Abstract
We have tested the hypothesis that a fermented beverage from the macroalgae Hydropuntia eucheumatoides exhibits antioxidant and enzymatic activity. The macroalga was hydrolyzed (maximum hydrolysis yield: 78%) with a mixture of the enzymes viscozyme and lactozyme. Then, the hydrolyzate was fermented with Lactobacillus casei and Saccharomyces boulardii. This beverage contained oligosaccharide prebiotics. The lactic acid, acetic acid, ethanol, methanol, cell count, pH, and heavy metal content of the beverage were determined. All tested heavy metals were either not detected (eg, As) or within the US Food and Drug Administration limits (eg, Fe).
Introduction
Fermented beverages promote health and prevent certain diseases. 1 They have anti-cancer 2 and anti-inflammatory 3 properties, and aid digestion. 4 Seaweed is a suitable raw material to produce fermented beverages, such as that produced from Gracilaria fisheri fermented by Lactobacillus plantarum DW3. 5 Laminaria japonica fermented by Saccharomyces cerevisiae produces a fermentation fluid with low alcohol content. 6
Producing fermented beverages from red seaweed has 2 main steps: hydrolysis and fermentation. Hydrolysis is achieved by chemical and biological methods. Polysaccharides such as cellulose, carrageenan, and agar, under the action of heat, inorganic acids, and enzyme preparations, are broken down into monomers such as glucose and galactose. For example, Kim et al 7 produced lactic acid by pretreatment and hydrolysis of 50 g/L red seaweed at 120°C, digestion for 15 min with 3% (v/v) H2SO4, then fermentation with Lactobacillus paracasei. Yanagisawa et al 8 produced ethanol by pretreatment of 300 g/L fresh Gelidium elegans with 0.1 M aqueous nitric acid at 120°C for 20 min, before adding 5 g of the enzyme meicelase, and then hydrolyzing the mixture at 50°C, pH 5.5 for 120 h. Furthermore, Amamou et al 9 produced ethanol by autoclaving 30 g/L dry Gelidium sesquipedale for 15 min, followed by the addition of 0.6 g haliatas, and then hydrolyzing at pH 5.5 at 37°C for 72 h.
Beverage fermentation of red seaweed involves the growth and development of probiotic strains on a hydrolyzed medium. The probiotic strains are usually lactic acid bacteria (LAB). 10 According to the proposed definition of FAO/WHO, probiotics must be alive and abundant once ingested. However, during recent years, new definitions have been added to probiotic terminology such as “paraprobiotics” (dead/inactive cells of probiotics) and “postbiotics” (healthful metabolites of probiotics), because findings have shown that dead cells (intact or ruptured) could also show significant health impacts on humans.11,12 Consumption of probiotic products has been associated with many health benefits. The effect of whey dairy beverage containing Lactobacillus casei on in vitro and in vivo anti-hyperglycemic activity was determined by inhibition of α-glucosidase and α-amylase activities. 13 Lactobacillus plantarum B719-fermented milk produced anti-osteoporotic effects. 14
According to Son et al, 10 active probiotics of LAB isolated from traditional Korean fermented foods, obtained from strains of L rhamnosus, L plantarum, and L brevis, contain antibiotics that exhibit high β-glucosidase activity and antioxidant properties, and can be safely consumed by people who are lactose-intolerant. Hwang et al 15 used 3 probiotic LAB strains, L rhamnosus, L casei, and L plantarum, to ferment the lactic acid of Gelidium amansii hydrolyzate. Hayisama-ae et al 16 produced fermented beverages from Gracilaria fisheri by treating the seaweed overnight with 0.5% (w/w) potassium metabisulfite, washing the treated seaweed, then mixing it with sugar and water at a mass ratio of 3:1:10. Additionally, L plantarum was fermented for 60 days. Furthermore, the resulting fermentation fluid of red seaweed contains oligosaccharide prebiotics, 16 such as galacto-oligosaccharides that affect bowel function. 17 Torres et al 18 reported the production, properties, applications, and pertinence of galacto-oligosaccharides as prebiotics. Cheong et al 19 reported the production, properties, and potential health and cosmetic applications of oligosaccharides derived from red seaweed.
