Abstract
Breast cancer is the most common invasive cancer in women and the second leading cause of cancer death in women. However, it is not clear about its effective treatments. As a potential anticancer agent, grape seed procyanidins (GSPs) have been shown to inhibit the proliferation of various cancer cells in vitro and in vivo. In this study, it was shown that GSPs significantly inhibit MCF-7 cell proliferation in a concentration/time-dependent manner. The flow cytometric data clearly demonstrated that GSPs cause cell cycle arrest in the G2/M phase, followed by cell apoptosis. Moreover, it also confirmed that growth inhibition mediated by treatment with GSPs is related to the induction of apoptosis due to p53 elevation, purportedly by inhibition of the epidermal growth factor receptor (EGFR)/vascular endothelial growth factor (VEGF)/matrix metalloproteinase 9 (MMP9) pathway. Taken together, these findings suggest that GSPs inhibit MCF-7 cells proliferation and induce cell apoptosis by suppressing EGFR/VEGF/MMP9 pathway.
Breast cancer, a hormone-responsive cancer that accounts for about one fourth of all female cancers, has become the leading cause of cancer-related death among women in the world. 1 One of the reasons for such a high mortality is the invasive behavior of cancer cells, which results in the metastasis of breast cancer. 2 Angiogenesis, as well as the formation of blood vessels by preexisting capillaries, plays an important role in tumor growth and metastasis. 3 Within the past decades, angiogenesis has become widely accepted to be essential for breast cancer cell proliferation and metastasis. 4 Efficient anti-angiogenic strategies have emerged as a new modality to treat cancers via the inhibition of pro-angiogenic molecules. Besides, cancer metastasis involves several interdependent processes, including uncontrolled cell proliferation, invasion of surrounding tissues, migration to distant parts of the body, as well as adhesion, invasion and colonization of other organs and tissues. 5 Thus, inhibition of growth, and aggressive behavior cancer will lead to inhibition of breast cancer metastasis and further increase the survival rate of the patients.
Traditional Chinese medicines have been recently recognized as a new source of anticancer drugs and neoadjuvant chemotherapy to enhance the efficacy of chemotherapy and to alleviate the side effects of cancer chemotherapy. 6 Procyanidins are the major group of polyphenols in the human diet because of their widespread occurrence in fruits, berries, nuts, beans, cocoa-based products, wine, and beer. 7,8 Grapes are one of the most widely consumed fruits in the world, which are rich in proanthocyanidins with 60% to 70% of the proanthocyanidins being contained in the seeds, and they have enormous health benefits. Grape seed proanthocyanidins (GSPs) are widely consumed as a dietary supplement and possess chemopreventive and/or chemotherapeutic effects in various cell culture and animal models. 9,10 There is growing evidence showing that GSPs have inhibitory effects in vitro and in vivo on various cancers without having adverse effects on normal cells. 11,12 However, it is unclear whether GSPs possess effective chemopreventive activities against breast cancer. Moreover, no evidence has been reported for the direct effect of breast cancer on MCF-7 cell proliferation and the mechanisms of this effect.
In the present study, we provided the evidence that GSPs inhibit cell proliferation in human cancer cells through cell cycle arrest at the G2/M phase and induction of apoptosis. Mechanistic studies revealed that interruption of the epidermal growth factor receptor (EGFR)/vascular endothelial growth factor (VEGF)/matrix metalloproteinase 9 (MMP9) signaling pathway plays a critical role in these GSPs-mediated cell cycle arrest and induction of apoptosis via activating p53 expression. Our results provide a novel explanation for GSPs as an angiopreventive agent against breast cancer.
