Abstract
Tyrosinase ultimately controls the melanogenesis rate of the skin, and tanning and haircare products generally induce the activation of tyrosinase. Moreover, various enzymes, including tyrosinase, tyrosinase-related protein 1 (TRP1), and tyrosinase-related protein 2 (TRP2), mediate melanogenesis in which microphthalmia-associated transcription factor (MITF) is a master regulator. One coumarin family member 7,8-dihydroxy-4-methylcoumarin (DHMC) shows extensive biological activities with beneficial health effects; however, it also induces cytotoxicity and its melanogenic effect has not been reported yet. Therefore, we first synthesized DHMC derivatives via methylation to obtain 7,8-dimethoxy-4-methylcoumairn (DMMC), and investigated the pro- or anti-melanogenic effects of DHMC and DMMC in B16-F10 melanoma cells as well as the underlying mechanism. DHMC showed cytotoxicity at all tested concentrations, whereas DMMC did not reduce cell viability, even at the high concentration. DMMC also drives the significant increase in intracellular melanin and tyrosinase activity. Moreover, DMMC induced MITF expression by significantly increasing tyrosinase activity, which activates the gene expression of TRP1 and TRP2. Western blotting confirmed that DMMC induced the activation of mitogen-activated protein kinase (MAPK) signaling by the phosphorylation of C-Jun N-terminal kinase (JNK), resulting in the increased melanin production and the decreased phosphorylation of protein kinase B. Collectively, this study showed the pro-melanogenic effect of DMMC and its potential as a safe tanning and dyeing agent.
Keywords
Introduction
Melanin is a polymer that is naturally present in animal and plant systems, which is a pigment responsible for skin color and is comprised of indole and other tyrosine-derived intermediates. Melanogenesis is initiated upon exposure to internal and external stimulators such as ultraviolet (UV) rays, chemicals, and bacteria, which occurs in the melanosomes and organs specialized for melanocytes in the skin.1,2 The expression of melanin is regulated by various cellular mechanisms and the pigment is then transmitted to the surrounding keratinocytes. Melanin can be classified into pheomelanin and eumelanin, with the latter largely responsible for determining the color of mammals.
Three main enzymes (tyrosinase, tyrosinase-related protein-1 [TRP1], and tyrosinase-related protein-2 [TRP2]) play a central role in melanin synthesis. L-tyrosine is a required precursor in the early stages of melanin synthesis, during which it is converted into dihydroxy-l-phenylalanine (l-DOPA) through a hydroxy reaction by tyrosinase. DOPA quinone, an oxidation form of l-DOPA to form DOPA chrome, is subsequently converted to 5,6-dihydroxy-2-indolylcarboxylic acid (DHICA) by TRP2. 3 Ultimately, indole 5,6-quinone-carboxylic acid is generated from DHICA by TRP1, result in the production of brown/black pigment eumelanin.6,7 Tyrosinase is a direct inhibitor of the synthesis of eumelanin and pheomelanin, whereas TRP1 and TRP2 are deeply involved in the synthesis of eumelanin. 8 In mice, it was reported that TRP1 oxidize 5,6-dihydroxyindole-2-carboxylic acid to produce indole-5,6-quinone-2-carboxylic acid, 9 although the activity of this enzyme differs in humans.