The literature does not report hydrolysis and fermentation of Hydropuntia eucheumatoides for beverage production, and thus the resulting prebiotic properties are unknown. Accordingly, we performed pretreatment and hydrolysis of this red alga with various enzyme preparations based on viscozyme L (vis), lactozyme 2600 L (lac), and leafgluco L (lea). We fermented H eucheumatoides with L casei and Saccharomyces boulardii, because these are common strains used in beverage fermentation. We analyzed the antioxidant activity and the activity of pertinent enzymes. The oligosaccharide is a prebiotic that increases the antioxidant activity and acts as a substrate that stimulates enzyme production in the fermented strains. 18 We also analyzed pertinent chemical properties.
Our goal was to produce new beverages from H eucheumatoides, and quantitate hydrolysis and fermentation parameters for this seaweed. The resulting beverage exhibits enzymatic and antioxidant activity, and the oligosaccharides increase the activity.
Materials and Methods
Red Seaweed, Bacterial Strains, and Commercial Enzymes
H eucheumatoides (Harvey) Gurgel & Fredericq, 2014—Phylum, Rhodophyta; class, Florideophyceae; order, Gracilariales; family, Gracilariaceae; genus, Hydropuntia, species, eucheumatoides—was collected from Hon Rua Island, Nha Trang City, Vietnam.
The Lactobacillus strain was L casei of KAN Phytochemicals Pvt. Ltd, India, and the Saccharomyces strain was S boulardii CNCM I-745 of Biocodex, France.
Vis [β-glucanase (endo-1,3(4)-) 100 FBG/g] and lac (β-galactosidase) were purchased from Sigma (Novozymes, Denmark), and Lea (glucoamylase) from Leaf Cleantech Pvt. Ltd, India.
Pretreatment and Enzyme Hydrolysis
Pretreatment
Dried H eucheumatoides was mixed with water to a concentration of 20 to 50 g/L, and then pretreated with 5 mM aqueous citric acid (Merck) at 120°C for 15 min.
Effect of Enzyme Formulation on Hydrolysis
The enzyme formulations were as follows: vis, lac, lea, vis + lac, and vis + lea (1 part enzyme per part dry H eucheumatoides, w/w). The hydrolysis conditions were as follows: 30 g dry H eucheumatoides/L, 96 h, 50°C, 150 r/min.
Effect of H eucheumatoides Concentration on Hydrolysis
The H eucheumatoides concentration was as follows: 20, 30, 40, and 50 g/L. Hydrolysis conditions were as follows: 1 part enzyme to 1 part vis and lac (w/w), 96 h, 50°C, 150 r/min.
Fermentation
Starter Inoculum
L casei was inoculated into 200 mL de Man, Rogosa, and Sharpe (MRS) broth and S boulardii into 200 mL malt extract broth (ME broth), then incubated at 30°C for 48 h at a rotation speed of 150 r/min. Then 50 mL aliquots were centrifuged at 6000 r/min for 15 min to collect the cells. The collected cells were then placed into 1 L of H echumatoides hydrolyzate to ferment.
Fermentation and Survey of Population Dynamics
H eucheumatoides hydrolyzate was fermented in a 5-L tank volume bioreactor (Ecell, model 3000, Anyang City, Korea). H eucheumatoides hydrolyzate was treated at 120°C for 15 min. Then the hydrolyzate was fermented at pH 5.2, at 32°C, 100 r/min—with an initial microbial count of 6.3 log colony-forming units (CFU)/mL of L casei and 6.2 log CFU/mL of S boulardii—for 120 h. The population dynamics, such as changes in pH, CFU, and sugar concentration, were followed during fermentation.
