Abstract
Objective
The study examined the impact of seasonal variation on the quality of bioethanol produced from bamboo juice through fermentation and distillation, focusing on differences between dry and rainy seasons. Bamboo juice samples were supplemented with small amounts of mango fruit at the verge-stage to enhance enzymatic hydrolysis and fermentation efficiency.
Methods
Fresh bamboo juice was collected from bamboo farms using “mbeta” and allowed to ferment at room temperature. To enhance hydrolysis and fermentation efficiency, the fermenting juice was supplemented with mango fruit at the verge stage. The fermented juice was distilled at a controlled temperature of 78 °C to obtain high-quality bioethanol.
Results
The total soluble solids (TSS) of bamboo juice were significantly higher in the dry season (10.01 ± 0.04 °Brix) than in the rainy season (5.30 ± 0.08 °Brix), reflecting a higher concentration of fermentable sugars during the dry period. The results demonstrated higher fermentation efficiency in the dry season, achieving a maximum alcohol by volume (%ABV) of 5.15%, compared with 4.14% during the rainy season. The findings showed bioethanol concentrations of 39% (v/v) in the dry season compared with 35% (v/v) in the rainy season for the first 100 mL aliquot from distillation. Subsequent redistillation increased bioethanol purity to 87% (v/v), rendering it suitable for fuel-grade applications.
Conclusion
These results demonstrate that bamboo juice harvested during the dry season contains higher levels of fermentable sugars and yields superior bioethanol quality, highlighting seasonal timing as a critical parameter for optimizing bamboo juice-based bioethanol production.
Introduction
The global shift toward renewable energy has emerged as one of the most pressing imperatives of the twenty-first century, driven by the urgent need to mitigate climate change and reduce the reliance on fossil fuels. 1 As sustainable energy alternatives, biofuels, particularly bioethanol, have gained popularity in accordance with international frameworks of the Paris Agreement. 2 A clean-burning, biodegradable fuel, bioethanol is well known for its ability to drastically reduce greenhouse gas emissions as compared to traditional petroleum-based fuels. 1 According to the International Energy Agency (IEA, 2023), global bioethanol production exceeds 110 billion liters annually, with forecasts indicating an increase to more than 140 billion liters by 2030. 3 The major producers, including the United States of America, Brazil, and China, continue to rely predominantly on first-generation feedstocks, including corn, sugarcane, and wheat, to meet production demands. 2
However, the growing concerns over food security, land use conflicts, and water scarcity have led to the exploration of non-food and regionally abundant biomass resources for sustainable bioethanol production. 1 Besides, the research focus has expanded to second-generation biofuels and underutilized plant materials and byproducts that do not compete with food supply. 4 In this context, tropical and subtropical countries, particularly those with bimodal rainfall patterns, offer unique opportunities and challenges related to seasonal variability in feedstock composition and fermentation behaviors. 5 Studies from Southeast Asia and Latin America demonstrate that bioethanol yield varies seasonally by 20–40% depending on environmental factors, including temperature, humidity, and rainfall. 6 Fluctuations are often linked to changes in the concentration of total soluble solids (TSS), microbial activity, and fermentation efficiency. 4 For instance, higher TSS in the dry seasons enhances fermentation rates but results in lower yields if temperatures exceed optimal microbial thresholds. 6 Besides, cooler, wetter conditions slow fermentation but lead to higher bioethanol purity due to more stable substrate-microbe interactions. 5 Despite such findings, seasonal dynamics are rarely accounted for in bioethanol production planning, especially in tropical developing countries where climatic variations are pronounced. In sub-Saharan Africa, the potential for bioethanol generation remains largely untapped. Although the region contributes less than 5% of global bioethanol production, it possesses vast biomass resources ranging from crop residues, grasses, to fruit wastes and wild plants that remain underexploited. 7 However, bioethanol pilot programs have been established in countries including Ghana, South Africa, Nigeria, and Kenya, mostly utilizing food-based feedstocks including maize and sugarcane. 8 However, these approaches ignore the fundamental problem of seasonally fluctuating biomass performance and create food versus fuel considerations. 8 Besides, most African settings still lack the technological capability to monitor and modify fermentation processes in response to seasonal variations. 7
In Tanzania, interest in bioethanol is steadily growing in alignment with the National Energy Policy (2015) and the Vision 2025 strategy, which emphasize the need to reduce the reliance on imported petroleum fuels and promote clean, affordable, and locally sourced energy alternatives. 9 The Southern Highlands of Tanzania, particularly Iringa, Njombe, and Mbeya regions are endowed with extensive stands of Oxytenanthera abyssinica, a native bamboo species recognized for its rapid growth, high biomass yield, and environmental adaptability. 10 While most studies on bamboo have focused on its structural fibers, shoots, and leaves for lignocellulosic bioethanol production using acid or base pretreatment, which is not environmentally friendly, the pure juice extracted from bamboo culms presents an underexplored but promising feedstock for bioethanol production. 11
Bamboo juice contains natural sugars, including glucose, sucrose, and fructose, and a significant amount of complex carbohydrates, primarily in the form of polysaccharides including cellulose, hemicellulose, and starch compounds. 12 These complex sugars are not readily fermentable and must first be broken down into simple fermentable sugars, including glucose and fructose, to enable efficient bioethanol production. 13 The present study utilized natural enzymes from mango fruit at the verge-stage (mangoes at the early ripening stage), including amylase, invertase, cellulase, and pectinase, to aid in the hydrolysis of complex polysaccharides into fermentable sugars. 13 At this stage, the fruit is nearly mature but still maintains firmness and a relatively lower sugar concentration compared to fully ripe mangoes. This unique biochemical state makes mangoes at the verge-stage a valuable, inexpensive biocatalyst for fermentation, primarily due to their enzymatic activity.
