Abstract
Ganpu tea is a novel type of beverage produced from Pu-erh tea stuffed in the pericarp of Citrus reticulata “Chachi”. It has gained considerable popularity in China owing to its inviting flavor and health effects. However, the in vivo metabolites of Ganpu tea, which may contribute to its overall health effects, are still unclear. In the present work, rat urine and feces samples were collected after oral administration of Ganpu tea extract (GTE), and then subjected to ultra-high-performance liquid chromatography/quadrupole-time-of-flight tandem mass spectrometry (UHPLC-Q-TOF-MS/MS)-based metabolite profiling. As a result, 27 prototype compounds and 41 metabolites derived from caffeic acid, gallic acid, p-coumaric acid, xanthine, catechin, polymethoxyflavone, (PMF) flavanone, and flavone were identified in rat urine and feces. Based on the detected metabolites, the ingested prototype compounds derived from Ganpu tea were found to undergo extensive phase II metabolism in rats, especially and sulfation. These results will be valuable for interpreting the health effects of Ganpu tea.
Introduction
Pu-erh tea is a variety of fermented tea made from the leaves of a large leaf tea species (Camellia sinensis Linn. var. assamica [Masters] Kitamura) in Yunnan Province of China. 1 After sufficient sun drying and being rolled, the leaves further undergo controlled microbial fermentation and accompanying oxidization, until the desired flavors are reached. 2 A No. of studies have documented that Pu-erh tea could exert multiple health-promoting effects, such as anti-oxidative, anti-inflammatory, anti-hyperlipidemic, and anti-diabetic activities. 3 Owing to its unique flavor, Pu-erh tea is hugely popular among consumers in China and Southeast Asia, with an annual output of more than 150,000 tons. In recent years, a novel type of tea beverage derived from Pu-erh tea, named Ganpu tea, has appeared on the market and gained considerable popularity in China. 4 Ganpu tea could be defined as Pu-erh tea stuffed in the pericarp of Citrus reticulata “Chachi” from Xinhui County in Guangdong Province of China. 5 The pericarp of C reticulata “Chachi”, called “Guangchenpi” in Chinese, is a widely used cooking spice and traditional herb in China due to its special flavor and beneficial health effects.6,7 According to the literature, the combination of tea leaves and citrus peel as a brewed tea has a history of thousands of years. The tangy aromas of the pericarp of C reticulata “Chachi” is a perfect match for the mellow earthiness of Pu-erh tea, leading to its inviting flavor. 8 Moreover, several studies have revealed the health effects of Ganpu tea, such as antioxidation, anti-proliferation, anti-depression, α-amylase inhibition, and modulating gut microbiota.9–12 Nowadays, the consumption of Ganpu tea is increasing rapidly, and thousands of enterprises are involved in its production.
In a previous study, we investigated the chemical profiles of Ganpu tea using ultra-high-performance liquid chromatography/quadrupole-time-of-flight tandem mass spectrometry (UHPLC-Q-TOF-MS/MS). 12 A total of 92 compounds were detected, including flavonoids, organic acids, alkaloids, and limonin. In addition, several other studies have investigated the volatile components, tea polyphenols, and rare earth elements of Ganpu tea.13–15 However, the in vivo metabolite profiles of Ganpu tea have not yet been investigated. During in vivo digestion and metabolism, phytochemicals would undergo structure modifications to generate a range of metabolites, which might have different biological activities from their initial compounds. 16 More and more studies have shown that derived metabolites are not negligible contributors to the health effects of dietary phytochemicals.17–19 Therefore, clarification of the in vivo metabolite profiles of Ganpu tea is a worthwhile study. In the present work, UHPLC-Q-TOF-MS/MS-based metabolite profiling was performed on rat urine and feces samples collected after the oral administration of Ganpu tea extract (GTE). The obtained results should be valuable for further study of interpreting the health effects of Ganpu tea.