Activity of Beverages
Pertinent enzyme activities (β-galactosidase, β-glucosidase, and α-glucosidase) were assayed using a published method. 20 The reaction mixture consisted of 2 mL substrate [0.05% (w/v) para-nitrophenyl b-D-glucopyranoside (p-NPG), lactose, agarose, carbon methylcellulose (CMC), and rice starch], 1 mL enzyme solution, and 0.1 mL citrate buffer (pH 4.8). The mixture was incubated at 50°C for 30 min. The Somogyi method 21 was used to measure the change in sugar concentration, referring to that of agarose, CMC, and rice starch. The resulting blue color was measured, at 500 nm, immediately after incubation. The p-NPG substrate has a yellow color that was measured, at 425 nm, immediately after incubation. The lactose substrate was measured by high-performance liquid chromatography (HPLC: anion-exchange column ICE-AS1 of Dionex, detection RID 6A, Shimadzu Systems of Japan). One unit of activity (U) was defined as the quantity of enzyme that liberated 1 µg sugar from the substrate per minute under standard assay conditions. 21
The percent antioxidant activity (AA%) of the fermentation fluid was assessed by the 2,2-diphenyl-1-picrylhydrazyl hydrate (DPPH) radical scavenging method.
22
The reaction mixture consisted of 0.5 mL sample, control solution, or blank; 3 mL ethanol; and 0.3 mL DPPH radical in 0.5 mM aqueous ethanol. AA% was measured using an ultraviolet–visible spectrophotometer (model 6405; Jenway, UK) at 517 nm:
Analysis
Determination of the chemical composition of the crude material followed AOAC methods, 23 as follows. The nitrogen content of the protein was measured by the Kjeldahl method. Ash was estimated after heating H eucheumatoides at 1500°C for 8 h. Lipid was extracted in a Soxhlet extractor (Soxhlet laboratory extractor EV6, Gerhardt Systems of Germany). Total carbohydrates followed the Dubois method. 24
Determination of the total sugar mass of the H eucheumatoides hydrolyzate followed the Dubois method. 24 The H eucheumatoides hydrolyzate was centrifuged and the supernatant collected for sugar analysis. The sugar concentration was measured using a UV–visible spectrophotometer (model 6405; Jenway, UK). The mass of oligosaccharide is the total mass of the sugar minus the sum of the total mass of the lactose, glucose, and galactose. 25
Determination of the mass of ethanol, methanol, lactose, lactic acid, acetic acid, and monosaccharides was by HPLC (Shimadzu systems of Japan); for example, LC 10AD pump, anion-exchange column ICE-AS1 of Dionex (250 mm × 9 mm column dimensions), detection RID 6A. The eluent was 5 mM aqueous H2SO4 at a flow rate of 0.8 mL/min, temperature 45°C. 8
Determination of the enzyme was by HPLC; for example, column YMC ODS AQ (250 mm × 4.6 mm column dimensions), detection SPD 10 Apv. The eluent was 0.8 mL/min, temperature 45°C, 0.05% (v/v) aqueous methanol prepared in deionized water, detection by UV–visible spectroscopy, at λ = 220 nm. 26
Determination of heavy metals in the fermented fluids refers to As, Pb, Cu, Fe, Zn, Cd, and Hg, which were analyzed by atomic absorption spectrometry (AAS; Thermo Elemental—Model Solaar M6 Dual Zeeman).
Sensory Analysis
The sensory analysis of the beverages was made by the QDA method, in accordance with the ISO 8586: 2012 standard (ISO 2012). Ten assessors had at least 1-y experience in the descriptive sensory evaluation of the fermentation products. The assessors were selected in accordance with their sensory sensitivity based on the correlation value. 27
The attributes examined were selected in a panel discussion and were comprised of smell (red seaweed—fermentative, sterilization, sweet, irritant, and other), color, density, smoothness and viscosity, and taste (red seaweed—fermentative, sterilization, sour, sweet, caprine, salty, bitter, irritant, and other). 28
Statistical Analysis
All experiments were repeated 3 × with duplicate samples, and all results are presented as the mean ± standard deviation. Excel 2010 was used for all statistical analyses.
