Abstract
Guiera senegalensis J.F. Gmel. (Combretaceae) is commonly used as a traditional medicine in Africa. In our previous study, the hydro-alcoholic extracts of the leaves of G. senegalensis showed potent free radical scavenging and enzyme inhibitory activities. Detailed chemical analysis of the 70% ethanol extract afforded 8 phenolic compounds including myricetin (
Guiera senegalensis J.F. Gmel. (Combretaceae) is a shrub commonly distributed in African countries including Senegal, Guinea, Mali, 1 Nigeria, 2 Burkina Faso, 3 and Sudan. 4 Traditionally, it is used in the treatment of cold, bronchitis, fever, 1 diabetes, vomiting, stomach pain, jaundice, kidney stones, and also as a tonic. 4,5 In our previous study, we analyzed the effect of extraction solvents on the antioxidant and enzyme inhibitory activities for 6 medicinal plants from Sudan and found that the hydro-ethanolic extract of the leaves of G. senegalensis showed potent activities. 6 In continuation to that, we herein report the isolation and identification of potent bioactive phenolic compounds from the leaves of G. senegalensis.
The leaves of G. senegalensis collected from Kordofan State, Sudan, were extracted twice with 70% EtOH. The combined extract was dried under reduced pressure and then subjected to repeated column chromatography (CC) on MCI gel CHP20P, Sephadex LH20, ODS, and silica gel to isolate 8 pure compounds (

Structures of compounds isolated from the leaves of G. senegalensis.
All of these isolated compounds were evaluated for their 1,1-diphenyl-2-picrylhydrazyl (DPPH) free radical scavenging activity, α-glucosidase and pancreatic lipase inhibitory activities. The IC50 values of active compounds for all these assays are given in Table 1. All compounds showed potent free radical scavenging activities and among them, compound
IC50 (μM) a Values of Compounds Isolated From G. senegalensis
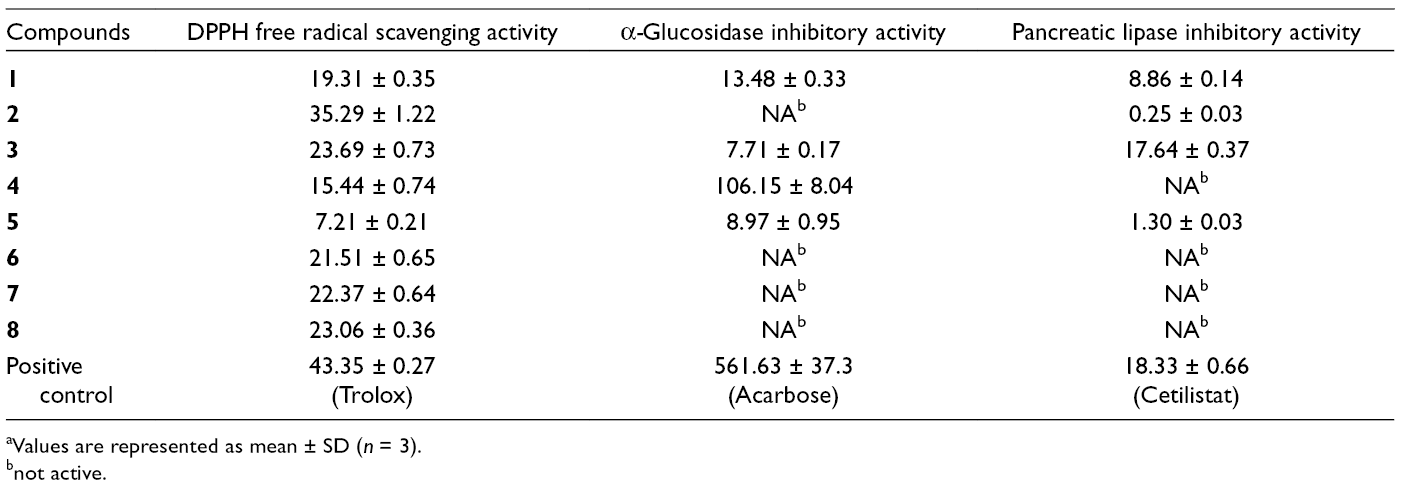
aValues are represented as mean ± SD (n = 3).
bnot active.