Materials and Methods
Chemicals and Materials
The reference standards of caffeic acid, gallic acid, caffeine, catechin, quercetin, and kaempferol were obtained from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). Naringenin, nobiletin, hesperetin, p-coumaric acid, and formic acid of mass spectrometry (MS) grade were acquired from Sigma-Aldrich (St. Louis, USA), apigenin from Shanghai Macklin Biochemical Co., Ltd (Shanghai, China), homoeriodictyol from Extrasynthese Chemical (Genay Cedex, France), and the stable isotope-labeled internal standard (IS) [2′,3′,5′,6′-D4]-naringenin from Artis-chem Co., Ltd (Shanghai, China).
Acetonitrile of LC-MS and HPLC grade were purchased from Fisher Scientific Inc. (Fair Lawn, USA) and Honeywell B&J Chemicals Inc. (New Jersey, USA), respectively. Water was prepared with a Milli-Q Plus system (specific electric conductivity = 18.2 MΩ) and filtered through a 0.22 μm membrane filter before use.
Preparation of Ganpu tea Extract
One hundred g of pulverized Ganpu tea sample was soaked in 1.5 L of boiling water for 15 min, and filtered to obtain the tea infusion. This extraction process was repeated twice and the tea infusions were mixed together. The mixed tea infusion was evaporated to 500 mL in a rotary evaporator (Eyela, Tokyo, Japan) to afford GTE with a concentration of 0.2 g/mL.
Sample Collection
Ten male Sprague-Dawley rats (weighing 180-220 g) were acquired from Guangdong Medical Laboratory Animal Center. The animals were housed in a standard environment at 20 to C, with a light-dark cycle of 12/12 h; food and water were supplied ad libitum. All experimental processes were approved by the Institutional Animal Care and Use Committee (IACUC) of Sun Yat-Sen University, and conducted in accordance with the National Institutes of Health guide for the care and use of laboratory animals (NIH Publications No. 8023, revised 1978).
After 1 week of adaptive feeding, rats were intragastrically given Ganpu tea extract (15 mL/kg, calculated as 3 g/kg of raw Ganpu tea), a dosage equivalent to 33 g/70 kg for an adult human. After administration, the rats were kept together in a metabolic cage, with free access to water, but no food. Urine and feces samples were collected within 12 h after the intervention. Feces sample was lyophilized, pulverized, and then mixed with saline (20 mL/1 g feces). After vortexing for 1 min, the mixtures underwent ultrasonic-assisted extraction for 15 min, and then centrifuged at 2348 × g for 1 min, so as to obtain the supernatant for further analysis. All biological samples were stored at − 70 °C until analysis.
Sample Preparation and Detection
For metabolite identification, an aliquot of 200 μL acetonitrile (containing IS at a concentration of 15 μg/mL) was added to 100 μL of either urine or feces extract, vortex-mixed for 3 min, and then centrifuged at 15,871 × g for 30 min to acquire the supernatant for UHPLC-Q-TOF-MS/MS analysis. The injection volume was set to 10 μL.
Sample detection was performed on a Shimadzu ultra-high-performance liquid chromatography system (UHPLC; 20ADXR; Shimadzu) in tandem with a hybrid triple quadrupole time-of-flight mass spectrometer (Q-TOF-MS/MS; Triple TOF 5600 plus, Sciex), equipped with an electrospray ionization (ESI) source. The chromatographic separation system and mass spectrometer detector were operated under the same parameters as our reported studies.20,21
Data Analysis
Raw data were acquired using Analyst® TF 1.6 software (AB Sciex) in information-dependent acquisition mode. Potential metabolites were characterized on the basis of chromatographic elution time, exact molecular mass, chemical formula, and MS/MS fragmentation pattern, as well as by comparison with available reference standards and published literature.
Results and Discussion
In a previous study, we investigated the chemical composition of GTE and finally identified 92 compounds, including 71 flavonoids (comprising 33 polymethoxyflavones [PMFs] and 8 catechins), 14 organic acids, 6 alkaloids, and limonin. 12 However, information concerning the in vivo metabolism of Ganpu tea is still scarce. In this work, GTE was orally administrated to rats to collect urine and feces samples for UHPLC-Q-TOF-MS/MS-based metabolite profiling. The typical basic peak chromatograms of rat urine collected after the intervention of Ganpu tea are shown in Figure 1, while the compound name, chemical formula, retention time (RT), exact molecular weight, and MS/MS fragment ions of the identified metabolites are listed in Table 1.