Results and Discussion
Raw Material Chemical Composition
The chemical composition of H echuematoides was as follows: protein-derived nitrogen, 6.2%; ash, 12%; lipid, 1.5%; and carbohydrate, 80.3% (all percentages are w/w). This seaweed has a quite high mineral content because the seaweed hulls are calcified by seawater and some sand particles adhere to the surface. The carbohydrates of this seaweed are mainly agar, as well as some starch and cellulose. Thus, when analyzing the composition of the monosaccharides, there will be only 2 main sugars: galactose and glucose. A high carbohydrate content indicates a suitable material for producing fermented beverages and volatile organic acids (VOAs). 29
Enzymatic Hydrolysis
Effect of Enzyme Formulation on Hydrolysis
Different enzyme formulations resulted in different concentrations of lactose, glucose, galactose, and oligosaccharide (Table 1).
Sugar Content and Yield of Enzymatic Hydrolysis. a

Hydrolysis yield = [mass of sugar total hydrolyzate (g/L)/mass of H echuematoides (g/L)] × 100%.
Alone, vis and lea enzymes tended to produce more glucose than galactose and lactose (although the vis enzyme produced more galactose than the other 2 sugars). In contrast, the lac enzyme produced more lactose and galactose than glucose. The lac + vis experiment had the highest hydrolysis yield, 78%; and a high concentration of glucose, galactose, and lactose. The other experiments resulted in lower efficiencies, ranging from 46% to 67%. Therefore, a combination of lac and vis is suitable for saccharification: reducing sugar concentration, 10.1 g/L; oligosaccharide concentration, 13.5 g/L. This formulation is suitable for fermentation by microorganisms.
Various researchers have studied enzymatic red seaweed hydrolysis; for example, Gelidium elegans with meicelase hydrolyzes cellulose, starch, and 1,3-glucan 8 ; and Gelidium amansii hydrolysis with a mixture of celluclast 1.5 L and viscozyme L enzyme preparations. 30 Thus, no specific enzyme is necessary to hydrolyze the agar and carragennan of red seaweed. 30 We used a combination of vis and lac because this enzyme formulation contains β-galactosidase, β-glucanase, α-glucanase, and pectinase, which hydrolyzes the cellulose, agar, and starch of H echuematoides. 31
Effect of H echuematoides Concentration on Hydrolysis
Different concentrations of H echuematoides resulted in different concentrations of lactose, glucose, galactose, and oligosaccharide (Table 2).
Sugar Concentrations and Hydrolysis Yields at Various H echuematoides Concentrations.
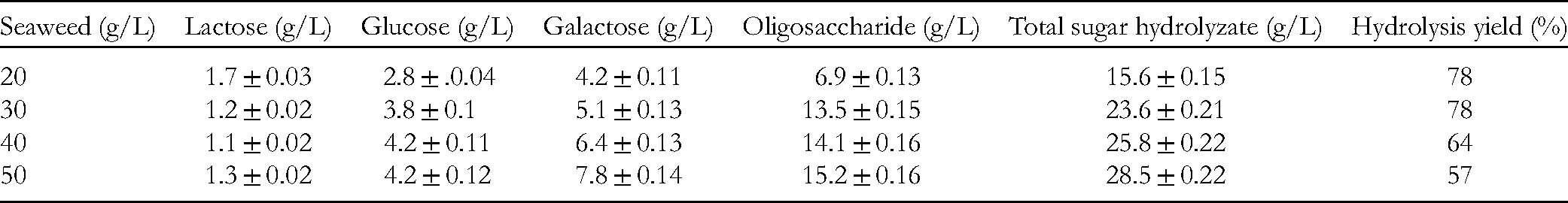
Table 2 shows that during saccharification, the sugar concentration increased in accordance with increasing H echuematoides concentration. However, higher H echuematoides concentrations decreased the hydrolysis yield. Seaweed concentrations of 40 to 50 g/L are highly viscous and because the enzyme is encapsulated in a polysaccharide, the enzymatic activity and hydrolysis yield (57%-64%) are low. However, a dilute hydrolyzed fluid is suitable for fermentation. At 20 and 30 g/L, the hydrolysis efficiency is nearly equal, but because 30 g/L produces a higher concentration of reducing sugars, it is more suitable for fermentation.
