Abstract
Angelica sinensis Radix (ASR), one of the most commonly used traditional Chinese medicines, contains many chemical components such as polysaccharides, volatile oil, flavonoids, amino acids, and organic acids, among which polysaccharides play an indispensable role in the therapeutic effect of ASR. A. sinensis polysaccharide (ASP) has many biological activities, for instance, hematopoietic, anti-tumor, and liver protection, which are closely related to the treatment of human diseases such as chronic anemia, leukemia, and diabetes. In addition, there are excellent application prospects for drug delivery in nanoparticles. This paper reviews the chemical compositions, extraction methods, biological activity, action mechanism, potential clinical applications, nanoparticles, and research prospect of ASP from 2010 to 2020, so as to provide references for its further development.
Angelica sinensis radix (ASR), the dried roots of Angelica sinensis (Oliv.) Diels (Apiaceae), has been widely used as a tonic to treat various diseases in China for thousands of years. 1,2 ASR has been reported to have a host of biological activities, such as activating blood circulation and replenishing blood, delaying senescence, and fighting cancer. 3,4 ASR contains more than 70 components, including polysaccharides, organic acids, volatile oils, and flavonoids. Among them, Angelica sinensis polysaccharides (ASP) are the primary components, which play an important role in the blood enrichment properties of ASR. 5 In addition, ASP have antioxidant, blood circulation invigorating, anti-tumor, hepatoprotective, anti-aging, and anti-inflammatory effects, as well as excellent drug delivery and immunomodulatory properties, which offer considerable research and application potential. 6,7 Jin et al. mainly reviewed the extraction and purification technology, structural characterization, and biological activity of ASP before 2010. 8 Therefore, this paper provides a comprehensive overview summarizing the chemical composition, extraction methods, biological activity, action mechanisms and applications of ASP from 2010 to 2020.
Search Strategy
For conducting a preliminary search, several databases were searched from 2010 to 2020, including Science Direct (Elsevier), PubMed Central, Web of Science, and Google Scholar to retrieve published studies regarding ASP. The following search keywords were used: (“Angelica sinensis polysaccharides”) and (“effects” OR “bioactivities” OR “structure” OR “extract” OR "“isolate”). First of all, the title and abstract of the obtained papers were screened. Then, the full text of the papers that passed the primary selection were screened again.
Chemical Composition and Characterization
The powdered roots were extracted with water. To the aqueous extract, ethanol was added and the precipitate obtained was dialyzed, freeze-thawed, and lyophilized to obtain crude polysaccharide. This was packed into a DEAE-Sephadex A-25 column (90 cm × 5 cm), which was eluted first with water, and then 0.3 M and 0.5 M NaCl in turn. The 0.5 M NaCl eluate was collected, dialyzed, and freeze-dried to obtain APS-3. This was further purified on a Sephacryl S-400 column (100 cm × 3.5 cm), eluting with 0.1 M NaCl, to obtain 3 sub-fractions, which were treated on Sephadex G100 (100 cm × 3.5 cm) columns. Three acidic polysaccharides, APS-3a, APS-3b and APS-3c, were obtained. Their sugar content was 63.7%, 67.9% and 61.0%, uronic acid content 25.1%, 30.3%, and 35.7%, and molecular weights 5.9 × 105, 2.3 × 105, and 1.4 × 104 Da, respectively. APS-3a and APS-3b were composed of glucose, galactose, arabinose, rhamnose, and mannose with molar ratios of 3.2:1.7:2.5:1.3:1.0 and 2.3:5.4:6.8:1.0:1.2, respectively, while APS-3c was composed of glucose, galactose, arabinose, rhamnose, mannose, and xylose with a molar ratio of 6.3:4.7:6.7:6.5:1.6:1.0. 3
Sliced roots were extracted with water, the pH of the extract was adjusted to 5-6, and then the crude polysaccharide was obtained by ethanol precipitation. The crude polysaccharide was then dissolved in water, freeze-thawed, filtered, and eluted with water on a Sephadex G-50 (80 cm × 3 cm) column. Mu et al. finally extracted an acidic heteropolysaccharide with a molecular weight of 8 × 104 Da, which was composed of glucuronic acid, glucose, arabinose, and galactose with molar ratios of 1.00:1.70:1.85:5.02. Its backbone was composed of 1, 3-linked Galp, 1, 6-linked Galp, and 2-OMe-1, 6-linked Galp with 3 branches connected to O-3 of 2-OMe-1, 6-linked Galp, and the ends are GlcpA and Araf; all the Araf and most of the Glcp were distributed in branched chains. The chemical structure is shown in Figure 1(A). 6
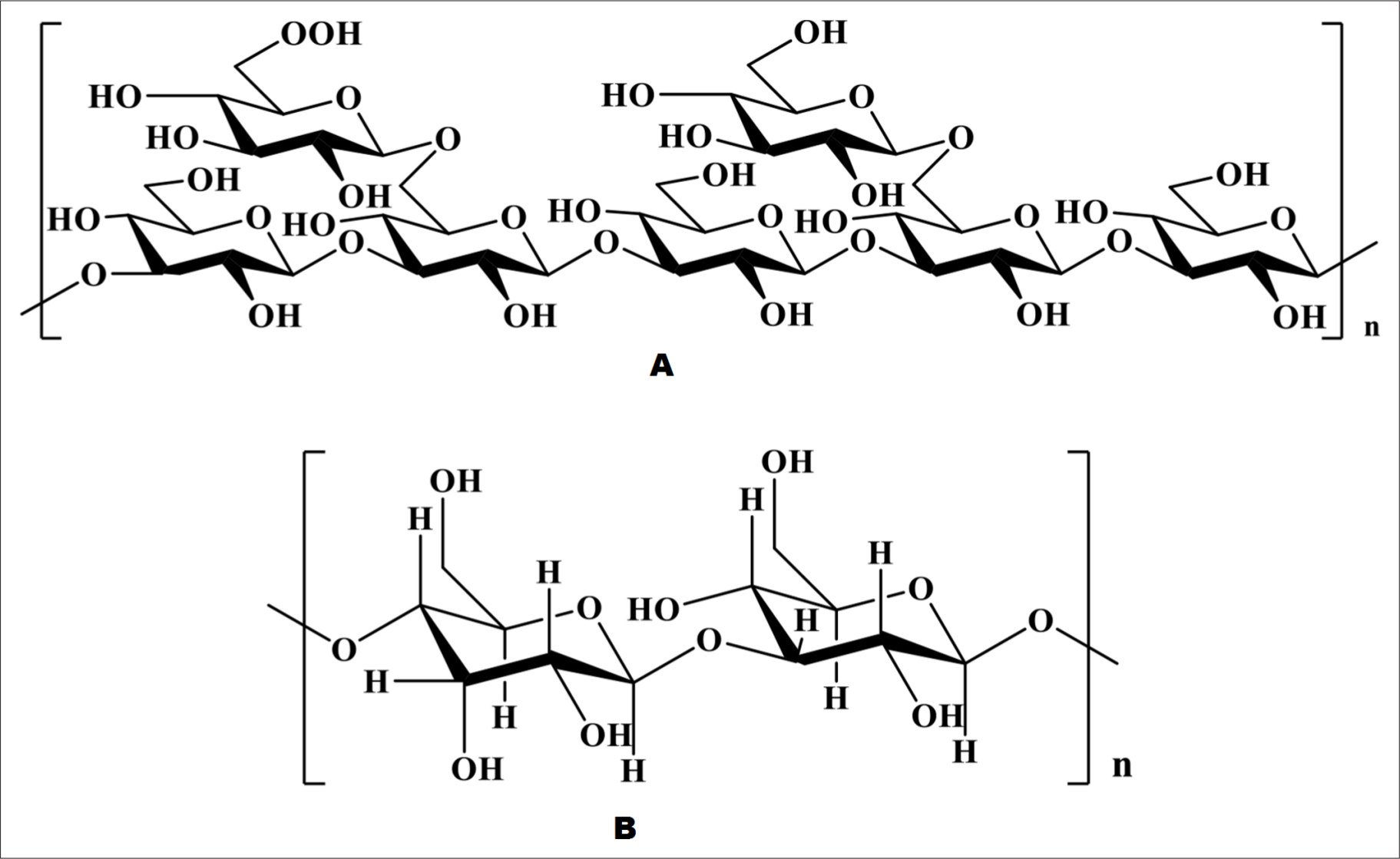
Chemical structure of ASP.