The basic peak chromatograms of rat urine samples were collected after the oral administration of Ganpu tea extract (GTE) in negative (a) and positive (b) ion modes.
Identification of Metabolites in rat Urine and Feces Samples After Oral Administration of Ganpu tea Extract (GTE).

Confirmation in comparison with authentic standards.
The losses are Glc = glucose moiety, Rha = rhamnose moiety and, ND = not detect.
Confirmation in comparison with mass spectral library (Natural Products HR-MS/MS Spectral Library, Version 1.0; AB Sciex).
Abbreviation: RT: retention time.
Herein, a total of 27 prototype compounds and 41 metabolites derived from caffeic acid, gallic acid, p-coumaric acid, xanthine, catechin, PMF, flavanone, and flavone were detected. These derivative metabolites included 21 glucuronides and 14 sulfates, revealing that ingested prototype compounds derived from Ganpu tea underwent extensive phase II metabolism in rats. Specifically, the hydroxyl groups in caffeic acid, gallic acid, p-coumaric acid, catechin, and flavonoids could conjugate with glucuronic or sulfuric acid moieties, giving rise to a No. of phase II metabolites.
Identification of Caffeic Acid Derivatives
Caffeic acid (3,4-dihydroxycinnamic acid) is widely distributed in plants and their products owing to its intermediate role in the biosynthesis of lignin.22,23 It was also detected in GCP and Puerh tea, the raw materials of Ganpu tea.12,24 As shown in Figure 2, caffeic acid has 3 hydroxyl sites, which can be combined with glucuronide, sulfate, and other groups. The caffeic acid reference standard gave a deprotonated quasi-molecular ion ([M-H]−) at m/z 179 (C9H8O4) and a characteristic fragment ion at m/z 135 by the loss of one CO2 molecule. In this work, compounds 1, 2, and 3 all displayed a [M-H]− ion at m/z 355 (C15H16O10), 176 Da (C6H8O6) more than that of caffeic acid. They also showed product ions at m/z 179 and m/z 135, which were consistent with the fragmentation pattern of caffeic acid. Hence, compounds 1, 2, and 3 were tentatively characterized as caffeic acid-O-glucuronide. Similarly, compounds 4, 5, and 6 were assigned as caffeic acid-O-sulfate. However, due to the unavailability of corresponding commercial standards, the exact binding sites of the mentioned glucuronides and sulfates have yet to be determined.
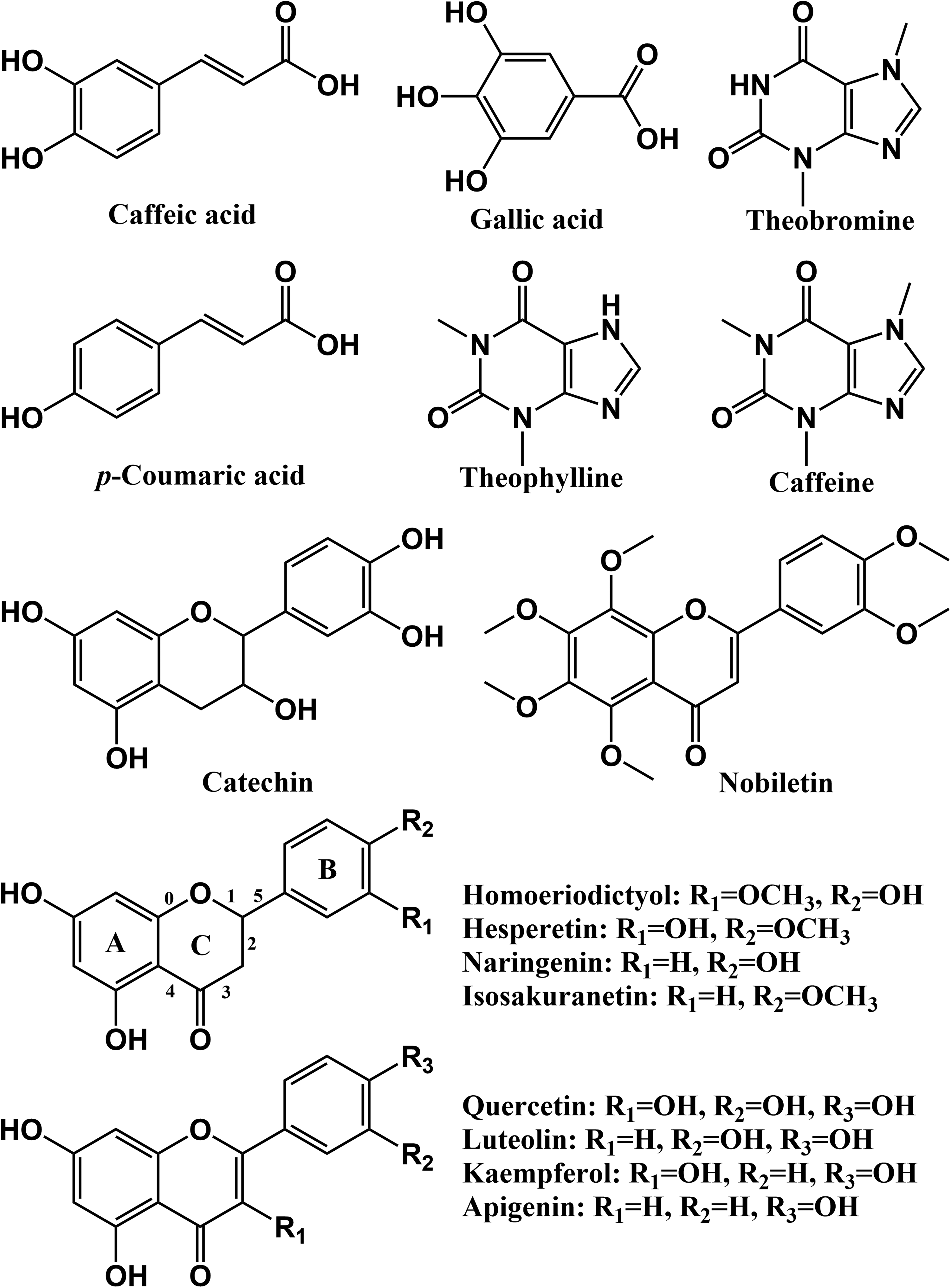
Structures of primary compounds detected in rat urine collected after the oral administration of Ganpu tea extract (GTE).