Thus, H echuematoides is easily hydrolyzed by enzymes and the hydrolyzate is suitable for processing fermented beverages. Hydrolysis is not thorough, which results in large quantities of oligosaccharides with prebiotic activity for the fermented product. The hydrolysis of our study differs from that of seaweed hydrolysis for the production of ethanol, lactic acid, and other VOAs. This is reflected by the lower sugar concentration of this study, 10 g/L; lower than the 40 g/L value of Yanagisawa et al 8 and 30 g/L of Kim et al. 7 In our study, the pretreatment conditions and enzymatic hydrolysis were straightforward and gentle, in contrast to the complex and high-acid conditions of other studies; for example, pretreatment and hydrolysis of red seaweed at a concentration of 30 to 50 g/L, hydrolysis at 120°C for 15 min with 3% (v/v) aqueous H2SO4, then fermentation with L paracasei for lactic acid production. 7 Another study 8 reported Gelidium elegans, at a concentration of 300 g fresh /L, pretreated with 0.1 M aqueous nitric acid at 120°C for 20 min, where the researchers added 5 g meicelase and then carried out hydrolysis at 50°C, pH 5.5. The study reported production of ethanol after 120 h. 8
Fermentation
Population Dynamics During Fermentation
We found considerable differences in the numbers of L casei and S boulardii (Figure 1a) over the course of fermentation. In the early phase of fermentation (hours: 0-24), the number of cells increased slowly; these strains were adapting to the fermentation medium. L casei had a cell count of 7.4 log CFU/mL and that of S boulardii was 6.4 log CFU/mL. During the next time frame (hours: 24-96), the cell count of the 2 strains increased sharply; L casei had a cell count of 9.2 log CFU/mL and that of S boulardii was 8.4 log CFU/mL. In the end time frame (hours: 96-120), the number no longer increased; L casei had a cell count of 9.4 log CFU/mL and that of S boulardii was 8.5 log CFU/mL. Thus these strains are well-adapted to the hydrolyzate of H echuematoides.

Population dynamics during 120 h of fermentation by L caseii and S boulardii: (a) log CFU/mL, (b) pH, and (c) sugar concentration.
We determined the pH change during fermentation (Figure 1b). In the early phase of fermentation (hours: 0-24), the pH only decreased a little; the pH of L casei fermentation broth was 4.9 and that of S boulardii was 5.03. In the next phase (hours: 24-72), the pH continued to decrease; the pH of L casei fermentation broth was 4.58 and that of S boulardii was 4.72. In the final phase (hours: 72-120), the pH of S boulardii broth no longer decreased by much: pH 4.61; whereas the pH of L casei broth was low, 3.52, because of the comparatively higher quantity of acids produced during fermentation for the latter (Table 3).
Concentrations of Substances Formed After 120

The initial quantity of sugar was 23.6 g. In the early phase (hours: 0-24), the rate of fermentation of the 2 strains was slow; the sugar content decreased slightly, where the residual sugar concentration was 20 to 21 g/L. In the next phase (hours: 24-72), the 2 strains used up all of the monosaccharides in the solution; the residual sugar concentration was 12 to 14 g/L. In the next phase (hours: 72-120), S boulardii used only a small quantity of sugar because monosaccharides were essentially unavailable in the residual sugar, remaining at a concentration of 12.8 g. However, L casei continued to hydrolyze and ferment; the residual sugar concentration was 5 g/L. Thus L casei has the ability to produce enzymes that hydrolyze oligosaccharides into simple sugars for use (Table 3).
In a fermentation dynamics study, we found that the 2 tested strains were suitable for use in H echuematoides hydrolyzate broth. The number of cells in the 2 strains increased over time and peaked at 96 h. The pH of both strains decreased, especially L casei, because this strain produced comparatively more acid over time. The 2 strains fully utilized the monosaccharide content, but L casei continued to hydrolyze and ferment the residual sugar. This fermentation indicates that in terms of the number of microorganisms, residual sugar, and pH, our protocol is suitable for making fermented drinks.