Results
GSPs Inhibit Cellular Growth
The cytotoxicity of GSPs was evaluated by a MTT growth inhibition assay, which was carried out with MCF-7 cells. The screening procedure was based on the standard MTT method, and the IC50 value, at which the compound produced 50% inhibition of cell growth. From Figure 1(A), the IC50 value of GSPs for inhibition of cell proliferation in MCF-7 cells was 0.10 ± 0.02 µM. To evaluate further the effects of GSPs on the growth of human breast cancer cells, the growth inhibitory potential of this compound with different concentrations (0, 0.05, 0.1, 0.2 µM) was determined in MCF-7 cells at various times. We found that GSPs significantly inhibited the growth vitality of MCF-7 cells, with suppression peaking at 0.2 µM at 48 hours (Figure 1(B)). Also, the results showed that treatment with GSPs for 48 hours at different concentrations led to a 12-fold decrease in cell viability (Figure 1(B)). Besides, we also found that 0.2 µM GSPs at different treatment times (12 h-48 h) significantly inhibited cell viability, which was decreased to 37.3% (Figure 1(B)). Together, it demonstrated that the cell viabilities were decreased in a dose-dependent, as well as time-dependent manner in cells treated with GSPs.
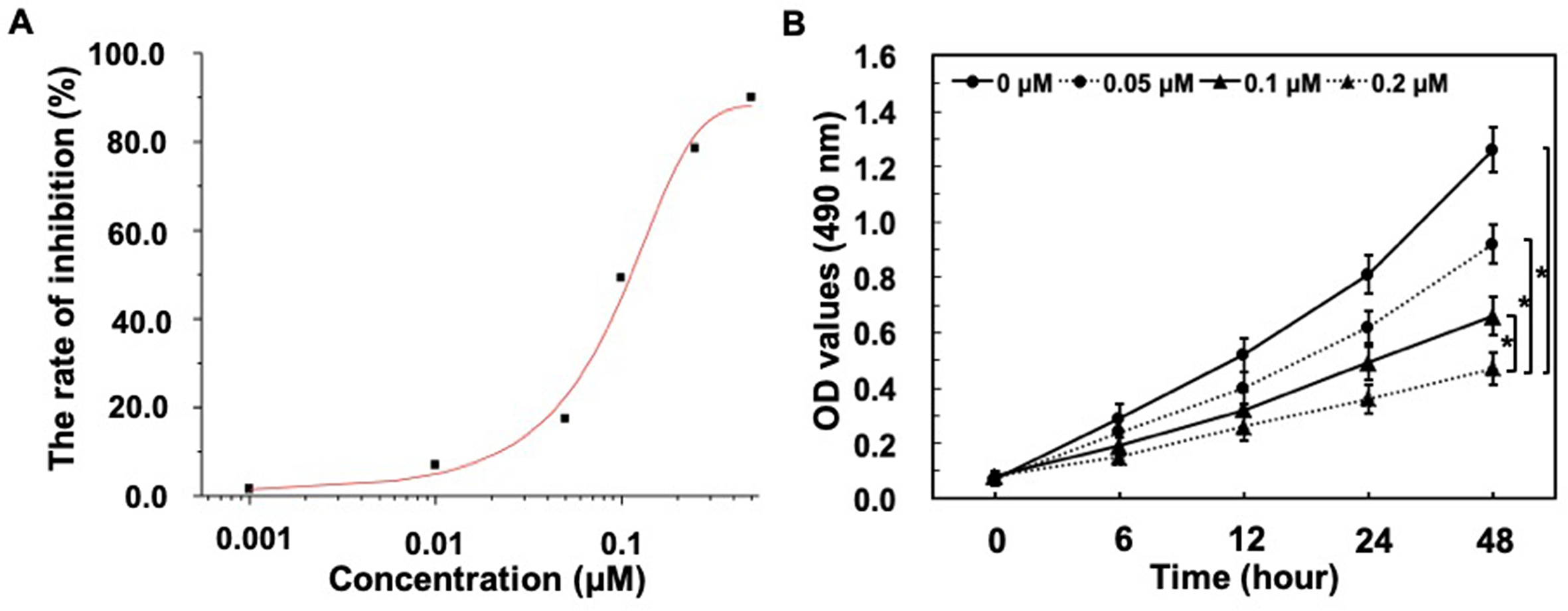
GSPs inhibit MCF-7 cells’ viability in vitro. (A) The IC50 value of GSPs was determined by MTT test. MCF-7 cells were treated with different concentrations of GSPs (0, 0.001, 0.01, 0.05, 0.1, 0.25, 1 µM) at 48 hours. Cell inhibition rate was calculated as 100% × (OD value in control group-OD value in experimental group)/OD value in control group. The IC50 value was obtained from origin 8.0 software. (B) Results of MTT assay, indicating the inhibition of MCF-7 cells viability by GSPs in a concentration-dependent and time-dependent manner. The cells in the control group were treated with DMEM +10% FBS, whereas the cells in the test groups were treated with DMEM +10% FBS +GSPs (0.05, 0.1, and 0.2 µM, respectively) for different times (6 h, 12 h, 24 h, and 48 h). Values are expressed as the means ± SD, n = 3, *P < .05.