The sliced roots were extracted with water and then the alkaline and acidic proteins were removed with Ca(OH)2 and 3 M H2SO4, followed by ethanol precipitation and dialysis, and finally, chromatography with Sephadex G-50. Thereafter, Wang et al. found a polysaccharide, the molecular weight of which was 7.29 × 104 Da, composed of arabinose, glucose and galactose with a ratio of 1:2.5:7.5. 9
The dried roots were extracted with water. After addition of ethanol to the extract, the precipitated crude polysaccharides were collected, then dialyzed, freeze-thawed, and lyophilized. A polysaccharide was extracted with a carbohydrate content of 97%, the monosaccharide components of which were arabinose, glucose, rhamnose, galactose, and galacturonic acid, with a ratio of 10.4:4.7:1.0: 5.2:8.0. 10
Zhuang et al. determined the chemical structure of novel polysaccharides, as shown in Figure 1(B). 11 Water extraction of powdered roots was carried out by regulating the pH to remove acidic and basic proteins, then the polysaccharides precipitated with ethanol were collected and separated on a Sephadex G-50 column (35 cm × 3 cm) by eluting with water. Then Liu et al. isolated a new polysaccharide with a molecular weight of 7.8 × 104 Da, and a total sugar content of 95%, composed of arabinose, glucose, and galactose, with a molar ratio of 1:5.68:3.91. 12,13 Crude polysaccharides were obtained from powdered roots by water extraction, ethanol precipitation, dialysis, freeze-thaw, and lyophilization. The crude polysaccharides were loaded into a DEAE Sephadex A-25 column (80 cm × 5 cm), and eluted with distilled water, and then 0.1 M, 0.3 M, and 0.5 M NaCl, respectively. Four polysaccharides, APS-0, APS-1, APS-3, and APS-5, were obtained. APS-1 and APS-3 were further separated by Superdex 200 (100 cm × 3 cm). Thereafter Zhang et al. isolated 2 hoursomogeneous polysaccharides, APS-1a and APS-3a, with molecular weights of 49 and 6.54 × 104 Da, which were both composed of galactose, arabinose and glucose, with molar ratios of 57.34, 27.67, 14.98 and 84.54, 6.50, 8.96, respectively. APS-1a and APS-3a were mainly composed of 1,4-linked galactose, 1,3,6-linked galactose, T-galactose and T-arabinose, and the molar ratio of each bond in APS-1a and APS-3a was different. 14
The sliced roots were extracted with water and then the alkaline and acidic proteins were removed by adjusting the pH to 4-5 with 3 M H2SO4, followed by ethanol precipitation and lyophilization. Cheng et al. found a homogeneous ASP with a molecular weight of 8.09 × 104 Da and a sugar content of 88.0%, mainly composed of arabinose, glucose, and galactose with a ratio of 1:1:1.75. 15 The roots were successively extracted with NaOH aqueous solution, the polysaccharides precipitated with ethanol, the protein removed with CHCl3-n-BuOH, and then dialyzed with water to obtain crude polysaccharide. This was packed into different columns in turn. A DEAE-Sephadex A-25 column (80 cm × 4 cm) was eluted with distilled water, a Sephadex G-100 column (100 cm × 5 cm) was eluted with 0.1 M NaCl, and a Sephadex G-100 column was eluted with distilled water. Liu et al. finally extracted a homogeneous polysaccharide APS-1II with a molecular weight of 4.21 × 104 Da, and determined that it was composed of arabinose, glucose, and fucose with a molar ratio of 2.42:1.05:1.00. The backbone of APS-1II consisted of 1,3-α-L-Araf and 1,6-α-D-Glcp and the branched chain contained 1,5-α-L-Araf, 1,4-β-D-Glcp, T-β-D-Glcp, 1,3-α-L-Fucp, and T-α-L-Fucp. 16
The roots were first extracted with water, the polysaccharides precipitated with ethanol, and the protein and pigments removed with papain, CHCl3-n-BuOH, and H2O2. After dialysis and freeze-drying, the material was separated on a Sephadex G-100 column (65 cm × 3 cm) by eluting with water.
Polysaccharides were extracted by Ji et al. from unprocessed Chinese Angelica (UCA), Angelica parched with alcohol (ACA), Chinese Angelica parched with sesame oil (OCA), Chinese Angelica parched with soil (SCA), and charred Chinese Angelica (CCA). The carbohydrate contents of UCA polysaccharide (UCAP), ACAP, OCAP, SCAP, and CCAP were 91.5%, 93.1%, 88.6%, 89.9%, and 88%. Their monosaccharide compositions were all rhamnose, arabinose, mannose, glucose, and galactose, but in ratios of 3.9:35.8:3.1:29.9:28.1, 4.5:41.2:3.6:32.3:32.1, 4.1:40.1:3.3:30.6:31.3, 4.4:39.5:3.2:31.9:29.2, and 4.7:55.4:3.7: 31.6:30.2, respectively. 17
Crude polysaccharides were obtained by water extraction and ethanol precipitation. The crude polysaccharides were extracted with water again, and precipitated by ethanol 1 step precipitation and fractional precipitation. Wang et al. separated Chinese Angelica sinensis polysaccharides 30 (CAPS30), CAPS50, CAPS70, CAPS80, and total CAPS (CAPSt), and determined their carbohydrate contents as 90.31%, 85.50%, 86.95%, 88.62%, and 90.74%, and their molecular weights as 2.621 × 104, 2.07 × 104, 2.082 × 104, 2.719 × 104, and 2.083 × 104 Da, respectively. CAPS30 and CAPS80 contained arabinose, mannose, glucose, and galactose with molar ratios of 1.21:33.06:63.99:1.29 and 2.31:3.68:91.29:2.14, respectively. Mannose and glucose were the main components of CAPS50 and CAPS70 with a molar ratio of 1.05:86.22 and 1.20:1.01; the molar ratios of xylose and mannose were 1.92:1.00 in CAPSt. 18
The sliced roots were soaked in NaOH solution and the polysaccharides precipitated with ethanol. After protein removal with CHCl3-n-BuOH, the material was freeze-dried to obtain the crude polysaccharides. The crude polysaccharides were packed into a DEAE Sephadex A-25 column and eluted with distilled water, then 0.5 M, 1 M, and 2 M NaCl solution in turn. Then the material was fractionated on a Sephadex G-100 column (100 cm × 5 cm) by eluting with 0.1 M NaCl, followed by a Sephadex G-100 column eluting with distilled water. Liu et al. found a new heterogeneous ASP, called APS-2I, whose molecular weight was 7.2 × 105 Da, the main components of which were mannose, rhamnose, galacturonic acid, glucose, galactose, and arabinose, with a ratio of 4:5:1:10:23:39. The backbone of APS-2I was composed of 1,4-GalpA, 1,3-Rhap, 1,2,6-Manp, 1,6-Galp, and 1,2-Manp, and the branched chains contained TAraf, 1,4-Galp, 1,3-Araf, 1,5-Araf, 1,3,5-Araf,and 1,6-Galp. 19
The sliced roots were extracted with water, and the crude polysaccharides precipitated with ethanol were collected dialyzed and lyophilized. The crude polysaccharides were packed into a DEAE-Sepharose CL6B column (D 30 cm × 2.6 cm) and eluted successively with distilled water and a NaCl gradient (0, 1 M). Sun et al. finally isolated neutral polysaccharide ASP1, as well as acidic polysaccharides ASP2 and ASP3 from ASR. The molecular weight of ASP3 was determined to be 3.4 × 104 Da. ASP1 was found to be mainly composed of (65.00) glucose, (3.66) galactose, and (9.15) arabinose, while the contents of galacturonic acid, rhamnose, arabinose, and galactose in ASP2 and ASP3 were 35.38, 1.11, 16.31, 15.75 and 58.27, 1.87, 10.50, 24.93, respectively. 20
The powdered roots were extracted with water, the protein removed by Sevag, and the polysaccharides precipitated with ethanol and lyophilized. The product was loaded into a DEAE-52 cellulose anion exchange column (30 cm × 2.6 cm) and eluted with phosphate buffer and a gradient of NaCl (0.05, 1 M), respectively. Pan et al. discovered a neutral heteropolysaccharide with a molecular weight 8.2 × 104 Da, which was composed of arabinose, galactose, and glucose in ratios of 6:1:1. 21 The monosaccharide composition is shown in Table 1.