Previous studies in B16-F10 melanoma cells have shown that microphthalmia-associated transcription factor (MITF) expression has a strong correlation with the phenotypes of melanogenic enzymes, thereby contributing to melanin production.10,11 Upon stimulation by UV irradiation, intracellular α-melanocyte-stimulating hormone (α-MSH) is produced and then binds to melanocortin receptor 1 (MC1R) to activate adenylated acid cyclase (AC) that in turn increases cyclic adenosine monophosphate (cAMP) levels. 12 MITF expression is further augmented by the increased phosphorylation of cAMP response element-binding protein (CREB) and protein kinase A (PKA). 13 Recent studies revealed that the three serine/threonine kinases of mitogen-activated protein kinases (MAPKs) such as p38 MAPK, extracellular signal-regulated kinase (ERK), and C-Jun N-terminal kinase (JNK), are involved in melanin production by regulating MITF expression.14,15 Specifically, ERK ubiquitination and phosphorylation induces the degradation of MITF, which then attenuates the expression of melanogenesis enzymes (tyrosinase, TRP1, and TRP2). Furthermore, ERK is known to negatively modulate the expression of MITF, indicating that several signals, including phosphatidylinositol 3-kinase (PI3 K)/protein kinase B (AKT) are associated with melanogenesis by regulating MITF expression.16,17
Tyrosinase is an enzyme that limits the melanogenesis rate. The activator of tyrosinase is used as a tanning agent and in haircare products. However, since hair coloring agents can lead to adverse side effects, 18 identifying nontoxic melanogenic agents has become a point of interest for the development of new functional cosmetics and treatments. Coumarin and derivatives belong to a group of colorless crystalline oxygen heteropolycyclic compounds which was initially extracted from plants.19,20 In addition, coumarin is thought to exert various biological properties, such as anti-inflammatory,21,22 antiviral, 23 anti-coagulant, 24 antimicrobial,25,26 and enzyme inhibition27,28 effects. In particular, one member of the coumarin family, 7,8-dihydroxy-4-methylcoumarin (DHMC), has been used to synthesize diacetoxy derivatives. 29 DHMC has been reported to have anti-leukemic, 30 anti-inflammatory, 31 and anti-lung cancer 32 effects; however, the exact role of DHMC in melanogenesis had not been studied to date. Here, a study on the melanogenesis properties of DHMC was conducted, but it was determined that melanin could not be produced due to cytotoxicity. Therefore, DHMC derivatives were synthesized to discover new melanin-induced activators. Specifically, we applied a methylation method to DHMC, which is the process of chemically substituting hydroxyl groups with methyl groups in compounds, forming a methyl ester bond in the derivative 7,8-dimethoxy-4-methylcoumarin (DMMC). We then investigated the effects of the synthesized DMMC on various melanin-producing activities in B16-F10 melanoma cells.
Results
Methylation of DHMC
As described below, DHMC was subject to methylation process. The obtained methylated reaction product was purified using preparative high-performance liquid chromatography (HPLC), and its yield is over about 90% (Figure 1a). In addition, as shown in the mass spectrum in Figure 1(b), the tallest peak at m/z 193 was identified as that of ionized DHMC, and it showed that an [M + H+] peak at m/z 221 was that of DMMC.

(a) HPLC chromatogram of 7,8-dihydroxy-4-methylcoumarin (DHMC) and 7,8-dimethoxy-4-methylcoumarin (DMMC). (b) Structure and mass analysis of DHMC and DMMC.
1H nuclear magnetic resonance (NMR) analyses further confirmed that the methylated product was DMMC, based on comparison with NMR data from a previous study in which DMMC was synthesized with the same approach. 33 7,8-dimethoxy-4-methylcoumarin (DMMC, 1): 1H NMR (DMSO-d6, 500 MHz): δ 7.49 (1H, d, J = 8.9 Hz, H-5), 7.12 (1H, d, J = 9.0 Hz, H-6), 6.24 (1H, s, H-3), 3.91 (3H, s, 8-OMe), 3.81 (3H, s, 7-OMe), and 2.40 (3H, s, 11) (see Supplemental Material).
Effects of DHMC and DMMC on the Viability of B16-F10 Melanoma Cells
The level of cell viabilities relative to non-treated controls was measured and compared using an 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) assay 34 and from which the appropriate treatment concentrations of DHMC and DMMC with no significant cytotoxicity were chosen. The cell viability was 78%, 38%, and 36% in the concentration of 50, 100, and 200 μM DHMC, respectively, compared with that of the corresponding controls (Dulbecco's modified eagle medium [DMEM]); 0.2 μM α-MSH was used as a positive control. By contrast, DMMC was nontoxic to B16-F10 cells at these concentrations with no decrease in viability (Figure 2). These results demonstrated that changes in methylation contributed to improving the safety of DHMC with no cytotoxicity.

Effect of DHMC and DMMC on cell viability determined by the MTT assay. Cells were treated with different concentrations (50, 100, and 200 μM) of DHMC and DMMC for 72 h. The values represent the mean ± SD of three repeated independent experiments performed.