Identification of Gallic Acid Derivatives
Gallic acid is a trihydroxybenzoic acid with the formula C7H6O5. In negative ion mode, it exhibited a main product ion at m/z 125 by losing a CO2 molecule. Herein, compound 7 showed an [M-H]− ion at m/z 249 (C7H6O8S), 80 Da (SO3) more than that of gallic acid, and yielded similar ion signals at m/z 169 and m/z 125. By comparison with reported data, 25 compound 7 was tentatively identified as gallic acid-O-sulfate.
Identification of p-Coumaric Acid Derivatives
Similar to caffeic acid, p-coumaric acid is also a hydroxy derivative of cinnamic acid. Compound 8 was unequivocally identified as p-coumaric acid. It eluted at 10.5 min and gave an [M-H]− ion at m/z 163 (C9H8O3), as well as a fragment ion at m/z 119 by the loss of one CO2 molecule. Compounds 9 and 10 had the same [M-H]− ion at m/z 243 (C9H8O6S), 80 Da (SO3) more than that of p-coumaric acid. Both of them produced product ion signals at m/z 163 and m/z 119. By comparison with the literature, 26 compounds 9 and 10 were presumed to be p-coumaric acid-O-sulfate.
Identification of Xanthine Derivatives
Xanthine is a common alkaloid encountered in tea and coffee. 27 It is an intermediate product of purine degradation, subsequently converted to methylxanthine and uric acid by the action of corresponding enzymes. 28 In this work, 8 xanthine derivatives were characterized. Compounds 11 and 12 both exhibited protonated quasi-molecular ions ([M + H]+) at m/z 181 (C7H8N4O2) and a characteristic product ion at m/z 163. Based on the obtained exact molecular mass and formula, these two compounds were proposed as dimethylxanthine. With reference to chromatographic RT and other information in the literature,29,30 compounds 11 and 12 were tentatively identified as theobromine and theophylline, respectively. Compounds 13 and 14 gave the same [M + H]+ ion at m/z 197, 16 Da (O) more than that of dimethylxanthine, and were presumed to be oxytheobromine and oxytheophylline, respectively. Similarly, compounds 15 and 16 were assigned as methylxanthine, the demethylated metabolite of theobromine or theophylline. Compared with the corresponding reference standard, compound 17 was explicitly identified as caffeine. It showed an [M + H]+ ion at m/z 195 and fragment ions at m/z 138, 123, and 110 by the successive losses of C2H3NO, CH3, and CO. Given the reported results, compound 18 was plausibly identified as 8-oxocaffeine, the oxidative product of compound 17.