Experimental
General Experimental Procedures
Optical rotations, JASCO DIP-1000KUY polarimeter; NMR, BRUKER AVANCE 600 spectrometer; CC, silica gel 60 (0.040-0.063 mm, Merck KGaA), MCI gel CHP20P (75-150 µm, Mitsubishi Chemical Industries, Japan), Sephadex LH-20 (Amersham Pharmacia Biotech, Tokyo, Japan), and Chromatorex ODS (30-50 µm, Fuji Silysia Chemical Co., Ltd., Aichi, Japan). Thin layer chromatography was performed on a precoated silica gel 60 F254 (0.2 mm, aluminum sheet, Merck KGaA, Darmstadt, Germany).
Chemicals
1,1-Diphenyl-2-picrylhydrazyl, enzymes α-glucosidase (from Saccharomyces cerevisiae), and porcine pancreatic lipase were purchased from Sigma Aldrich, Co. (Tokyo, Japan). 6-Hydroxy-2,5,7,8-tetramethylchromancarboxylic acid (Trolox) and acarbose were from Wako Pure Chemical Industries, Ltd (Tokyo, Japan), and 2-(N-morpholino)ethanesulfonic acid buffer was purchased from Dojindo Chemical Research, Kumamoto, Japan. 4-Nitrophenyl-α-d-glucopyranoside was purchased from Nacalai Tesque, Inc., Kyoto, Japan. Cetilistat was obtained from Combi-Blocks, San Diego, USA.
Plant Material
The leaves of G. senegalensis were collected from Kordofan state in March 2017. The plant species was identified by Mr. Yahya Sulieman Mohamed, Taxonomist, Medicinal, Aromatic Plants and Traditional Medicine Research Institute (MAPTRI), Khartoum, Sudan. The voucher specimen of the plant species (Voucher No.: MPRTMI-H G/2/82) was deposited at the Herbarium of MAPTRI, Khartoum, Sudan.
Extraction and Isolation
The leaves of G. senegalensis were shade dried for 2 weeks. The dried leaves (2.6 kg) were extracted twice with 70% EtOH (22 L, 48 hours). The filtered extracts were combined and evaporated under reduced pressure to give 586.2 g of extract. The extract was then subjected to MCI gel CHP20P CC and eluted successively with water, 40%, 70% and 100% MeOH to give 6 fractions (Fr. 1-Fr. 6). Fraction 2 (256.7 g, water eluate), and fraction 3 (46.6 g, water eluate) were combined and subjected to MCI gel CHP20P CC and eluted with water, 10%, 20%, 30%, 40%, 70% and 100% MeOH to obtain 9 fractions (SubFr. 2-1-SubFr. 2-9). The subtractions 2-3 (7.8 g, water eluate) and 2-4 (17.8 g, 10% MeOH eluate) were combined and subjected to Sephadex LH-20 CC (water, 50% and 100% MeOH) to obtain 7 subfractions (SubFr. 2-3-1-SubFr. 2-3-7). Subfraction 2-3-3 (8.3 g, water eluate) was subjected on ODS CC (water, 10%, 20%, 50%, 100% MeOH) to obtain compound
Optical Rotations
Myricitrin (
Evaluation of Bioactivities
The DPPH radical-scavenging activity, α-glucosidase inhibitory and pancreatic lipase inhibitory activities were performed as reported previously. 6
Supplemental Material
Supplementary Data - Supplemental material for Phenolic Compounds as Potent Free Radical Scavenging and Enzyme Inhibitory Components From the Leaves of Guiera senegalensis
Supplemental material, Supplementary Data, for Phenolic Compounds as Potent Free Radical Scavenging and Enzyme Inhibitory Components From the Leaves of Guiera senegalensis by Amina Ibrahim Dirar, Anjana Adhikari-Devkota, Md. Mahadi Hassan, Mikiyo Wada, Takashi Watanabe, and Hari Prasad Devkota in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to acknowledge the JSPS RONPAKU Program for fellowship support to Ms Amina Ibrahim Dirar. We are also grateful to Mr Yahya Sulieman Mohamed, Taxonomist, Herbarium, Medicinal, Aromatic Plants and Traditional Medicine Research Institute (MAPTRI) Sudan, for the identification of the plant.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Program for Leading Graduate Schools, Health Life Science: Interdisciplinary and Glocal Oriented (HIGO) Program, MEXT, Japan and Program for Building Regional Innovation Ecosystems at Kumamoto University, Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