Comparison of Effects of DHMC and DMMC on Melanin Production
In B16-F10 melanoma cells without α-MSH treatment, DHMC did not impact the synthesis of melanin due to its cytotoxicity, whereas DMMC increased melanin production over the increasing order of DMMC concentration (Figure 3). Based on this result, we further focused on the synthesized DMMC that caused the most significant change in melanin production.

Effects of DHMC and DMMC on melanin production in B16-F10 melanoma cells treated with or without α-MSH (0.2 μM; positive control). Cells were treated with different concentrations (50, 100, and 200 μM) of DHMC and DMMC for 72 h. The values represent the mean ± SD of three repeated independent experiments performed.
Effects of DHMC and DMMC on Tyrosinase Activity
Increased tyrosinase expression and activities are an indicator for the increased production of melanin. The activity of tyrosinase was enhanced by 242.8%, 345.1%, and 442.8% after DMMC treatment at 50, 100, and 200 μM, respectively, relative to the untreated control (Figure 4). These results indicated that DMMC treatment enhances melanogenesis in accompany by the increase of tyrosinase activity in B16-F10 melanoma cells. This posed DMMC as a potential dyeing agent.

Effects of DHMC and DMMC on intercellular tyrosinase activity in B16-F10 melanoma cells treated with or without α-MSH (positive control). The cells were treated with different concentrations (50, 100, and 200 μM) of DHMC and DMMC for 72 h. The values represent the mean ± SD of three repeated independent experiments performed.
DMMC Induces the Upregulation of Melanogenic Enzymes and MITF
The transcription factor MITF regulates melanogenic enzymes involved in melanogenesis, such as tyrosinase, TRP1, and TRP2. Relative to that of the untreated cells, MITF protein expression was increased in the increasing order of DMMC concentration at 48 h post-DMMC (Figure 5a). Similarly, the expression levels of other melanogenic proteins were increased after DMMC treatment for 48 h in a concentration-dependent manner relative to untreated cells (Figure 5b to d). These data suggest that DMMC treatment upregulated TRP1, TRP2, and tyrosinase production by increasing MITF expression.
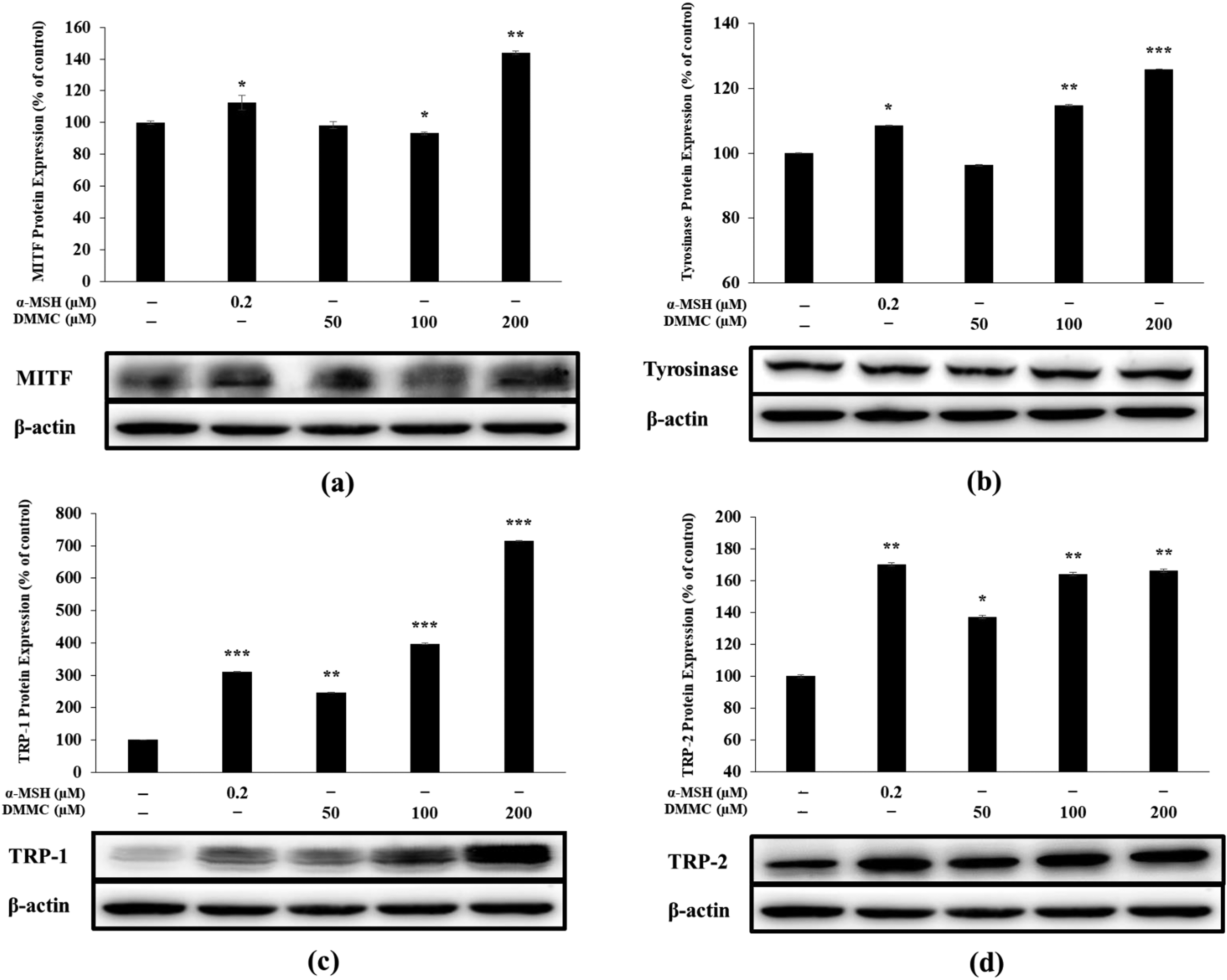
Effect of DMMC on (a) MITF, (b) tyrosinase, (c) TRP1, and (d) TRP2 expression in B16-F10 melanoma cells. Cells were treated with different concentrations of DMMC (50, 100, and 200 µM) for 48 h. Protein levels were measured through western blotting. The values represent the mean ± SD of three repeated independent experiments performed in duplicate.
DMMC Influences the AKT Signaling Pathway in B16F10 Melanoma Cells
