 50, or 100 µg/mL of C-Phlo; 25 or 50 µM of each phlorotannin; or 200 µM of arbutin as a positive control, for 2 hours. After treatment, cells were stimulated with 2 µM α-MSH (Sigma-Aldrich) for 72 hours. Cells were then washed twice with PBS and lysed with 100 µL of 1 n NaOH at 50 °C for 1 hours. Absorbance was measured at a wavelength of 475 nm using a microplate reader (Varioscan LuxTM). The melanin content was calculated from a standard curve prepared using synthetic melanin, and normalized by the protein concentration of each cell, which was determined using a DC Protein Assay Kit (Bio-Rad, Hercules, CA, USA) as a standard.
Tyrosinase and Tyrosinase-Related Protein-2 Activity
Fully confluent HMV-II cells were washed twice with PBS and centrifuged at 145 × g for 3 minutes. Cell pellets (1.0 × 107 cells) were suspended in 500 µL of RIPA buffer and disrupted using a sonicator (Ultrasonic cell disruptor, Wakenyaku, Kyoto, Japan) on ice. The suspension was further centrifuged at 13,500 × g for 15 minutes at 4 °C to prepare the crude enzyme solution. To measure tyrosinase activity, 50 µL of C-Phlo, FF-A, or PFF-A (final concentration: 10‐100 µM) was added to 25 µL of 15% L-3, 4-dihydroxyphenylalanine (L-DOPA, Sigma-Aldrich). To measure TRP-2 activity, the same volume of dopachrome was used as a substrate instead of L-DOPA. Dopachrome was prepared by oxidizing L-DOPA with Ag2O in the presence of 0.1 mM urea in a 96-well plate at 37 °C for 10 minutes. After incubation, either a crude enzyme solution or 1 mg of mushroom tyrosinase (Sigma-Aldrich, 2400 units/mg) in 1 ml of 0.1 M phosphate buffer was added to each well at 37 °C for 15 minutes. The absorbance was measured using a microplate reader, and tyrosinase activity was calculated using the following formula:
RNA Isolation and Quantitative Real-Time PCR
To determine the expression levels of Mitf, Tyr, Trp-1, and Trp-2, total RNA was prepared using TRIzolTM reagent (Life Technologies) and quantified using a NanoDrop Lite (Thermo Fisher Scientific). We prepared cDNA from total RNA samples using a High-Capacity RNA-to-cDNA Kit (Applied Biosystems, Foster City, CA, USA). Subsequently, quantitative real-time PCR was performed using the StepOneTM Real Time PCR System (Applied Biosystems) with Power SYBR® Green PCR master mix (Applied Biosystems) under the following cycling conditions: 45 cycles at 95 °C for 15 s and at 60 °C for 1 minutes. The primer sequences were as follows: Gapdh forward, 5′-GCACCGTCAAGGCTGAGAAC-3′ and reverse, 5′-TGGTGAAGACGCCAGTGGA-3′; Mitf forward, 5′-ACTATGGCCAAGGCAGAGCAAC-3′ and reverse, 5′-TCAGAGGCTACAAGCCAAGGTAATG-3′; tyrosinase forward, 5′-CAAAGTACAGGGATCGGCCAAC-3′ and reverse, 5′-GGTGCATTGGCTTCTGGGTAA-3′; Trp-1-forward, 5′-TGATGCGGTCTTTGACGAATG-3′ and reverse, 5′-GTTGGTAACTGGAGGCCAGAATG-3′; Trp-2-forward, 5′-AACCGCAGAGCAACTTGGCTAC-3′ and reverse, 5′-CTCCCAGGATTCCAATGACCAC-3′. Gapdh was used as an internal standard.
Western Blot Analysis
Whole cell lysates were obtained using RIPA buffer containing protease inhibitors (Roche Diagnostics, GmbH, Mannheim, Germany) and a phosphatase inhibitor (Sigma-Aldrich). Protein concentration was measured using a DC Protein Assay Kit (Bio-Rad). Equal amounts of protein were resuspended in lysis buffer [125 mM Tris-HCl (pH 6.8), 4% sodium dodecyl sulfate (SDS), 20% glycerol, 10% 2-mercaptoethanol] and denatured at 98 °C for 5 minutes. Samples were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and electroblotted onto polyvinylidene difluoride (PVDF) membranes. After blocking non-specific binding sites for 1 hours with 5% (w/v) nonfat milk (Becton, Dickinson and Company, Sparks, MD, USA) in Tris-buffered saline containing 0.1% (w/v) Tween 20 (FUJIFILM Wako Pure Chemical Co., Osaka, Japan, TBS-T), the PVDF membrane was incubated overnight at 4 °C with various primary antibodies. The membranes were then washed 3 times with TBS-T and incubated with the appropriate horseradish peroxidase-conjugated secondary antibodies at 25 °C. Immunolabeled proteins were detected using an ECL Prime Chemiluminescence Kit (GE Healthcare, Piscataway, NJ, USA) and a Davinch-Chemi imager™ (CAS-400SM, Davinch-K, Seoul, Korea).
Statistical Analysis
All data are expressed as the mean ± standard error (SE). Statistical comparisons between different treatments were assessed using the Tukey-Kramer test, and differences were considered significant when P values were less than 0.01.
Supplemental Material
Online supplementary file 1 - Supplemental material for Effect of Phlorotannins Isolated From Eisenia bicyclis on Melanogenesis in Mouse B16 Melanoma Cells
Supplemental material, Online supplementary file 1, for Effect of Phlorotannins Isolated From Eisenia bicyclis on Melanogenesis in Mouse B16 Melanoma Cells by Yuki Ohno, Shiori Kondo, Kiho Tajima, Toshiyuki Shibata and Tomohiro Itoh in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.