Composition and Characterizations of Polysaccharides.
Extraction Methods and Optimization
Extraction is a key step for obtaining polysaccharides from ASR and there are more than one extraction methods, including hot water, acid-alkali, and enzymatic extraction methods. There are many factors affecting the extraction efficiency of polysaccharides, the most important of which are the ratio of liquid to solid, extraction time, extraction temperature, and ultrasonic power for ultrasonic-assisted extraction. 22,23
Hot water is used for extraction, and the relationship between response function and process variables is identified by Box-Behnken design (BBD) and orthogonal array experimental design (OAD) to maximize the extraction yield. The results show that the best extraction conditions are as follows: extraction time 130 minutes (min), ratio of liquid to solid 5, and extraction number 5, then the extraction rate of ASP can be as high as 5.83%, but is actually 5.6% 24 ; extraction time 180 minutes, ratio of water to solid 6, extraction number 4 and extraction temperature 100 °C, then the extraction rate of ASP can reach up to 5.6%. 25
When ultrasonic-assisted extraction is used, the extraction method is analyzed and optimized by single factor experiment, OAD and BBD. The optimal extraction conditions are as follows: ratio of liquid to solid 43.31, ultrasound power 396.83 w, extraction time 28.06 minutes, and ASP yield 21.89% 23 ; ratio of liquid to solid 7, ultrasound power 180 w, extraction time 45 minutes, extraction temperature 90 °C, and ASP yield 6.96%. 26 These methods are presented in Table 2.
ASP Extraction Methods.

From the above results, it can be found that the actual extraction rates of the 2 water extraction methods are basically the same, and the reason may be that the extraction of ASP by this method has reached the limit. Therefore, with increasing ASP study, the extraction method also needs to be further improved.
Through comparison of the ASP yields mentioned above, we may come to a reliable conclusion that compared with water extraction, ultrasound-assisted extraction has a shorter extraction time and higher extraction rate. Ultrasonic-assisted extraction is cheap, easy to implement and more efficient. It can also exert mechanical effects to make more solvent penetrate into the tissue and increase the contact surface area between solids and liquids. Accordingly, the solute diffuses rapidly from the solid phase to the solvent. 26
Biological Activities
Hematopoietic Activity
Anemia caused by iron metabolism disorder is a prevalent clinical disease, which can be divided into 2 major categories: one is iron metabolism abnormality, the other is iron deficiency anemia (IDA), but hepcidin can regulate plasma iron levels. As a result, hepcidin has gradually become a potential target for the therapies of iron deficiency diseases and is affected by iron status, erythropoiesis, hypoxia, and inflammation. 27
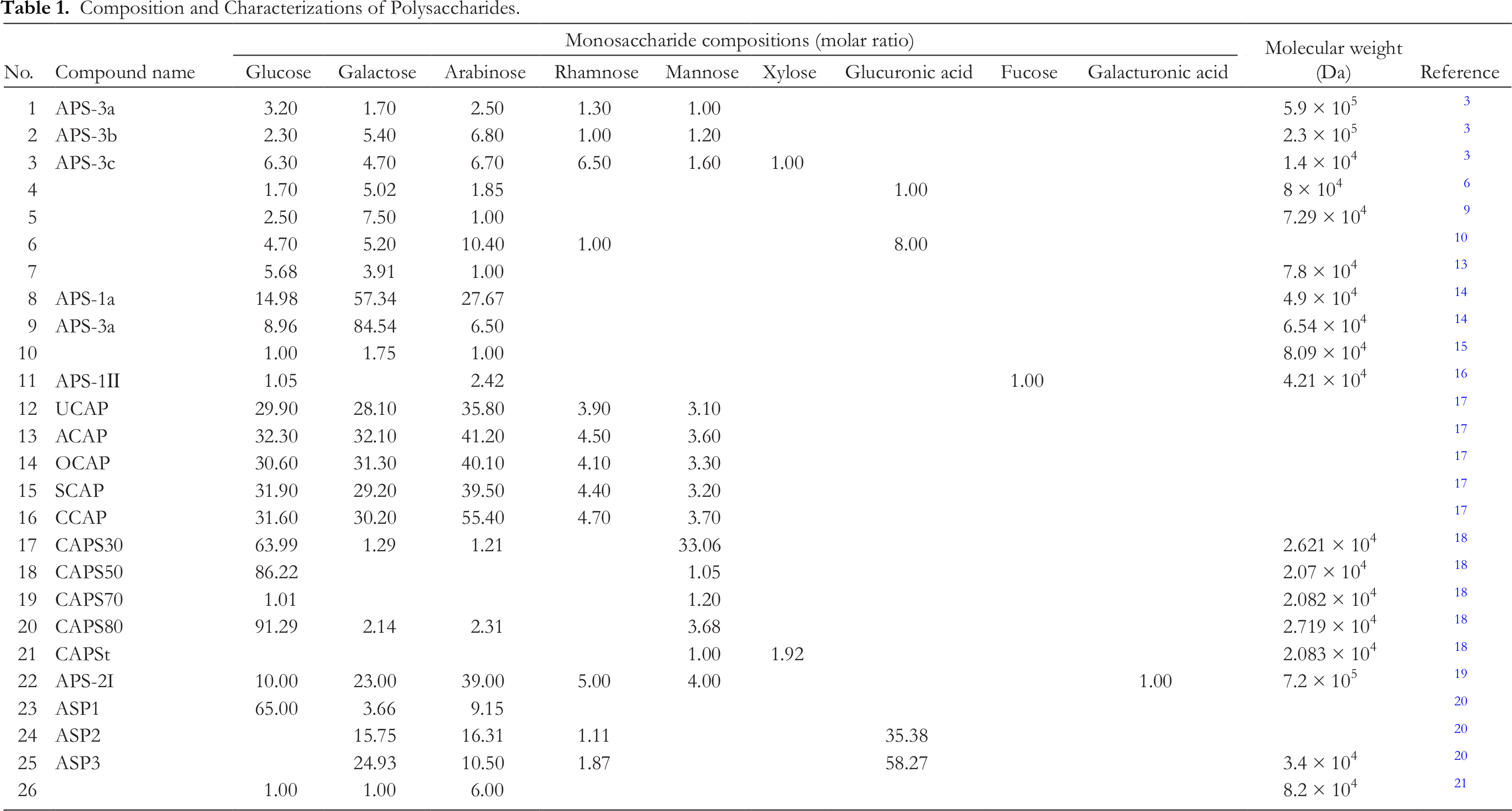
More recently, erythropoietin (EPO) has been shown to inhibit effectively the expression of hepcidin by promoting erythropoiesis in vivo, and is used to treat anemia caused by hepcidin, while ASP can promote the secretion of EPO and down-regulate the level of hepcidin in IDA rats. 13 Related studies have shown that ASP can promote the recovery of hemoglobin in mice with acute blood loss, and hematopoietic activity is achieved through the secretion stimulation of hematopoietic growth factors, such as granulocyte macrophage-colony stimulating factor (GM-CSF) and interleukin-3 (IL-3). 2,28 ASP can enhance EPO secretion, decrease the level of hepcidin and alleviate hematopoietic injury in mice after whole body irradiation. 14,29 It can also block Janus-kinase/signal transducers and activators of the transcription proteins (JAK-STAT), extracellular signal-regulated kinase (ERK), 27,29,30 IL-6/STAT3, and bone morphogenetic protein-mothers against the decapentaplegic protein (BMP-SMAD) pathways, thus inhibiting the expression of hepcidin, improving the utilization of iron, eliminating the suppression of inflammatory cytokines on erythropoiesis, and blocking the activation of inflammatory hepcidin and nuclear factor kappa-B (NF-κB) to treat rats with anemia of chronic disease (ACD). 31,32 Furthermore, ASP promotes the production of EPO in kidney and liver by stabilizing hypoxia inducible transcription factor-2α (HIF-2α) protein and weakening inflammatory inhibition, and it repairs the EPO receptor (EPOR) signaling system and increases iron utilization by inhibiting hepcidin and inflammation, thus treating anemia in rats with chronic nephropathy. 33
Liu et al. suggested that the hematopoietic and thrombopoietic effects of ASP may result from the phosphatidylinositol 3-kinase/protein kinase B (PI3K/Akt) pathways. 34 In addition, ASP can increase the number of red blood cells and the content of hemoglobin 35 -37 by up-regulating theexpression of cyclin D1 and Bcl-2 protein. 38 At the same time, ASP can stimulate EPO secretion and regulate the pathway of erythropoiesis, and so the level ofhepcidin mRNA decreased with the increase in erythropoiesis rate. 30 Additionally, ASP can restore the function of hematopoietic stem cells viainhibiting aberrant T-cell immunity and inducing anti-mitochondrial apoptosisin patients with aplastic anemia. 39 The mechanism of hematopoietic activity is shown in Figure 2 and relevant pathways are summarized in Table 3.