We next focused on its effects on molecules in the AKT signaling pathway, which has been reported to regulate the pigmentation process.16,17 Western blotting showed a reduction of AKT phosphorylation after DMMC treatment to melanoma cells in a concentration-dependent manner and its maximum reduction was achieved at 200 μM (Figure 6). This result indicated that DMMC distinctly enhances melanogenesis by significantly reducing AKT phosphorylation.

Effects of DMMC on the phosphorylation of AKT. B16-F10 cells were treated with DMMC for 3 h at the indicated concentrations and western blotting was performed. The values represent the mean ± SD of three repeated independent experiments performed in duplicate.
DMMC Influences the MAPK Pathway in B16F10 Melanoma Cells
Western blotting showed that JNK protein expression levels increased with the increase in DMMC concentration (Figure 7a). However, DMMC decreased the expression level of ERK, and p38 (Figure 7b and c).

Effect of DMMC on the phosphorylation of (a) JNK, (b) p38, and (c) ERK. B16-F10 cells were treated with DMMC at the indicated concentrations for 3 h. The values represent the mean ± SD of three repeated independent experiments performed in triplicate.
Discussion
The skin plays a key role in body protection against potentially harmful external stimulations. Melanin creates a barrier, for example, against the penetration of UV rays, and determines the color of the skin, hair, and eyes. In this study, we allowed DHMC to undergo methylation process and obtained the black compound DMMC. The structure was then confirmed by HPLC, ESI-MS, and NMR analyses. In contrast to DHMC, DMMC exhibited significantly enhanced melanin production in B16-F10 melanoma cells without significant cytotoxicity. The increased melanin content was accompanied by the increase in DMMC concentration, and also increased the activity and/or protein expression level of the melanogenesis enzymes TRP1, TRP2, and tyrosinase, and MITF a master regulator in melanogenesis. Based on previous studies,35,36 AKT signaling pathway was shown to play a role in melanin production in B16-F10 cells,16,17 and reduced AKT phosphorylation increases MITF expression by inducing CREB phosphorylation. 37 Therefore, our results indicate that DMMC increases MITF expression through inhibition of AKT phosphorylation, thus inducing melanogenesis. Specifically, these results suggest that DMMC impacts the MAPK signaling pathway mainly and increased JNK phosphorylation may bind to the promoter region of MITF gene and boost MITF gene expression, in turn increasing the protein expression of tyrosinase, TRP1, and TRP2, and ultimately inducing melanin production.