GSPs-Induced Cell Cycle Arrest at the G2/M Phase and Apoptosis in the MCF-7 Cell Line
To explore the mechanism by which GSPs inhibit cell proliferation, we next investigated their effect on cell cycle progression in human breast cancer MCF-7 cells by using flow cytometry assay. The typical histograms of cell cycle distribution in cells treated either without or with GSPs for 24 hours are shown in Figure 2(A). A concentration dependent change was observed in the cell cycle pattern (P < .05), which is shown in Figure 2(C). Our results demonstrated that treating MCF-7 cells with GSPs increased the percentage of cells in the G2/M phase from 17.3 ± 3.1% to 69.4 ± 4.1% as the concentration increased from 0.05 to 0.2 µM. These results suggested that the GSPs-mediated inhibitory effects of cell proliferation were accompanied by cell cycle arrest at the G2/M phase.

GSPs induce cell cycle arrest at G2/M and apoptosis in MCF-7 cells. (A) MCF-7 cells in log phase were harvested and subjected to cell cycle analysis. The cell cycle phase distribution was evaluated by FACS. The data were analyzed with ModFit software. The results shown are representative images of three separate experiments. (B) Representative flow cytometric profiles demonstrating cell apoptosis of MCF-7 cells. Cells were incubated for 48 hours and induction of apoptosis was measured by Annexin-V/PI double-staining assay. (C) The percentage of cells in G2/M phase. Values represent the means ± SD, n = 3, *P < .05 compared with control group. (D) The rate of apoptosis was calculated and expressed as means ± SD of three separate experiments. *P < .05 by Student’s t-test.
We next investigated by flow cytometry assay whether GSPs inhibited cell proliferation through induction of apoptosis. As shown in Figure 2(B), the percentage of normal cells (left lower section of fluorocytogram) remaining after treatment with 0.2 µM of GSPs represented (21.5 ± 2.4)% of the total cells, compared to (68.1 ± 3.6)% of MCF-7 cells treated with 0.1% dimethyl sulfoxide (DMSO) as a control. Also, it revealed a basal apoptotic population, including early apoptosis (right lower section of fluorocytogram) and late apoptosis (right upper section of fluorocytogram) in the untreated culture as (29.9 ± 3.9)%. However, after treatment with GSPs a dose dependent increase in apoptotic population was observed, which was found to be 41.8 ± 3.1%, 63.1 ± 3.1% and 76.7 ± 4.1%, respectively, at 0.05, 0.1 and 0.2 µM after 48 hours of incubation. Moreover, there was a statistically significant difference in the degree of apoptosis (P < .05) (Figure 2(D)). Taken together, these results confirmed that growth inhibition mediated by treatment with GSPs was related to the induction of apoptosis.