Theobromine and theophylline are isomers with the formula C7H8N4O2, both of which are dimethylated derivatives of xanthine. They can be further oxidized or demethylated, yielding the corresponding dimethyluric acid and monomethylxanthine. Moreover, methylation at either position N-1 of theobromine or position N-7 of theophylline would generate caffeine (1,3,7-trimethylxanthine). Therefore, it can be seen from Table 1 that theobromine and theophylline mainly underwent oxidation, methylation, and demethylation in rats.
Identification of Catechin Derivatives
Catechin is an abundant secondary metabolite in tea. It belongs to the group of flavan-3-ols, a type of natural phenol and antioxidant. 31 In our preliminary study, 8 flavan-3-ols (including catechin) were detected in Ganpu tea. 12 Herein, compounds 19 and 20 both showed an [M + H]+ ion at m/z 467, 176 Da (C6H8O6) more than that of catechin, and gave similar ion signals at m/z 291, 139, and 123 in positive ion mode. Referring to reported results,32,33 compounds 19 and 20 were tentatively identified as catechin-O-glucuronide. Retro-Diels–Alder (RDA) reactions were observed in the MS/MS fragmentation of these two compounds. Taking compound 19 as an example, after the neutral loss of a glucuronyl moiety (GlcUA), a characteristic ion signal at m/z 291 was observed, which was presumed to be protonated catechin. Subsequently, protonated catechin gave a product ion at m/z 139 through an RDA reaction involving the cleavage of bonds 1 and 3 of C ring (Figure 2), while the signal at m/z 123 could be explained by cleavage of bonds 0 and 3. Besides, protonated catechin also yielded a fragment ion at m/z 181 in consequence of the cleavage of bond 5.
Identification of PMF Derivatives
PMFs are kinds of flavonoids bearing two or more methoxy groups on their basic C6-C3-C6 skeleton, with a carbonyl group at position C-4. 34 They vary in the number and position of the hydroxyl groups (-OH) and/or methoxyl groups (-OCH3) on the basic skeleton. PMFs exist almost exclusively in citrus plants, especially in the peels. 35 In fact, we have identified a series of PMFs in CRP and Ganpu tea, such as nobiletin, tangeretin, sinensetin, and natsudaidain.12,24 In this work, 32 PMFs and derived metabolites were detected. As shown in Table 1, these PMFs basically generated the diagnostic fragment ions of [M + H-nCH3]+, [M + H-nCH3-H2O]+, [M + H-nCH3-CO]+, and [M + H-nCH3-H2O-CO]+ in positive ion mode, aligned with reported studies.36,37 Similar MS/MS fragmentation patterns were observed in negative ion mode. For example, compound 39 was assuredly identified as nobiletin by comparison with the corresponding reference standard. Protonated nobiletin displayed its ion signal at m/z 403, and yielded characteristic product ions at m/z 388, 373, 345, and 330 with the successive loss of CH3 and CO.