Overall, DHMC showed cytotoxicity, whereas DMMC did not show cytotoxicity and increased melanin production. In this study, DMMC-induced melanogenesis occurred through increased phosphorylation of JNK, ERK, and p38, while decreasing AKT phosphorylation. Therefore, the results of this study suggest the potential use of DMMC as a tanning or pigmentation agent in cosmetics. However, animal studies need to be followed to ensure the safety and assess any arising side effects.
Materials and Methods
Methylation of DHMC
All chemicals were purchased from Sigma-Aldrich (St. Louis, MA, USA). In a typical methylation experiment, DHMC (50 mg) in dimethyl carbonate (4 mL) and 1,8-diazabicyclo[5.4.0]undec-7-ene (0.6 mmol, 10 mL) was heated at 110 °C overnight with magnetic stirring. Next, methanol (3 mL) was added and completely mixed by shaking. The mixture was then subject to evaporation. The resulting residues were dissolved in 1N HCL (5 mL) and ethyl acetate (10 mL) solvents. Finally, the ethyl acetate layer was vacuum-dried for 2 h at 40 °C.
HPLC Analysis and Purification of DMMC
Using Shimadzu SpectroMonitor 3200 digital UV-Vis detector coupled with a Shim-pack GIS 0.5 mm ODS C18 column (250 × 4.5 mm) (Shimadzu, Kyoto, JP), HPLC chromatograms were analyzed. The two degassed mobile phases are double distilled water supplemented with 0.1% trifluoroacetic acid (solvent A) and acetonitrile (solvent B). The overall percentage of solvent B was gradiently changed from 10% to 100% for 30 min with a flow rate of 1 mL/min.
Liquid Chromatography–Mass Spectrometry (LC-MS) and NMR Analysis of DMMC
The LC system, Perkin-Elmer API (Norwalk, CT, USA) single-quadrupole MS, composed of an 1100 HPLC pump and 1100 HPLC Agilent Autosampler (Agilent Technologies) with an 1100 HPLC Agilent DAD Detector (220 to 500 nm). A reversed-phase column (Acquity BEH C18 2.1 mm × 100 mm [1.7 μm] was used to separate the samples. ESI/MS detection of the samples: positive ion mode ESI+, acquisition range: 50-1, m/z 400, capillary voltage: 2.5 kV, cone voltage: 30 V, source temperature: 120°C, desolvation gas temp: 600°C, cone gas flow: 20 L h−1, desolvation gas flow: 800 L h−1. Analysis software MassLynx version 4.1 [Waters Corp.]. NMR spectra were generated using a VNMRS 500 NMR spectrometer [Agilent Technology] with a comparison of the residual solvent peak [DMSO-d6, 500 MHz] of the reference peak from deuterated NMR solvents.
Cell Culture and Viability Assay
B16-F10 melanoma cells get from ATCC (American Type Culture Collection, Manassas, VA, USA) were cultured in phenol red-free DMEM supplemented with 10% v/v heat-inactivated fetal bovine serum (FBS, Welgene, Fresh MediaTM, Gyeongsang, Korea) and 1% v/v penicillin and streptomycin (Welgene, Fresh MediaTM, Gyeongsang, Korea) at 5% CO2, 37 °C. The cells (1.0 × 104 cells/well) were seeded into 24-well plates and incubated 24 h. Three different concentrations of DHMC and DMMC (50, 100, and 200 µM) or α-MSH (0.2 μM) were applied to the cells and incubated for 72 h. To assess cell viabilities, 1 mg/mL MTT reagent (VWR Life Science) was added to each well of the plates and incubated for 3 h. After the complete removal of culture medium and the remaining formazan crystals were solubilized in dimethyl sulfoxide (DMSO). Absorbance was read at 570 nm on a microplate reader (Thermo Fisher Scientific).