EGFR/VEGF/MMP9 Pathway Down-Regulation in GSPs-Treated MCF-7 Cells
Angiogenesis is essential for tumor growth and metastasis. Two key elements in the growth and dissemination of tumors are VEGF and EGFR. VEGF is a potent inducer of angiogenesis and is identified to be associated with tumor angiogenesis. 13 EGFR is a member of the human epidermal growth factor receptor (HER) family of transmembrane receptor tyrosine kinases that is linked to growth control, cell adhesion, mobility, and apoptosis. 14 The VEGF and EGFR pathways are closely related, sharing common down-stream signaling pathways. 15 Furthermore, one of the key EGFR ligands, EGF is one of the many growth factors that drive VEGF expression. 16 Matrix metalloproteinases (MMPs), which can degrade extracellular matrix components, play a critical role in tissue remodeling during development in pathological process, including inflammation, tissue repair, tumor invasion, and metastasis. 17,18 Among MMPs, MMP9 is of particular interest, because it is thought to be a tumor angiogenic factor that signals through the VEGF-VEGFR system. 19 Considering that GSPs inhibit MCF-7 cells proliferation, we sought to determine to what extent GSPs treatment would inhibit signaling throughout this pathway in the proliferation of MCF-7 cells and assess the baseline inactivation status of the EGFR/VEGF pathway. After cells were treated with different concentrations of GSPs (0, 0.05, 0.1, 0.2 µM) for 48 hours, Western blot analysis revealed low levels of expression of EGFR with the greatest reduction evident at 0.2 µM and demonstrated a dose-dependent decrease (P < .05) (Figure 3). Also, GSPs at all doses suppressed VEGF protein levels, with complete inhibition at 0.2 µM (Figure 3(A)). Note as well that levels of MMP9, a VEGF downstream molecule, were decreased along with EGFR and VEGF in a dose-dependent change (P < .05) (Figure 3(B)). These results suggested that the inhibitory effect of GSPs on cell proliferation in MCF-7 cells was partly through suppressing the EGFR/VEGF/MMP9 pathway that initiates tumor angiogenesis. p53 plays an essential role in integrating multiple stress signals in the process of diverse anti-proliferative responses. 20 One of the most important p53 functions is its ability to induce apoptosis, and thus, it serves as a regulator of the apoptotic process that can modulate key control points in both the extrinsic and intrinsic pathways. 21 Downregulation of MMP is closely associated with cell cycle arrest and apoptosis. 22 We therefore evaluated whether GSPs-mediated EGFR/VEGF/MMP9 pathway suppression would affect the expression of p53 after MCF-7 cells stimulated by the indicated concentration of GSPs (0.05, 0.1 and 0.2 µM) for 48 hours. Western blot analysis revealed that the expression of p53 was elevated by all GSPs-treated MCF-7 cells (Figure 3(A)) and the difference between these was significant (P < .05) (Figure 3(B)). Caspase-3 is the main downstream effector caspase that plays an important role in degrading the majority of essential cellular components in apoptotic cells, 23 indicating its crucial role in the cell death process. As shown in Figure 3(A), 0.05 µM GSPs markedly induced Caspase-3 expression in MCF-7 cells and was followed by a robust increase in proteins levels at 0.1 and 0.2 µM (P < .05) (Figure 3(B)). Our observations indicated that activation of p53 and Caspase-3 induced by GSPs might partially be attributed to inhibition of the EGFR/VEGF/MMP9 pathway.

GSPs inhibit EGFR/VEGF/MMP9 pathway, followed by p53 and Caspase-3 activation. (A) MCF-7 cells were treated with different concentrations of GSPs (0, 0.05, 0.1, and 0.2 µM). Then cells were harvested at 48 hours. Whole cell extracts were subjected to immunoblot analysis with EGFR, VEGF, MMP9, p53 and Caspase-3 antibodies. β-Actin was used as loading control. (B) Data were analyzed by SPSS 13.0 software. Data are means ± SD. Representative results from at least three individual experiments are shown. *, &, #, $, @ Student’s t-test, P < .05 (VS. Control).