Hematopoietic and antioxidant mechanism.
Related Signal Pathways of ASP.

Antioxidant Activity
Oxidative stress, a key participant that leads to apoptosis, is closely related to ischemia-reperfusion injury, high-fat diet, and exhaustive exercise. 26,40,57
ASP could enhance the antioxidant components in rat models with myocardial ischemia reperfusion (MIR), such as superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px), and glutathione (GSH). 41,57 -59 The concentration of malondialdehyde (MDA), reactive oxygen species (ROS), and nitric oxide (NO) in brain tissue of cerebral ischemia reperfusion (CIR) rats treated with ASP also significantly decreased, while the activities of antioxidant enzymes increased, 25,60 suggesting that ASP could significantly reduce oxidative stress injury both in brain and heart caused by ischemia-reperfusion. In other studies, ASP could remarkably lower concentrations of ROS and MDA, as well as increase the levels of GSH and GSH-Px in livers of rat models with high-fat diet. 40 In addition, ASP also could strengthen the activities of SOD, CAT, GSH, GSH-Px, and glutathione reductase (GR) in the skeletal muscles of exhaustive exercise rats. 26
Lai et al. found that oxidized low density lipoprotein (ox-LDL) increased intracellular oxidative stress, which could be weakened by ASP. Moreover, ASP down-regulated the expression of gp91-phox, thus inhibiting the activity of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase. This inhibition decreased intracellular superoxide ion levels and oxidative stress in endothelial progenitor cells (EPCs). 10 In addition, the results showed that ASP had a scavenging effect on 2,2-diphenyl-1-picrylhydrazyl (DPPH), hydroxyl radical, and superoxide anion radical, thereby showing antioxidation. 42,61 ASP suppressed H2O2-mediated oxidative stress injury of human chondrocytes by up-regulating PPARγ and down-regulating inducible nitric oxide synthase (iNOS). 11 Furthermore, it also could significantly increase the level of total antioxidant capacity (T-AOC), 49 upregulate MicroRNA (miR)−126 and decrease lactate dehydrogenase and ROS products in H9c2 and HaCaT cells treated with H2O2, thus decreasing oxidative damage. 41,43 The mechanism of antioxidant activity is shown in Figure 2 and relevant pathways are summarized in Table 3.
Antitumor Activity
Cancer is one of the most pervasive and deadly human diseases in the world, killing more than 10 million people each year. Current cancer treatments mainly include surgery, radiotherapy and chemotherapy, which often kill healthy cells and cause poisoning. Hence, researchers are looking for ways to eliminate only cancer cells. 62
Zhuang et al. found that ASP could effectively induce apoptosis of human breast cancer cells T47D and Hs578T and inhibit the growth of breast cancer xenografts. Its effects are related to the activation of the cyclic AMP response element binding protein (CREB), caspase-3, rho-associated coiled-coil-containing protein kinase1 (ROCK1) activation, and myosin light chain phosphorylation. 12 ASP was also capable of inhibiting the proliferation of tumor cell lines HepG2, MCF-7, and A549 in vitro. 6 Additionally, it also could exert its anti-tumor activity by regulating iron metabolism, which might make hepcidin, IL-6, ferritin, transferrin (Tf), transferrin receptor 1 (TfR1), and TfR2 become targets of tumor therapy. 15,32 ASP could inhibit the proliferation, migration, invasion, and induce apoptosis of SH-SY5Y cells. It might inactivate PI3K/Akt and JAK/STAT signaling pathways via down-regulating miR-675 in SH-SY5Y cells, thereby inhibiting the tumorigenicity of SH-SY5Y cells. 44 Furthermore, ASP could block the growth of SH-SY5Y cells, the process of epithelial-mesenchymal transformation, as well as the PI3K/Akt and ERK1/2 signal pathways, which were achieved by up-regulating miR-205 with zinc finger E-box binding homeobox 1 as the target gene. 45 At the same time, ASP showed an inhibitory effect on the proliferation and cell cycle progression of U251 glioma cells, and induced apoptosis by stimulating the expression of E-cadherin through the transformation of the growth factor (TGF)-β signal pathway. 46
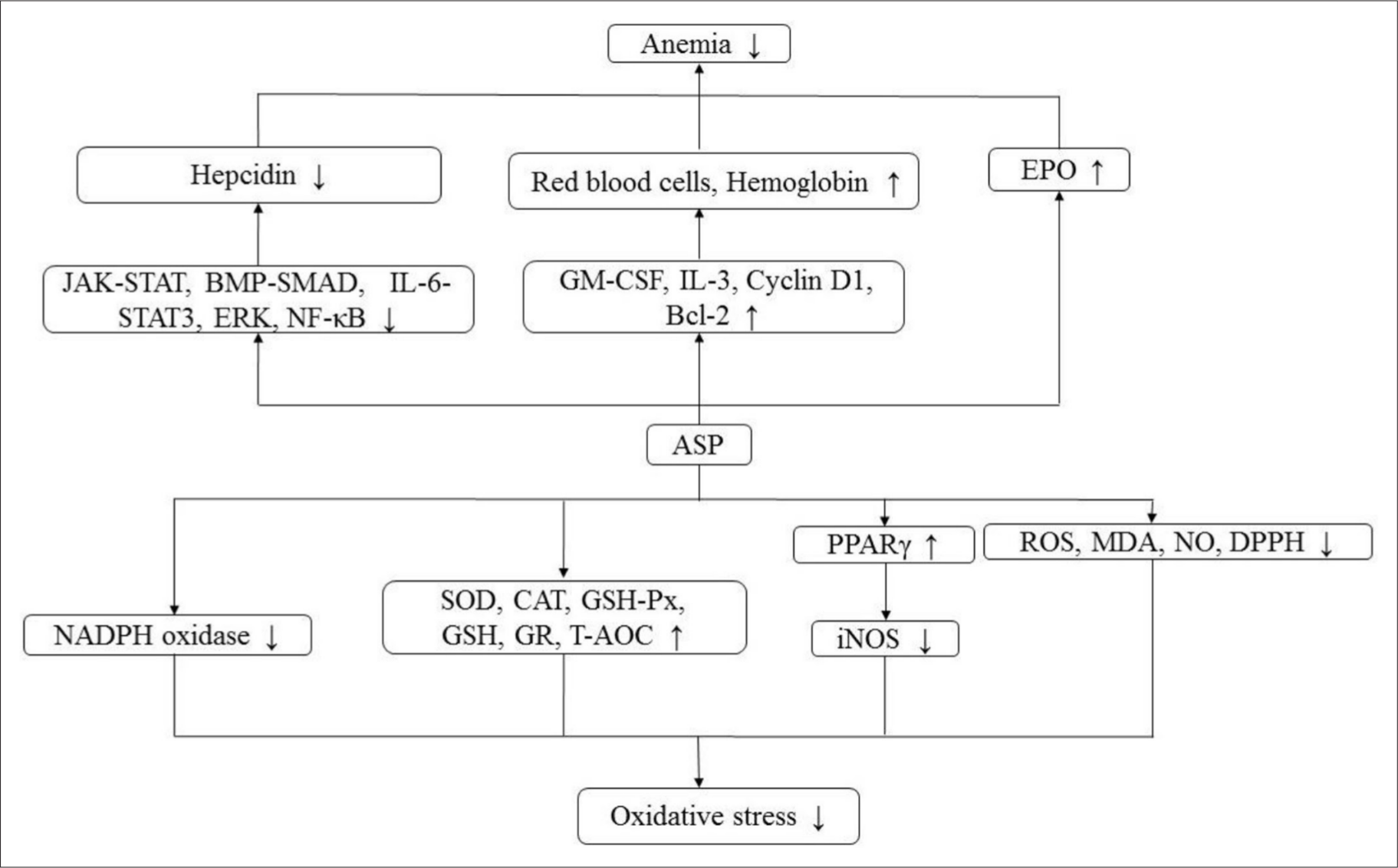
ASP could dramatically prolong the survival time of L1210 tumor-bearing mice. Its anti-leukemia effect was connected with the activation of leukocytes and lymphocytes in peripheral blood, inhibiting the secretion of cytokines tumor necrosis factor (TNF-α), interferon-γ (IFN-γ), and IL-2, thereby facilitating the proliferation of splenocytes, strengthening the phagocytic activity of peritoneal macrophages, and killing activity of natural killer (NK) cells. 16 ASP could dramatically suppress the proliferation of human acute myelogenous leukemia (AML) CD34+CD38-cells in vitro. In addition, ASP had an obvious cytotoxic effect on AML K562 cells, especially CD34+CD38-cells enriched by leukemia stem cells (LSC). It could significantly inhibit the colony formation of AML CD34+ CD38-cells. The potential mechanism of ASP-induced cell senescence was closely correlated to the upregulation of p53, p16, p21, and Rb genes, the changes of related cell cycle regulatory proteins P16, P21, cyclin E and Cell Cycle Dependent Kinase 4, telomere wear, and inhibition of telomerase activity. 63 ASP could suppress the proliferation of chronic myeloid leukemia K562 cells, and its mechanism had association with the induction of erythroid differentiation. In the process of erythroid differentiation of K562 cells induced by ASP, the results suggested that the JAK2/STAT5 signal played an indispensable role in erythroid differentiation of human myeloid leukemia cell line K562 induced by ASP. ASP induced erythroid differentiation of K562 cells through the JAK2/STAT5 signal pathway, and its potential mechanism might be attributed to the increased EPO response. 64 The mechanism of antitumor activity is shown in Figure 3 and relevant pathways are summarized in Table 3.