Measurement of Melanin Content
B16-F10 melanoma cells were seeded (4.0 × 104 cells per well in 6-well plates) and cultured for 24 h. DHMC and DMMC (50, 100, and 200 µM) were added at different concentrations in each well and cultured for 72 h with α-MSH (0.2 μM) as the positive control. To quantify intracellular melanin content, the spent medium was completely aspirated off followed by washing with phosphate-buffered saline. The resulting cell sediments were lysed in 1 mL of 1 M NaOH (added with 10% DMSO) at 90 °C for 1 h. Finally, the absorbance at 405 nm was read as described above.
Assessment of Intracellular Tyrosinase Activity
B16-F10 melanoma cell culture and sample treatments were made as described above. After culture was completed, the cells were trypsinized and pelleted by centrifugation at 848 g, 3 min. The proteins were isolated from the cells using 1% protease inhibitor cocktail-added 1X RIPA cell lysis buffer (Biosesang). The protein extraction process was facilitated by vortexing six times every 10 min, and then spun down at 15,928 g for 30 min, −4 °C. The samples were equalized by solvating l-DOPA-(phenyl-d3) using sodium phosphate buffer (0.1 M, pH 6.8). l-DOPA (80 μL, 10.14 mM) and 20 μL of each lysate were mixed, and the reaction continued for 2 h at 37 °C on a shaker (100 rpm). The experiment was repeated in triplicate and the absorbance was measured at 490 nm as described above.
Western Blotting
The western blotting procedures and primary and secondary antibodies used in this study were previously published. 37 B16-F10 melanoma cells seeded and cultured for 72 h as described above. The cells were treated with three different concentrations of DMMC (50, 100, and 200 µM). The DMMC-induced melanin production was compared against α-MSH (0.2 μM) only-treated positive control. Proteins were extracted in RIPA buffer containing 1% protease inhibitors. The lysates were spun down at 15,928 g at −4 °C for 30 min. Protein amounts present in the supernatants were quantified using PierceTM BCA Protein Assay Kit (Thermo Fisher Scientific). The supernatant was mixed with 2X Laemli sample buffer to prepare a sample for western blotting. After heating at 90 °C for 5 min, and 20 μL of the sample was loaded onto a 10% sodium dodecyl sulfate gel for electrophoresis (SDS-PAGE) to separate proteins according to their size. The SDS-PAGE was subject to 110 V for 2 h and electrophoretically transferred to a polyvinylidene difluoride membrane (PVDF). The blotted PVDF membrane was then blocked with 5% skim milk in Tris-buffered saline with Tween (TSBT) buffer with 1 h shaking. Washed 6 times for 10 minutes using TBST buffer. The membranes were incubated overnight at 4 °C with primary antibodies. After that, washed with TBST buffer six times 10 min each. Secondary Antibody incubation for 2 h with the anti-mouse IgG or anti-rabbit IgG (Cell Signaling, USA) antibodies. Target proteins were visualized using an enhanced chemiluminescence kit (Bio-Rad) and band intensities were quantified with the aid of ImageQuant 1.3 (LAS-4000 mini, Fujifilm, Tokyo, Japan).
Statistical Analysis
All experiments were conducted three times, and the results were presented as the mean ± standard errors from triplicate experiments. The significant difference from the untreated group: *P < .05, **P < .01, ***P < .005.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221076647 - Supplemental material for Synthesis and Melanogenesis Effect of 7,8-Dimethoxy-4-Methylcoumarin via MAPK Signaling-Mediated Microphthalmia-Associated Transcription Factor Upregulation
Supplemental material, sj-docx-1-npx-10.1177_1934578X221076647 for Synthesis and Melanogenesis Effect of 7,8-Dimethoxy-4-Methylcoumarin via MAPK Signaling-Mediated Microphthalmia-Associated Transcription Factor Upregulation by Min-Sung Kang, Sung-Chan Jang, Taejin Park, Min-Seon Kim, Jin-Soo Park, Won-Jae Chi and Seung-Young Kim in Natural Product Communications
Footnotes
Acknowledgments
National Institute of Biological Resources (NIBR), funded by the Ministry of Environment (MOE) of the Republic of Korea (NIBR202102109) granted funding for this studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Biological Resources (NIBR), funded by the Ministry of Environment (MOE) of the Republic of Korea (NIBR202102109) granted funding for this studies (grant number NIBR202102109).
Ethical Approval
In applicable for this article.
Statement of Human and Animal Rights
This study contains no human or animal subjects.
Statement of Informed Consent
Informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