GSPs Enhance P53 Activity in MCF-7 Cell Lines
The p53 pathway responds to various cellular stress signals (the input) by activating p53 as a transcription factor (increasing its levels and protein modifications) and transcribing a program of genes (the output) to accomplish a number of functions. 24 The p53 tumor suppressor protein coordinates multiple functions, including the ability to promote cell-cycle arrest, apoptosis, and senescence through its target genes such as p21, Puma, Bax, and Bid. 25 Then, we examined the effect of GSPs on p53’s transcriptional activity in vitro using MCF-7 cells. The cells were treated with GSPs with different concentrations (0.05, 0.1 and 0.2 µM) for 48 hours. RNA was isolated from each sample and subjected to qRT-PCR to assess transcription of the p53-targeted genes stated above. Transcription of these genes was elevated in the treated-MCF-7 cells compared to the control. Statistically significant differences (P < .05) between treatment and non-treatment were observed for p21, Puma, Bax, and Bid according to Student’s t-tests (Figure 4), confirming that GSPs could actually enhance p53 activity. Also, these results demonstrated that the inhibitory effect of GSPs on MCF-7 cells proliferation was at least in part attributed to the enhancement of p53 transcription activity and the consequent actisvation of its target gene expression, such as p21, Puma, Bax, and Bid.
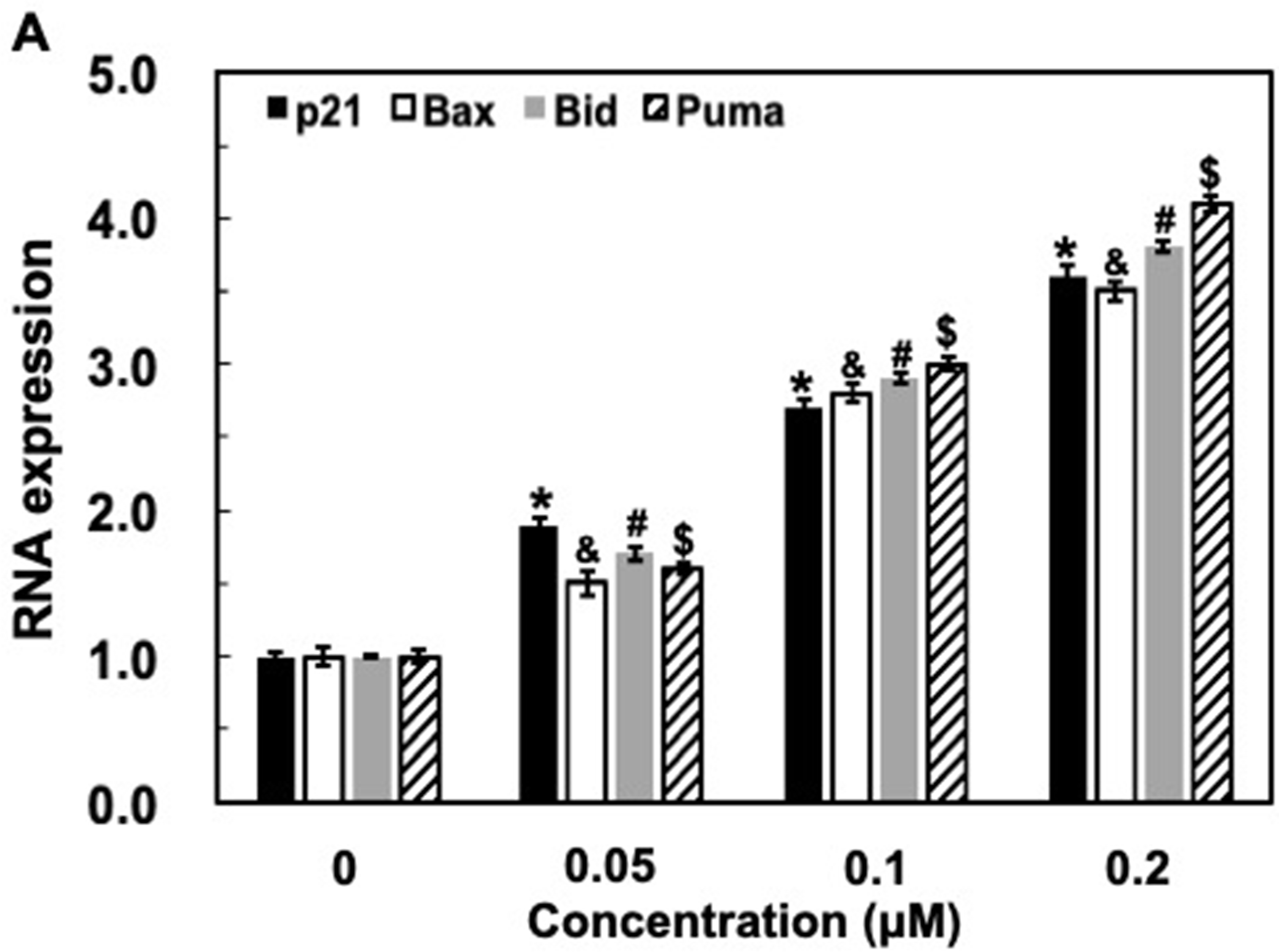
GSPs significantly promote the expression of p53 target genes at the RNA level. MCF-7 cells were treated with GSPs (0, 0.05, 0.1, and 0.2 µM) for 48 hours and harvested for RNA extraction and q-PCR analyses for specific p53 target genes, including p21, Puma, Bax, and Bid. GAPDH was used as negative control. Three independent experiments were performed in duplicates. Differences between the treatment and non-treatment for each target were analyzed using Student’s t-test (n = 3, *, &, #, $ P < .05).