Antitumor and hepatoprotective mechanism.
Hepatoprotective Activity
In China, liver diseases, mainly viral hepatitis (primarily hepatitis B virus), non-alcoholic fatty liver disease, and alcoholic liver disease affect about 300 million people, which have a significant impact on the global burden. Therefore, it is imperative to explore liver protective drugs. 65
ASP could remarkably reduce the concentration of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and γ-glutamyl transpeptidase (γ-GT) in plasma, 66 and also significantly alleviate liver injury caused by hepatocyte apoptosis 67 and diabetes, whose mechanism might be related to the regulation of lipids, including a decrease in serum total cholesterol (TC) and low-density lipoprotein cholesterol (LDL-C) concentrations. 22 ASP exerted its hepatoprotective effect by inhibiting the secretion of pro-inflammatory cytokines and apoptosis in liver injury induced by ConA, which includes serum TNF-α, IFN-γ, IL-2, and IL-6 levels, and the potential mechanism may block the signal transduction pathways mediated by NF-κB, IL-6/STAT3, caspase-8, and c-JunN-terminal kinase (JNK), and up-regulate the level of Bcl-2. 4,47 ASP remarkably decreased plasma MDA and alkaline phosphatase (ALP) activities, while the antioxidant defense capacity of major enzymes was significantly enhanced, such as SOD, GSH-Px, and CAT in the livers of rats treated with CCl4 24. It also could significantly increase the level of serum total protein (TP) and inhibit the expression of p-ERK, p-JNK, and p-p38 in the mitogen-activated protein kinase (MAPK) signal pathway to resist liver dysfunction induced by CCl4 in mice. 42 The mechanism of hepatoprotective activity is shown in Figure 3 and relevant pathways are summarized in Table 3.
Anti-aging Activity
Cell senescence was first proposed by Hayflick and Moorhead in the 1960s as the irreversible stagnation of cells after long-term culture. Many factors, including various forms of stress, can accelerate cell senescence, such as oxidative stress, so how to delay and treat cell senescence has become a major concern in life and social sciences. 68
Mu et al. suggested that ASP could prevent the senescence of hematopoietic stem/progenitor cells (HSC/HPCs) induced by D-galactose (D-Gal) in mice via reducing the level of serum advanced glycation end products (AGEs) and the number of senescence associated β-galactosidase (SA-β-Gal) positive cells, as well as via promoting the formation of mixed colony forming unit (CFU-Mix). ASP prevented oxidative stress by increasing T-AOC and reducing the levels of ROS, 8-hydroxydeoxyguanosine and 4-hydroxynonenal. It decreased the expression of phosphorylated H2A.X (γ-H2A.X), which was the marker of DNA double-strand breaks, and lowered the subsequent ectopic expression of effectors in p16INK4a-retinoblastoma (RB) and p19Arf-p21Cip1/Waf senescence pathways. Additionally, ASP could reduce the expression of β-catenin, Ser9-phosphorylated GSK-3β (phospho-GSK-3β), and T cell factor (TCF)−4, and inhibit cyto-nuclear translocation of β-catenin, thus blocking the over-activation of Wnt/β-catenin signal in aged HSC/HPCs. 48 Furthermore, ASP suppressed the senescence of EPCs mediated by ox-LDL, which is related to the decrease of SA-β-Gal positive cells, the recovery of telomerase activity and Akt/ human telomerase reverse transcriptase (hTERT) pathway. 10 Aging is associated with oxidative damage and chronic inflammation. However, in D-gal-induced neural stem cells (NSCs), ASP could increase the activity of SOD and decrease the levels of MDA, IL-1b, IL-6, and TNF-α to play a protective role. It also could regulate the expression of these genes through decreasing the levels of p53 and p21 proteins, thereby delaying the aging of hippocampus and NSCs. 49 The mechanism of anti-aging activity is shown in Figure 4 and relevant pathways are summarized in Table 3.

Anti-aging and anti-inflammatory mechanism.
Anti-inflammatory Activity
ASP could enhance the expression of insulin like growth factor 1 (IGF1)/ IGF1 receptor (IGF1R) gene by promoting the expression of the UDP-sugar synthases (USSs) gene, thus accelerating glycosaminoglycan (GAG) synthesis and cartilage repair to exert its anti-osteoarthritis (OA) activity. 50 At the same time, ASP could facilitate and up-regulate the expression of aggrecan and GTs genes, thereby promoting proteoglycans (PGs) biosynthesis to treat rat OA. 69
In addition, ASP could remarkably inhibit the activation of RBL-2H3 cells, and it had a significant inhibitory effect on the release of pro-inflammatory cytokines, allergic mediators, and Ca2+ entry from RBL-2H3 cells stimulated by anti-dinitrophenol (DNP) immunoglobulin E (IgE). Treatment of ASP could suppress the expression of cytokines IL-1, IL-6, TNF-α, and NF-κB p65 inflammatory pathways. Furthermore, ASP could down-regulate the phosphorylation of Fyn in anti-DNP IgE-stimulated RBL-2H3 cells. The downstream phosphorylated signal molecules Gab2/PI3K/Akt and Fyn/Syk pathways, including p-Akt and p-P38, were also down-regulated in anti-DNP IgE-stimulated RBL-2H3 cells. 51
ASP could attenuate lipopolysaccharide (LPS)-induced inflammatory damage in PC12 cells by decreasing IL-1β and IL-8 levels and cyclooxygenase-1 (COX-1). It might protect PC-12 cells from LPS-induced inflammatory injury by suppressing miR223, and inactivating the NF-κB pathway. 52,53 ASP could up-regulate the expression of miR-10a in HT22 cells and then inhibit NF-κB and JAK2/STAT3 pathways to alleviate LPS-treated inflammation. It also could attenuate inflammatory injury through up-regulating sirtuin 1(SIRT1) expression and activating the nuclear factor-erythroid 2-related factor (NrF2)/heme oxygenase-1 (HO-1) pathway in HACaT cells induced by LPS. 54,55 The mechanism of anti-inflammatory activity is shown in Figure 4 and relevant pathways are summarized in Table 3.
