Abstract
In this work, we prepared a transparent dispersion of fullerene nanoparticles by sonication with anionic phospholipids of 1,2-dipalmitoyl-sn-glycero-3-phosphorylglycerol (DPPG) with fluorescently NBD-labeled 1,2-dipalmitoyl-sn-glycero-3-phosphorylethanolamine (DPPE). Upon incubation of the fullerene nanoparticles with a rat skin, the nanoparticles successfully penetrated the stratum corneum and reached the epidermis.
Fullerenes are carbon cage molecules considered to be three-dimensional analogues of benzene. Buckminsterfullerene (C60) is a representative fullerene with 60 carbon atoms in the spherical structure (Figure 1). In fullerene, there are two types of bonds: C5–C5 single bonds in the pentagons and C5–C6 double bonds in the hexagons. Particular attention has recently been paid to the unique chemical and physical features of C60 as biomaterials. Many fullerene-based compounds with different biological activities have been reported for anticancer or antimicrobial therapy, cytoprotection, enzyme inhibition, contrast- or radioactivity-based diagnosis, and controlled drug delivery.1-8 Especially, one of the promising applications of fullerenes as a biomaterial is in skin care materials since fullerenes, having high antioxidant activity, show good protection against UV radiation if effectively penetrated into skin tissue.1,7 Skin tissue is composed of stratum corneum, epidermis, and dermis. In particular, the 10- to 40-μm-thick stratum corneum, consisting of densely packed cells, provides a barrier to protect the underlying tissue from infection, dehydration, chemicals, and mechanical stress, which prevents permeation of large nanoparticles across the skin tissue. Previously, we reported that hydrophobic bioactive compounds were dispersed by 1,2-dipalmitoyl-sn-glycero-3-phosphorylglycerol (DPPG; Figure 1), an anionic phospholipid, affording small-sized nanoparticles.9,10 Because DPPG having C16 chains can form kinetically stable phospholipid bilayers due to a high phase transition temperature (Tm = 41 °C), DPPG would be useful for the preparation of stable fullerene nanoparticles. Based on the previous study, we here demonstrated transdermal delivery of DPPG nanoparticle containing fullerene. By using a fluorescent 4-chloro-7-nitrobenzo-2-oxa-1,3-diazole (NBD)-labeled DPPG (Figure 1), we successfully visualized that the nanoparticle passed through the stratum corneum and reached the epidermis.

Molecular structures of fullerenene (C60) and phospholipids of DPPG and fluorescently-labelled DPPE-NBD used in this work.
First of all, we prepared fluorescent FLDPPG-fullerene nanoparticles containing NBD-labeled DPPE for visualization of subcutaneous permeability (Figure 2a). NBD is a typical non-ionic dye showing a green fluorescence with enough quantum yield. 11 Typically, DPPG (4.85 wt%), DPPE-NBD (0.15 wt%), and fullerene powder (0.1 wt%) were dispersed in water, and sonicated at 100 W for 2 min, and then cooled to room temperature. Visual observation of the prepared sample showed a transparent aqueous solution (Figure 2c) with dispersed fullerene, which was clearly different when fullerene was precipitated without the phospholipids (Figure 2b). Dispersibility of FLDPPG-fullerene was similar to that dispersed with DPPG (Figure 2d). We also confirmed that C70 fullerene could be dispersed by DPPG (Figure 2e). Next, we carried out transdermal delivery of FLDPPG-fullerene nanoparticles (Figure 3a). For this purpose, the nanoparticle samples were incubated with rat skin tissue placed on a Franz diffusion cell, and a section was prepared from the skin samples after 24 hours for fluorescent microscopy. Surprisingly, fluorescence derived from DPPE-NBD was observed in the epidermis, as well as in the stratum corneum of the skin tissue (Figure 3b and c). In this study, we did not directly observe fullerene, but the fluorescent DPPE-NBD nanoparticle because fluorescence labeling would largely affect the aggregation behavior of the fullerene molecule. However, considering the fact that fullerene is well dispersed by amphiphilic DPPG and DPPE-NBD, we can expect that fullerene also penetrates into the epidermis.