Discussion
Tumor angiogenesis plays a crucial role in tumor growth and progression. More importantly, most tumors cannot grow more than 2‐3 mm3 without forming new blood vessels. 26 Breast cancer is a kind of angiogenesis-dependent tumor. The prognosis of primary tumor patients is closely related to the degree of angiogenesis. 27 Therefore, the antiangiogenic therapy for breast cancer has been studied for decades in preclinical and clinical studies. However, the clinical impact is not very effective until today.
Proanthocyanidins are naturally occurring compounds that are widely available in fruits, vegetables, nuts, seeds, flowers, and bark. 28 Grape seeds are a particularly rich source of proanthocyanidins. GSPs, which are composed mainly of dimers, trimers, tetramers, and oligomers of monomeric catechins, 29,30 are a nutritional supplement with known disease prevention characteristics. 31,32 A series of preclinical and mechanistic studies have showed that GSPs could have effective anticarcinogenic activity, inhibit proliferation, and induce apoptosis in a wide variety of tumor models via modulation of multiple cell signaling events involving critical molecular candidates associated with growth, differentiation, apoptosis, and oncogenesis. 33 -35 The in vitro studies presented here indicate that treatment of MCF-7 cells with GSPs results in a dose/time-dependent inhibition of cell viability and proliferation (Figure 1) together with G2/M phase arrest and cell apoptosis (Figure 2). Therefore, the cell cycle arrest and induction of apoptosis in MCF-7 cells may be one of the mechanisms of the antitumor effects of GSPs.