Chemical Properties
L casei and S boulardii are suitable for hydrolysis and grow well under our experimental fermentation conditions (Table 3). The fermentation product of L casei produced mainly lactic acid, lowering the pH to 3.52. In contrast, S boulardii produced some lactic acid and ethanol, and the pH was 4.6. Both the quantity and type of oligosaccharide differed. The fermentation fluid of L casei had a low oligosaccharide content, whereas that of S boulardii had a high oligosaccharide content.
Thus, L casei is compatible with hydrolysis and converting the oligosaccharide to lactic acid. In contrast, S boulardii can only ferment reducing sugars in the hydrolyzate, rather than oligosaccharides in the fluid. With L casei, the HPLC oligosaccharide peak decreased in height, corresponding to an increase in the lactic acid peak. However, with S boulardii, the oligosaccharide peak was unchanged and the lactic acid peak small. Fermentation of beverages from H echuematoides using L casei and S boulardii converts the hydrolyzate into lactic acid and ethanol. The products of this process include the biomass of the 2 strains, oligosaccharides, and enzymes.
The fermentation time of 120 h is sufficient for metabolism. This is similar to the study of fermented agar substrate from red seaweed for 72 h 7 and fermented beverages from Gracilaria fisheri with L plantarum over the course of 60 days.5,16 A longer fermentation time was used because Gracilaria fisheri ferments directly without hydrolysis. In our research, the production time of fermented beverages from H echuematoides is short and from that perspective is suitable for industrial-scale production.
The chemical composition of the fermentation fluid shows that many compounds have been produced: organic acids and oligosaccharides. These substances function to enhance the antioxidant activity of fermented beverages; for example, lactic and acetic acids have strong antioxidant activity, 32 and galacto-oligosaccharide has antioxidant activity. 1 The combination of organic acids and galacto-oligosaccharide will increase the antioxidant activity of seaweed fermented beverages.
Activity of Fermented Beverage
Antioxidant Activity
DPPH free-radical results of fermented fluids from L casei and S boulardii differed in terms of AA%. The AA% coefficients were as follows: S boulardii, 64.8 ± 0.4%; L casei, 85.9 ± 0.6%; and 2 mg/L ascorbic acid, 95.5 ± 0.7%. The difference between the 2 samples was considerable, yet still within ∼20% of each other. Fermentation with L casei produced a large quantity of lactic acid; thus, it had higher antioxidant activity than the S boulardii product. The antioxidant capacity of this was also high and mainly depended on the quantity of oligosaccharides in the fluid. Fermented beverages from the 2 strains have high antioxidant capabilities. For example, Suryavanshi et al 33 reported the antioxidant activity of S boulardii ATCC796 (20%-86% with 25-500 µL of sample), and Chooruk et al 34 reported the antioxidant activity of L casei (29.4% with 100 µL of sample). The antioxidant activity of this study is different from other studies due to the oligosaccharide-containing fermentation fluid; for example, Cheong et al 19 reported the antioxidant activity of galactose oligosaccharide (65% with 200 µL of sample).
Enzymatic Activity
During fermentation, the H echuematoides hydrolyzate of L casei and S boulardii secreted several enzymes able to convert sugars into fermented products. In this study, we analyzed the activity of β-galactosidase (for lactose and agarose), β-glucosidase (for p-NPG and CMC), and α-glucosidase (for rice starch). Figure 2 shows the results.

Enzymatic activity of L casei and S boulardii in fermentation fluid.
L casei performed well in the H echuematoides hydrolyzate medium and secreted many enzymes. β-Galactosidase performed strongly with agarose and moderately with lactose. The total activity of β-galactosidase, 15 UI, was considerably higher than that of β-glucosidase, 2.4 UI; and α-glucosidase, 0.3 UI. On HPLC examination, a peak with a retention time of 5.365 min (corresponds to β-galactosidase) had a much larger area than that with a retention time of 3.848 min (corresponds to β-glucosidase).