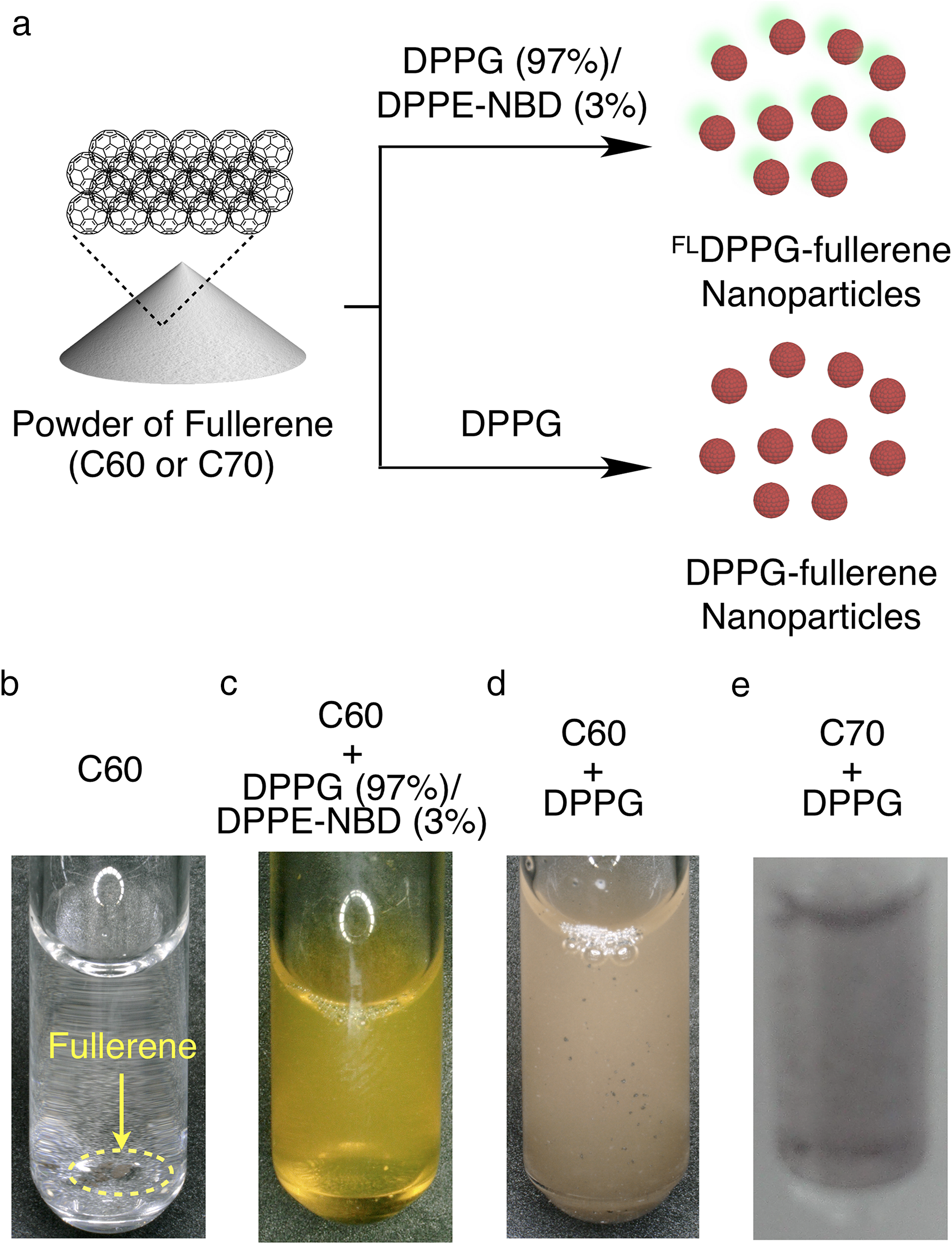
(a) Synthetic illustrations for the preparation of fluorescent and non fluorescent fullerene nanoparticles. Photograph of fullerene (C60) (b), fluorescent fullerene (C60) nanoparticle dispersed with DPPG and DPPE-NBD ([DPPG]/[DPPE-NBD] = 97/3) (c), or dispersed with DPPG (d). Photograph of fullerene (C70) dispersed with DPPG.

Synthetic illustrations for transdermal delivery of FLDPPG-fullerene nanoparticle (a). Bright field (b) and fluorescence image (c) of a sliced of rat skin tissue 24 hours after incubation with FLDPPG-fullerene nanoparticle.
In this study, we have demonstrated a transdermal delivery of FLDPPG nanoparticles containing fullerene. FLDPPG-fullerene composite nanoparticles could be prepared by heating followed by cooling of aqueous mixtures of DPPG, DPPE-NBD, and fullerene. Upon addition to rat skin tissue, FLDPPG-fullerene nanoparticles reached the epidermis of the skin tissue. Although fullerene is a well-known anti-oxidant, it has still been challenging to design fullerene-based skin care materials because of difficulty in transdermal delivery. FLDPPG-fullerene nanoparticles with skin permeability demonstrated in this study could be a new candidate not only for skin care materials, but also as reagents for phototherapy of diseased tissues such as cancer. 12
Experimental
Footnotes
Author Contributions
N.U and M.Y. performed all experiments and H.H. designed and wrote the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