Then, we studied the defined molecular mechanisms underlying GSPs-regulated suppression of human breast cancer by measuring the expression levels of several growth factors and proteins in MCF-7 cells. EGFR activation is involved in cancer cell proliferation, migration, and invasion, and it is overexpressed in various cancers, and then can activate several downstream signaling pathways. 36 The results showed that the EGFR expression level is significantly reduced in the GSPs-treated cells in a dose-dependent manner compared with the control group (Figure 3). Under pathological conditions, VEGF is secreted by tumor cells in the majority of cancers and acts on endothelial cells of existing blood vessels to promote new blood vessel formation. VEGF released by tumor cells initiates the angiogenic process by activating endothelial cells and promoting their migration. 37 Also, it is reported that EGFR is a major inducer of VEGF. 38 EGFR signaling can upregulate VEGF, while EGFR inhibition decreases VEGF expression and tumor vascularization. 39,40 Indeed, our data showed VEGF reduction is consistent with the change of EGFR expression after GSPs treatment (Figure 3). A decrease in MMP-9 protein expression is found after VEGF blockade, meaning that MMP-9 is positively regulated by the VEGF pathway, which plays a key role in cancer metastasis. 41 We also observed that GSPs downregulate the expression of MMP-9 (Figure 3). p53 serves as the major obstruction for tumorigenesis by inducing apoptosis and programmed cell death and inhibiting the growth of some cells by cell cycle arrest. 42 Caspase-3 has been reported to be the most frequently activated caspase protease in apoptotic cells. 43 p53 and Caspase-3 proteins expression in MCF-7 cells were increased following 48 hours treatment with GSPs. This response, which was evident even at low doses, was amplified with increasing doses (Figure 3), implying that p53, as well as Caspase-3, plays an essential role in the GSPs’ mechanisms of action. These results suggest that GSPs inhibit the growth of MCF-7 cells through EGFR/VEGF/MMP-9 signaling pathway suppression, followed by p53 and Caspase-3 activation. The tumor suppressor gene p53 has been shown to regulate the cell cycle at the G2/M checkpoint in response to DNA damage. 44 Upregulation of p53 leads to G2/M arrest through the induction of p21. In addition, p53 has also been shown to regulate apoptosis through inhibiting the expression of anti-apoptotic proteins (i.e., Puma, Bax, and Bid, etc.,.) 45 As shown in Figure 4, the mRNA expression of p21, Puma, Bax, and Bid was significantly increased by GSPs in a dose-dependent manner. These results suggest that the cell cycle arrest and apoptotic response induced by GSPs may be associated with p53 transcriptional activation.
In summary, we report that GSPs can simultaneously inhibit proliferation of breast tumor cells. Importantly, we further confirm that the EGFR/VEGF/MMP-9 pathway could be involved in the control of GSPs-induced proliferation inhibition, followed by cell apoptosis. Our studies provide novel insights into the anticancer activities of GSPs and their potential uses in anticancer therapy.
Materials and Methods
Cell Lines, Reagents and Antibodies
GSPs were purchased from Tianjin Jianfeng Natural Products Co. (proanthocyanidins contents > 95%) (Tianjin, China). The possible contaminants of endotoxin contained in GSPs were removed using Affi-Prep Polymyxin Matrix (Bio-Rad). The human breast cancer cell line MCF-7 was obtained from the American Type culture collection (ATCC). The cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin, in a humidified atmosphere with 5% CO2 at 37 °C. Anti-EGFR, anti-VEGF, and anti-MMP9, as well as anti-p53, anti-Caspase-3, anti-β-actin antibodies, and HRP-labeled goat anti-mouse IgG were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). MTT (3-[4, 5-dimethylthia- zol-2-yl]−2, 5-diphenyl tetrazolium bromide) and all other chemicals employed in this study were of analytical grade and purchased from Sigma Chemical (St Louis, MO).
Estimation of Cell Viability
Cells were harvested from the culture during the experimental growth phase and seeded in a 96-well plate at a density of 5 × 103 ~ 1×104 cells/mL in fresh medium and incubated overnight. GSPs were then added to attain the final concentration: (1) 0, 0.001, 0.01, 0.05, 0.1, 0.25, 1 µM for testing IC50 value; (2) 0, 1/2×IC50, IC50, 2×IC50 for analyzing dose-response relationship. After that, the cells were incubated for a further period (1) 48 hours for testing for the IC50 value; (2) 6, 12, 24, or 48 hours for analyzing time–response relationship. At the end of the stipulated period, 20 µl MTT (5 mg/mL) was added to each well and incubated at 37 °C for 4 hours. MTT assay was performed according to the manufacturer’s instruction. The absorbance was measured with a multifunctional microplate reader at 490 nm. The cell viabilities were normalized to the control group. The IC50 value was calculated with origin 8.0 software. All assays were performed in triplicate and mean ± standard deviation (SD) values were used to evaluate cell viability.