Myocardial Protective Activity
Myocardial infarction is one of the diseases with high morbidity and mortality worldwide, which is featured with cell death caused by long-term ischemia after coronary artery occlusion. In addition, reperfusion usually cannot be treated in time, and it can also cause myocardial injury, that is, ischemia reperfusion injury. Therefore, it is of great clinical significance to find a drug to protect the heart. 56
ASP stimulated the activating transcription factor 6 (ATF6) branch of unfolded protein response (UPR) and attenuated harmful endoplasmic reticulum (ER) stress in the ischemic heart animal model in vivo and oxygen-glucose deprivation (OGD)-induced H9c2 cells in vitro. The results showed that ASP could target the ATF6 branch of UPR through adenosine monophosphate-activated protein kinase (AMPK)-peroxisome proliferator-activated receptor gamma coactivator 1α (PGC1α) pathway, thus improving the heart injury caused by ischemia. Furthermore, ASP might alleviate hypoxia-induced H9c2 cell injury via down-regulating the expression of miR-22. ASP also could significantly enhance the pro-survival molecules in the ER stress response system, including glucose-regulated protein 78 (GRP78), GRP94, and protein disulfide isomerase associated 6 (PDIA6), and reduce the expression of pro-apoptotic factors. In addition, ASP could alleviate myocardial ischemic injury through its antioxidant and anti-inflammatory activities. 56,70 The mechanism of myocardial protective activity is shown in Figure 5 and relevant pathways are summarized in Table 3.
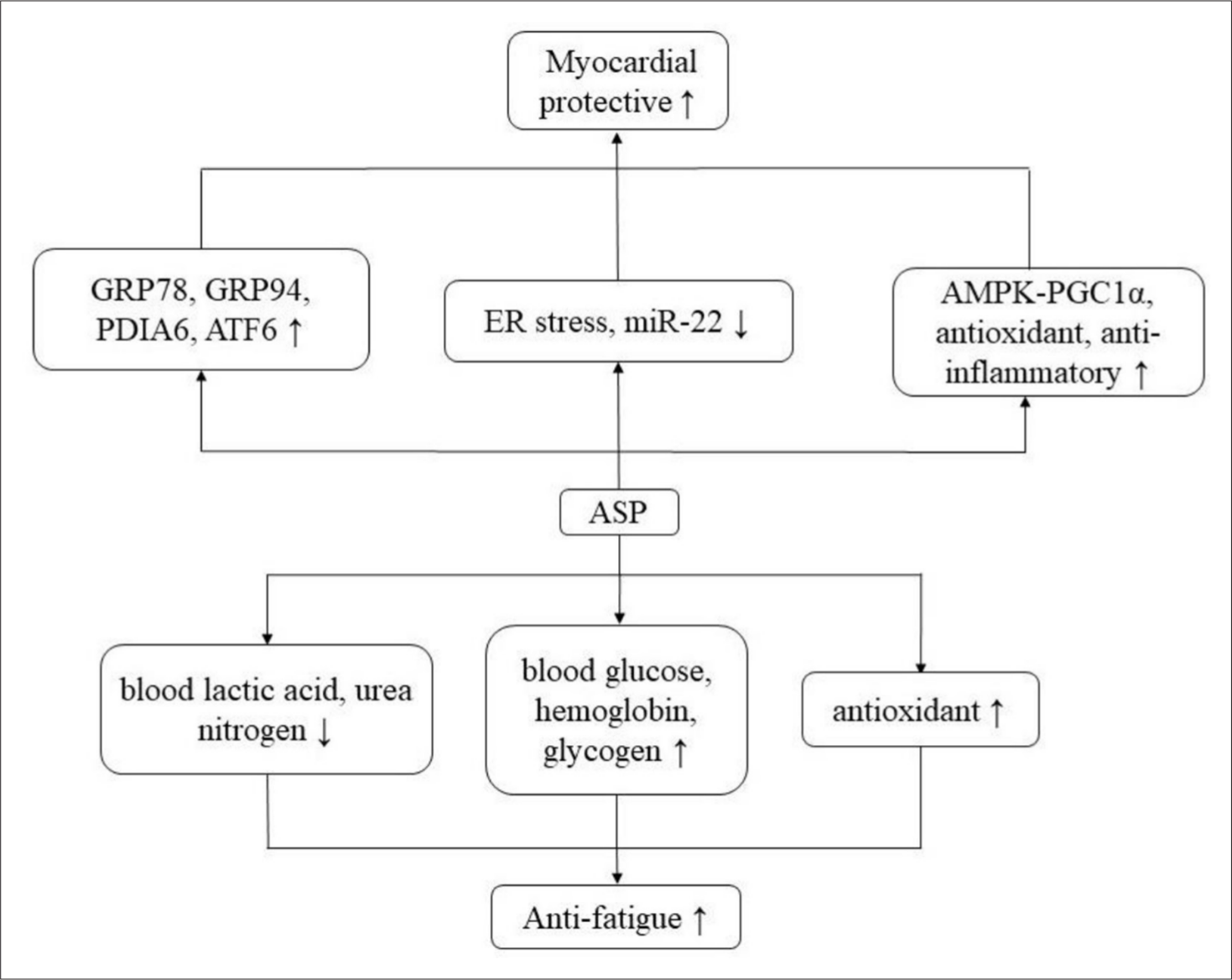
Myocardial protective and anti-fatigue mechanism.
Anti-fatigue Activity
Related articles suggest that ASP has an anti-fatigue effect, which could prolong the exhaustive swimming time of mice, decrease the production of blood lactic acid or promote the clearance of blood lactic acid, reduce the content of serum urea nitrogen, and increase the activities of blood glucose, hemoglobin and content of glycogen in liver and muscle. 61,71 Additionally, ASP could increase the glycogen reserve of liver and muscle and maintain the dynamic balance of the blood glucose level, which could guarantee the energy supply of the central nervous system, muscles, and erythrocytes and postpone the appearance of fatigue. Furthermore, ASP played a crucial role in fat metabolism, including the regulation of free fatty acid formation and the utilization of fat oxidation. At the same time, the antioxidant activity of ASP was helpful to protect the body from oxidative stress, thus to exert an anti-fatigue effect. 59 The mechanism of anti-fatigue activity is shown in Figure 5.
Other Activity
Sun et al. pretreated mice with ASP and then treated them with radiation. The results showed that the radiation-induced damage of mice in the ASP treatment group was significantly lower than that of the radiation control group, which proved that ASP had an anti-radiation effect, and the mechanism might be related to the protection of leukocytes and lymphocytes in mice. 20 Through in vitro assay, Xiao et al. found that ASP could improve the oxidative damage of bone marrow stromal cells and enhance their hematopoietic function to protect hematopoietic cells from stress-induced premature senescence, thus providing an effective treatment for chemotherapy-induced myelosuppression. 72 Furthermore, ASP could significantly inhibit the formation of calcium oxalate crystals in cells. In vivo, the renal pathological changes and calcium oxalate deposition in ASP treated rats were significantly alleviated, and the levels of urinary oxidative stress, oxalic acid, creatinine, and urea in rats treated with ASP were significantly lower than those of ethylene glycol-induced renal stone rats. ASP may play this role by regulating the expression of P-JNK and kidney injury molecule-1 in the kidney of rats, which reflects the potential anti-urolithiasis effect of ASP. 73 By feeding white shrimp with ASP, Pan et al. found that ASP could improve the activities of phenoloxidase, SOD, and GSH-Px, increase the total number of blood cells and the number of clear cells, semigranular cells and granulosa cells, and enhance disease resistance to Vibrio alginolyticus. The potential immunostimulatory activity of ASP was demonstrated. 21
Potential Clinical Applications
Anemia
IDA is a late symptom of iron deficiency and is the most prevalent category of anemia caused by inadequate iron storage, while hepcidin, as the main negative regulator of iron homeostasis, is mainly produced by the liver. Recently, hepcidin therapy for anemia has gradually become a research hotspot. Some studies showed that the stimulation of erythropoietin and the inhibition of SMAD4 expression participated in the inhibition of hepcidin expression induced by ASP, which blocked the phosphorylation of CCAAT/enhancer-binding protein α (C/EBPα) and STAT3/5, and then suppressed the expression of hepcidin in IDA rats. 13 Further research showed that ASP could significantly reduce the expression of hepcidin through inhibiting SMAD, JAK, and ERK pathway, which might have a potential clinical value in the alleviation of diseases caused by overexpression of hepcidin. 27,30
ACD is the second most widespread anemia, which is an acquired disease that occurs concurrently with chronic or acute immune activation, such as infection, autoimmune disease, and cancer. Wang et al. found that ASP could suppress the inflammatory hepcidin response of HepG2 cells and ACD rats by inhibiting IL-6/STAT3 and BMP/SMAD signaling pathways. After ASP treatment, the expression of iron transport protein, serum iron level, and serum EPO in ACD rats increased. Moreover, iron from the liver and spleen was activated, and symptoms of anemia were significantly alleviated. What is more, ASP could suppress the activation of NF-κB p65 through the IκB kinase-IκBα pathway, thus decreasing the secretion of TNF-α, as well as alleviating the inhibition of erythropoiesis accordingly. 31