Based on previous studies, demethylation or hydroxylation is considered to be the major in vivo metabolic pathway of PMFs.38,39 Nobiletin (5,6,7,8,3′,4′-hexamethoxyflavone) is a primary PMF in CPR and has been shown to exert antioxidant, anti-inflammatory, antitumor, and neuroprotective properties.6,40 Herein, compounds 40, 41, and 42 all gave [M + H]+ ions at m/z 389, 14 Da (CH2) less than that of protonated nobiletin, and therefore they were proposed as the mono-demethylated metabolite of nobiletin. Based on the RT reported in literature, 41 compounds 40, 41, and 42 were tentatively assigned as 6-demethylnobiletin, 7-demethylnobiletin, and 4′-demethylnobiletin, respectively. These metabolites could be further demethylated into several dihydroxy-tetramethoxyflavones (compounds 36, 37, and 38). To date, a No. of in vivo biotransformation studies have shown that the number and location of -OCH3 and -OH in PMFs have important effects on their bioactivity. For example, some hydroxylated PMFs were found to exert better anticancer and anti-inflammatory activities than that of their corresponding PMFs.42,43 Therefore, metabolites probably play an indispensable role in the overall bioactivity of phytochemicals. It is important to take corresponding metabolites into consideration when investigating the biological activities of phytochemicals. In addition, these hydroxylated PMFs could further conjugate with either glucuronic acid or sulfuric acid, leading to the corresponding phase II metabolites.
Identification of Flavanone and Flavone Derivatives
Flavanones and flavones are both abundant in citrus fruits. 44 Flavanones are regarded as the first intermediate products of the flavonoid biosynthetic pathway, and are characterized by the presence of a chiral center at C2 and a notable absence of the C2-C3 double bond. 45 The flavanone structure is highly reactive, and is prone to undergo dehydrogenation, yielding corresponding flavones. Flavones are another class of flavonoids based on the backbone of 2-phenylchromen-4-one, with the presence of a C2-C3 double bond. 46
Compared with corresponding reference standards, compounds 53, 54, 59, 62, 64, and 66 were undoubtedly identified as homoeriodictyol, hesperetin, naringenin, quercetin, kaempferol, and apigenin. Compounds 61 and 63 were assigned as isosakuranetin and luteolin, respectively, by comparison with the Natural Products HR-MS/MS Spectral Library (Version 1.0; AB Sciex). Compounds 55 and 56 produced the same [M + H]+ ion at m/z 479, and gave characteristic fragment ions at m/z 303, 285, and 153. Based on reported results, compounds 55 and 56 could be tentatively identified as either hesperetin-O-glucuronide or homoeriodictyol-O-glucuronide. 47 Accordingly, compound 57 and 58 were identified as hesperetin-O-sulfate or homoeriodictyol-O-sulfate. Compound 60 exhibited an [M-H]− ion at m/z 351, 80 Da (SO3) more than that of deprotonated naringenin, and generated product ion signals at m/z 151 and 107. Hence, compound 60 was tentatively identified as naringenin-O-sulfate. Similarly, compounds 65, 67, and 68 were presumed to be kaempferol-O-glucuronide/luteolin-O-glucuronide, apigenin-O-glucuronide, and apigenin-O-sulfate, respectively.
Four flavanone aglycones (eg homoeriodictyol, hesperetin, naringenin, and isosakuranetin) and 4 flavone aglycones (eg quercetin, luteolin, kaempferol, and apigenin) were screened in rat urine and feces samples. These aglycones were further engaged in glucuronidation and sulfation, giving rise to several conjugates. Typical RDA reactions were found in the MS/MS fragmentation of these flavanone and flavone derivatives, which were consistent with published results.48,49 For instance, the characteristic ion at m/z 153 in positive ion mode (m/z 151 in negative ion mode) could be defined as the product of RDA reaction by cleavage of bonds 1 and 3 of the C ring in homoeriodictyol, hesperetin, naringenin, quercetin, luteolin, kaempferol, apigenin, and their derivatives.
Conclusions
In this work, the in vivo metabolite profiles of Ganpu tea were investigated for the first time. Ganpu tea was extracted with boiling water and then orally administrated to rats in a dosage of 3 g/kg. Urine and feces samples were collected and analyzed by high-resolution UHPLC-Q-TOF-MS/MS. As a result, 68 compounds, including 6 caffeic acid derivatives, 1 gallic acid derivative, 3 p-coumaric acid derivatives, 8 xanthine derivatives, 2 catechin derivatives, 32 PMF derivatives, 9 flavanone derivatives, and 7 flavone derivatives, were either identified or tentatively characterized in rat urine and feces. Based on the detected metabolites, prototype compounds derived from GTE were found to undergo extensive phase II metabolism in rats, especially glucuronidation and sulfation. The obtained results should be helpful for further study of the health values of Ganpu tea.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities, Sun Yat-sen University (2022).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