Flow Cytometric Analysis
MCF-7 cells (2 × 105) cultured in 25 cm2 flasks with or without GSPs for 24 hours were harvested, fixed with 70% ethanol at −20 °C for 1 hour, and then stained with propidium iodide (PI) solution (100 mg/mL of Rnase A and 40 µg/mL of PI) at 37 °C for 30 minutes. The cell cycle distribution was measured by using the Becton-Dickinson FACScan cytofluorometer (Mansfield, MA, USA).
MCF-7 cells (2 × 105) were cultured in 25 cm2 flasks, and treated with vehicle alone (0.1% DMSO) and various concentrations of GSPs for 48 hours. Then cells were washed twice with phosphate-buffered saline (PBS) and stained with 2 µl of Annexin V-FITC and 5 µl of PI (50 µg/mL) in 1 × binding buffer (10 mM HEPES, pH 7.4, 140 mM NaOH, and 2.5 mM CaCl2) for 15 minutes at room temperature in the dark. Samples were then analyzed using a flow cytometer (Becton Dickinson, Mansfield, MA, USA) and evaluated based on the percentage of cells positive for Annexin V.
Western Blot Analysis
MCF-7 cells were exposed to different concentrations of GSPs for 48 hours. Cell lysates with 50 µg of protein were fractionated and transferred to nitrocellulose membranes. After being blocked with blocking buffer (TBS containing 0.1% Tween 20% and 5% non-fat milk) for 60 minutes at room temperature, the membrane was probed with antibodies, as indicated. The membrane was then incubated with horseradish peroxidase-conjugated secondary antibodies. After repeating the washes in triplicate, the proteins of interest were detected using a chemiluminescence reagent kit (Amersham Pharmacia, Piscataway, NJ).
RNA Isolation and Quantitative RT-PCR (QRT-PCR) Analysis
Total RNA was extracted from harvested cells using Tri-Reagent (Sigma), according to the manufacturer’s instructions. The quality of total RNA was calculated by a NanoDrop 1000 Spectrophotometer (Thermo Scientific). cDNA was obtained from total RNA using a reverse-transcription kit (Thermo Scientific). Primers used in the present study were as follows: 5'-TGA GCC GCG ACT GTG ATG-3' and 5'-GTC TCG GTG ACA AAG TCG AAG TT-3' for human p21; 5'-ATG GAC GGG TCC GGG GAG CA-3' and 5'-CCC AGT TGA AGT TGC CGT CA-3' for human Bax; 5'-GAG TGC ATC ACA AAC CTA CTG G-3' and 5'-TCT CCA TGT CTC TAG GGT AGG C-3' for human Bid; 5'-ACC TCA ACG CGC AGT ACG A-3' and 5'- TGA GGG TCG GTG TCG ATG CT-3' for human Puma; 5'-AGC CTC AAG ATC ATC AGC AAT G-3' and 5'-ATG GAC TGT GGT CAT GAG TCC TT-3' for human GAPDH. qRT-PCR was performed on the RT-PCR 7500 machine (Applied Biosystem, Rotkreuz Zug, Switzerland) using SYBR green and Quantitec primer assays (QIAGEN, Hilden, Germany). Results were analyzed by the comparative ΔΔCt method. GAPDH expression remained relatively consistent across the test samples and can be considered as an internal control.
Statistical Analysis
All data are expressed as means ± SD and were analyzed using the Student’s t-test. Differences were considered significant at the level of P < .05. Statistical analysis was performed by using SPSS 13.0 software (SPSS, Chicago, IL, USA).
Footnotes
Acknowledgments
This work was supported by Key Scientific and Technological Research Plan of Hebei Provincial Department of Health (NO. 20190175). We wish to thank the editor, the associate editor, and the three anonymous reviewers for their helpful comments and suggestions, which have led to an improvement of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Key scientific and technological research plan of Hebei Provincial Department of Health (20190175).