Study on anemia in chronic nephropathy has shown that ASP could promote hypoxia-induced EPO production in Hep3B cells and its mechanism might be related to the equilibrium of HIF-2α protein. Wang et al. proposed that ASP could increase the stockpile of HIF-2α protein, decrease the expression of NF-κB, GATA binding protein 2 (GATA2) and pro-inflammatory cytokines, and stimulate the production of EPO in kidney and liver, thus leading to the increase of serum EPO. Meanwhile, ASP could relieve the inhibitory effect of pro-inflammatory factor TNF-α on EPO by suppressing the activation of GATA2 and NF-κB. In addition, ASP could repair the production of EPO and the expression of EPOR mRNA, induce the expression of target genes B-cell lymphoma-extra-large, Fam132b and Tfrc, and increase the ratio of Bcl-2/Bax in bone marrow mononuclear cells of chronic nephropathy rats, which might be involved in erythroid progenitor cell apoptosis, as well as iron ingestion and utilization in hemoglobin synthesis. 33
Osteoarthritis
Oxidative stress is involved in the apoptosis of chondrocytes, and ROS damages the cell membrane, protein, and DNA, resulting in pathological changes in the morphology and function of chondrocytes, which in turn leads to the apoptosis of chondrocytes. In addition, inflammatory cytokines are crucial to the occurrence and development of OA, while ASP could significantly reduce the levels of IL-1β and TNF-α, and apoptosis of chondrocytes. Furthermore, ASP might inhibit oxidative stress mediated by H2O2 through NF-κB and MAPKs signaling pathways in OA chondrocytes, increase the expression of anabolism genes Collagen type II alpha 1, aggrecan and SOX9, and suppress the expression of matrix metalloproteinase (MMPs) genes MMP-1, MMP-3, and MMP-9, thereby reducing the response of chondrocytes to H2O2, protecting chondrocytes, and promoting the recovery of injured chondrocytes. 1 ASP could inhibit the production of MDA, so as to reduce lipid peroxidation, up-regulate the expression of PPARγ, and down-regulate iNOS, then, consequently, the NO synthesis is decreased and H2O2-mediated oxidative stress damage of chondrocytes is alleviated. 11
The loss of GAG can result in continuous degeneration of joint cartilage. ASP could significantly promote GAG synthesis and cartilage repair, and its mechanism is related to the stimulation of USSs gene expression. It could also stimulate the expression of IGF1/IGF1R genes both in vivo and in vitro. Moreover, IGF1 can facilitate the synthesis and secretion of GAG in chondrocytes, thus improving the level of USSs mRNA and eliminating the inhibitory effect of IL-1β on USSs mRNA expression. ASP could increase the content of GAG in cartilage of OA rats and alleviate synovitis. 50 The loss of PGs is one of the characteristics of OA, which results in tissue destruction and cell loss, and finally leads to the loss of joint function. However, ASP might facilitate the expression of aggrecan and GTs genes, and thus promote PGs synthesis in chondrocytes. 69
Diabetes
Prediabetes is an extremely dangerous condition for developing diabetes, which is determined by blood glucose variables that are higher than normal, but below the diabetes threshold. 74
Related research showed that after intragastric administration of ASP, the level of fasting blood glucose was decreased and the abnormal level of fasting serum insulin was improved in streptozotocin (STZ)-induced diabetic and prediabetic mice. Further research found that the homeostasis model evaluation-insulin resistance (HOMA-IR) index of prediabetic mice was decreased significantly and the concentration of serum TC or triglyceride in prediabetic mice or STZ-induced diabetic mice was increased after ASP treatment. At the same time, the concentrations of liver glycogen and muscle glycogen in STZ-induced diabetic mice were increased, but IR-related inflammatory factors IL-6 and TNF-α were decreased. After ASP treatment, the damaged pancreas, liver or adipose tissue of STZ-induced diabetic mice or prediabetic mice were visibly recovered. 9
Wang et al. found that ASP could observably repair the glucose-producing ability of mice fed with a high-fat diet. The mechanism might be related to the increase of glucokinase expression and the decrease of pyruvate kinase activity. Furthermore, ASP could dramatically increase the activity of hexokinase (HK) in mice fed with a high-fat diet, suggesting that the recovery of HK may also be involved in the process. In addition, ASP notably attenuated IR induced by hyperlipidemia, suggesting that the hypoglycemic ability of ASP was connected with the elimination of IR. It can also alleviate IR via regulating IR/Insulin Receptor Substrate (IRS)/PI3K/Akt/glucose transporter /GSK-3β signal protein and inhibiting the expression of p-JNK in mice fed with a high-fat diet. 40,67
HOMA-β is one of the indicators of promoting insulin secretion, which is often regarded as an evaluation criterion to assess β-cell capacity. Islet β-cells are gradually considered to be the leading part of hypoglycemia by producing and secreting insulin, while ASP might improve islets and resist diabetes through inhibiting β-cell apoptosis. ASP could increase the ratio of Bcl-2/Bax to block the cascade of caspase 9-caspase 3 in islet β-cells. At the same time, inhibition of the exogenous apoptosis pathway driven by caspase-8 further suppresses the activation of subsequent substrate proteins, such as poly [ADP-ribose] polymerase (PARP). 22
Ischemia Reperfusion Injury
Ischemia myocardial reperfusion leads to irreversible tissue damage and cell necrosis, resulting in decreased cardiac function, while cardiotrophin-1 (CT-1) is a cytokine associated with hypertension, myocardial infarction, and other heart diseases. ASP administration could dramatically decrease the myocardial infarction area, increase the activities of CT-1 and antioxidant enzymes, and down-regulate the expression of caspase-12mRNA to alleviate myocardial ischemia-reperfusion injury in rats. 57
Myocardial ischemia leads to grievous ER stress, such as increased expression of pro-apoptotic C/EBP homologous protein and caspase-12 proteins. ASP could activate the ATF6 arm of UPR and alleviate the harmful ER stress in OGD-induced H9c2 cells and ischemic cardiomyocytes in vivo, as well as remarkably improve the survival molecules of the ER stress response system, including GRP78, GRP94, and PDIA6. Furthermore, it also could decrease pro-apoptotic factors. Accordingly, ASP can attenuate myocardial ischemic injury by restoring ER function, and target UPR ATF6 arm through the AMPK- PGC1α signal. 56
CIR usually leads to oxidative damage of brain tissue and neurons damage. However, ASP could remarkably increase the activity of antioxidant enzymes, and thus might attenuate the oxidative damage to the brain tissue of CIR animals. In addition, ASP also observably increased the activities of Ach, Na+, K+-ATP, Ca2+, and Mg2+-ATP in the brain tissue of CIR animals, and suppressed the activity of acetylcholinesterase, so as to regulate glycogen metabolism and to increase the level of ACh. 25,60,75