In contrast, fermentation by S boulardii produced very little β-galactosidase, yet it produced more than that by L casei. β-Glucosidase from S boulardii had an activity of ∼7.1 UI, whereas that of β-galactosidase was only 0.5 UI and that of α-glucosidase 3.1 UI. Because of the low β-galactosidase activity, the residual oligosaccharide cannot be hydrolyzed; thus, the oligosaccharide concentration of 12.8 g/L in the fermentation fluid remained unchanged compared with before fermentation. In contrast, L casei secreted many β-galactosidase enzymes; thus, oligosaccharide was hydrolyzed into glucose, galactose, and lactose, available for L casei to use.
Thus, fermentation with L casei produced substantial levels of β-galactosidase and β-glucosidase, but fewer oligosaccharides, whereas S boulardii produced β-glucosidase and many oligosaccharides. Based on these results, the fermented beverages from L casei and S boulardii are probiotic drinks.
Son et al 10 reported the enzymatic activity of Lactobacillus strains that make use of β-galactosidase and β-glucosidase. LAB strains were isolated from mushrooms and fermented in an MRS medium. Yuksekdag et al 35 showed that β-glucosidase, produced by L casei SC1, bioconverted isoflavone glycosides to aglycones. Arevalo-Villena et al 36 showed that a Saccharomyces strain produced glucanase and pectinase. In our study, 2 strains fermented in the hydrolyzed fluid also produced these enzymes.
Heavy Metals in Fermented Fluid
In the fermentation solutions of L casei and S boulardii, As, Pb, Cd, and Hg were not detected. The fermented fluid of L casei contained Cu, 0.011 mg/L; Fe, 0.16 mg/L; and Zn, 0.14 mg/L, and that of S boulardii Cu, 0.011 mg/L; Fe, 0.09 mg/L; and Zn, 0.14 mg/L. These concentrations are below the permitted limits of the US Food and Drug Administration (FDA). 37
Sensory Characteristics of Probiotic Fermented Beverage From H echuematoides
In Table 4, the sensory evaluation results of the assessors for the red seaweed fermented beverage show some main characteristics of this product. The seaweed fermented beverage from L casei and S boulardii has the same color, density, smoothness, and viscosity. The color is similar to that of white agar, the density is dense/runny, the smoothness is sandy/floury/smooth, and the viscosity is ductile. The difference between the 2 strains is the smell and taste. The smell of L casei is characteristic of Lactobacillus fermentation. The S boulardii product smells like honey and sugar; a smell of light isoamyl acetate. The taste of the L casei product is sour, whereas that of S boulardii has a slightly sour and slightly sweet taste.
Sensory Characteristics of Probiotic Fermented Beverage From H echuematoides.

The main distinctions of this drink from yogurt and yakult are the smell, color, and viscosity. The smell of this product is characteristic of agar saccharificaton and fermentation. The color is similar to that of agar. The product is brighter than yogurt and yakult, and the viscosity is higher than yakult, but lower than yogurt. Future studies should focus on investigating consumers’ sensory acceptance of the red seaweed fermented beverage.
Conclusions
Our goal was to produce a fermented beverage based on H eucheumatoides, an agar producing species with high mineral content. We optimized the enzyme formulation and H eucheumatoides concentration to maximize the hydrolysis yield. Various microbes are suitable for fermentation, have appropriate antioxidant and enzymatic activity, and are probiotic. The heavy metal content is within US FDA specifications. We thus confirmed our hypothesis that a fermented beverage from H eucheumatoides exhibits antioxidant and enzymatic activity. Future studies should focus on the oligosaccharide activity of fermented seaweed beverages, and investigate consumers’ sensory acceptance of this product.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211066145 - Supplemental material for Probiotic Fermented Beverage From Macroalgae
Supplemental material, sj-docx-1-npx-10.1177_1934578X211066145 for Probiotic Fermented Beverage From Macroalgae by Vo T. Trung, Tran Van Huynh, Pham D. Thinh, Pham T. San, Truong H. Bang and Nguyen T. Hang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vietnam Academy of Science and Technology (grant no. ĐLTE00.05/20-21) and ĐTCS 2020 of NITRA.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