Tumor
Neuroblastoma is an extracranial solid malignant tumor, the most prevalent tumor in children and infants, and accounts for about 15% of all childhood cancer deaths. Recent findings suggested that ASP has anti-tumor activity against neuroblastoma SH-SY5Y cells, and its mechanism was related to the suppression of cell proliferation, migration and invasion, as well as the induction of apoptosis. ASP might inhibit neoplasia of SH-SY5Y cells via miR-205 and miR-675-mediated inactivation of PI3K/Akt, ERK1/2, and JAK/STAT pathways. In the meanwhile, KIF1B isoform β (KIF1Bβ) has been shown to be a target for miR-675, while KIF1Bβ is considered an inhibitor of neuroblastoma. 44,45
Leukemia is a heterogeneous neoplastic leukocytic disease, which has negative effects on blood, bone marrow, and the lymphatic system. There are 4 primary types of leukemia, acute lymphoblastic leukemia, AML, chronic lymphocytic leukemia, and myeloid leukemia. ASP could significantly suppress the proliferation of human AML CD34+CD38-cells cultured in vitro. In addition, ASP had an obvious cytotoxic effect on AML K562 cells, especially CD34+CD38-cells enriched by LSC. It was further shown that ASP could exert a significant inhibitory effect on the colony formation of AML CD34 + CD38 cells. 63 Furthermore, another study indicated that ASP was capable of inhibiting the proliferation of chronic myeloid leukemia K562 cells, and the mechanism was related to the induction of erythroid differentiation. 16,64
Nanoparticles
Researchers have chemically modified ASP with branched low molecular weight polyethylenimine (PEI) to obtain cationized ASP (cASP) (the reaction process is shown in Figure 6(A)), which was used as a gene vector to transfer the plasmid DNA encoding TGF-β1 into mesenchymal stem cells. cASP is combined with TGF-β1 plasmid to form cASP-pTGF-β1 nanoparticles. Subsequently, rat bone marrow mesenchymal stem cells and human umbilical cord mesenchymal stem cells were used to test the transfection activity of nanoparticles. The results showed that compared with the classical transfection agent liposome 2000, cASP-pTGF-β1 nanoparticles could deliver exogenous genes into cells faster and more effectively with less toxicity. 76
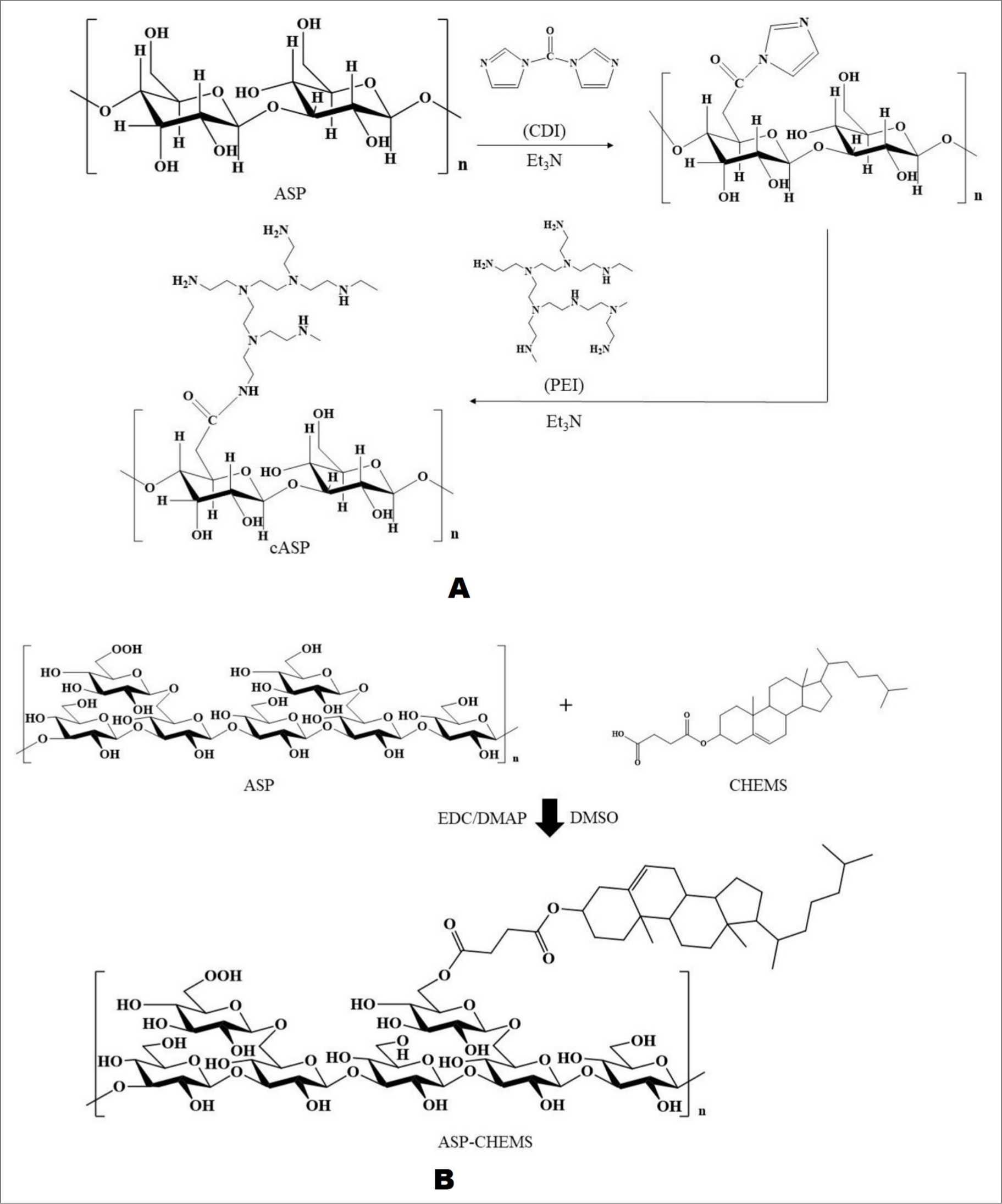
Chemical modification process.
Poly (lactic-co-glycolicacid) (PLGA) is a prevalently used synthetic biodegradable polymer, which is frequently applied to drug delivery systems because of its remarkable biocompatibility, biodegradability, and controllability of encapsulated drug release, so as to obtain better bioavailability and drug targeting. Gu et al. encapsulated ASP into PLGA to form ASP-PLGA nanoparticles, and then found that ASP-PLGA could significantly promote the proliferation of T and B cells compared with either ASP or PLGA alone. 77 Subsequently, Gu et al. embedded ASP and model protein antigen ovalbumin (OVA) in PLGA to constitute ASP-PLGA/OVA, and then injected it subcutaneously into mice. The results showed that ASP-PLGA/OVA could stimulate strong and persistent antibody response and induce cellular immune response. 78 Then, the author embedded ASP-PLGA into PEI to obtain ASP-PLGA-PEI nanoparticles, and proved that the antigen nanoparticles adsorbed by ASP-PLGA-PEI could be effectively internalized by macrophages, resulting in high concentrations of antigens and ASP in macrophages, so that antigens and ASP were transmitted to antigen presenting cells together. 79
Zhang et al. modified ASP with deoxycholic acid to obtain amphiphilic coupling compound ASP-DOCA, and then embedded doxorubicin (DOX) into ASP-DOCA to obtain DOX/ASP-DOCA nanoparticles. DOX/ASP-DOCA releases DOX in a continuous and pH-dependent manner, and it actively targets HepG2 tumors through the asialoglycoprotein receptor (ASGPR). In an animal model, DOX/ASP-DOCA passively accumulates into tumors through the enhanced permeability and retention effect, which significantly inhibits tumor growth and reduces the side effects of free DOX. 80 Wang et al. directly linked cholesteryl hemisuccinate (CHEMS) to ASP by esterification to obtain ASP-CHEMS nanoparticles (ACNPs); the reaction process is shown in Figure 6(B). Cur, as a model drug to protect the liver, was then encapsulated into ACNPs to constitute Cur/ACNPs. Through ASGPR-mediated endocytosis, ASP-derived ACNPs have high liver targeting, which could enhance the accumulation of Cur in the liver. Moreover, once Cur is encapsulated into ACNPs, then it has high drug load and good stability, and Cur can be continuously released from Cur/ACNPs, which improves the bioavailability of Cur. 7
Discussion and Research Prospect
It is known that polysaccharides are one of the most important bioactive constituents in ASR, and there are many extraction methods. In general, the ultrasonic-assisted method may be more efficient for extracting polysaccharides in comparison with the traditional hot water extraction. In order to improve the extraction efficiency of polysaccharides, it is essential that some extraction conditions still need further optimization, such as ultrasonic power, ultrasonic time, and liquid-solid ratio. As the main bioactive components of ASR for tonifying blood, the hematopoietic effect of ASP has attracted much attention. Many studies have proved that ASP could promote the secretion of EPO, increase red blood cell count and hemoglobin content and regulate the release of hepcidin. These effects are associated with hematopoietic factors such as GM-CSF and IL-3, as well as protein pathways including PI3K-Akt, JAK-STAT, and BMP-SMAD. In addition, ASP also could enhance antioxidant enzymes, inhibit tumor cell proliferation, immunostimulation, and protect cardiomyocytes and hepatocytes. In the meantime, the application of ASP nanoparticles in drug delivery has been reported and demonstrated good liver targeting, which provides potential research topics for the interested scholars.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (81773882, 81974522) and Key Research and Development Program of Shaanxi Province (2019ZDLSF04-05). This research was also financially supported by Subject Innovation Team of Shaanxi University of Chinese Medicine (2019-YL10) and The Youth Innovation Team of Shaanxi Universities (2020).