Investigating the influence of seasonal variations is critical to optimizing bioethanol yield and purity. The amylase enzymes naturally present in mangoes at the verge-stage actively facilitate the hydrolysis and fermentation of bamboo juice, thereby improving sugar conversion efficiency. The process not only increases the overall bioethanol output but also reduces dependence on costly commercial enzymes. Moreover, the approach promotes sustainable, locally driven clean energy solutions. This study investigates seasonal variations and evaluates verge-stage mango supplementation as an effective bio-enhancement strategy for bioethanol production from bamboo juice under contrasting seasonal conditions. By integrating locally accessible resources including mangoes at the verge stage, bioethanol production becomes more environmentally friendly, economically viable, and adaptable to regional climatic and feedstock conditions.
Materials and Methods
Collection of Bamboo Juice and mango Fruits
Bamboo juice samples were collected from local farmers in Kipaduka Ward, Kilolo District, Iringa Region, Tanzania. The site is geographically located at latitude −7.59759° (7° 35’ 51” S) and longitude 36.03576° (36° 2’ 9” E), with the Open Location Code grid reference 6G4RC22P + X8. Bamboo culms were manually cut using a sharp knife to extract the juice, following the method previously described. 10 The locally recognized bamboo container known as Mbeta was used for collecting and transporting the juice into clean storage containers. To reduce impurities, the juice was filtered to remove suspended particulates before measurement. Key parameters, including pH, TSS, temperature, specific gravity (SG), and percentage alcohol by volume (%ABV), were recorded after collection. Fresh bamboo juice was then transported to the Chemistry Laboratory for processing and fermentation. Figure 1 provides an overview of the collection and fermentation procedures. Besides, mango fruits at the verge-stage were collected from Mashine Tatu market in Iringa Municipality. The fruits were thoroughly washed with distilled water, peeled using a sterilized knife, and then ground using a mortar and pestle for use as a natural enzyme source in the fermentation process. The same procedures were consistently followed throughout the rainy and dry seasons.

Bamboo juice collection for laboratory fermentation.
Optimization of the Fermentation Process Using mango Fruit at the Verge Stage
To determine the optimal supplementation amount for the dry and rainy seasons, four different quantities of mango fruit at the verge-stage (10 g, 20 g, 30 g, and 40 g) were prepared. The fruits were peeled, washed with distilled water, ground into a fine pulp using a clean mortar and pestle, and then thoroughly mixed with 1 liter of freshly filtered bamboo juice. Each mixture was poured into separate 2.5-liter fermentation vessels and allowed to ferment at room temperature for over 312 h in both seasons. A 312-h fermentation period was selected based on preliminary trials, which monitored sugar consumption and bioethanol production, indicating that bioethanol levels plateaued at this point, signifying the completion of fermentation. 13
Fermentation performance was assessed by monitoring key parameters, including pH, TSS, SG, and %ABV, throughout the process in the dry and rainy seasons. The experimental design enabled the evaluation of how varying concentrations of mango-derived enzymes influence the rate and efficiency of fermentation, ultimately identifying the most effective mango supplementation amount for maximizing bioethanol yield from bamboo juice.
Fermentation of Bamboo Juice
Fermentation of bamboo juice was carried out at room temperature in both seasons. Each 10-liter fermentation vessel was filled with 8 liters of freshly extracted bamboo juice and supplemented with 240 g of mango fruit at the verge-stage in each season. This supplementation amount was determined based on the optimization experiments described in section 2.2. Fermentation proceeded under anaerobic conditions to facilitate optimal microbial activity. The entire process was monitored over 312 h, during which critical parameters, including temperature, pH, SG, %ABV, and TSS, were measured every 12 h to evaluate fermentation performance and bioethanol yield in both the dry and rainy seasons.
Distillation of Fermented Bamboo Juice Broths
Upon completion of the fermentation process, bamboo juice broths were carefully filtered using a clean, fine-mesh sieve to remove suspended solids and fruit residues, resulting in a clear liquid suitable for distillation. The clear broth was then subjected to a distillation process that exploits the differences in boiling points among the mixture's components, primarily to isolate bioethanol from water and other volatile compounds. Distillation was carried out at a constant boiling temperature of 78 °C, corresponding to the boiling point of bioethanol under atmospheric pressure. From 3 L of fermented bamboo juice broth, the first 100 mL fraction of bioethanol was collected after approximately 15 min of distillation, while subsequent aliquots were collected at 10-min intervals. The resulting liquid was distilled using the temperature-controlled laboratory distiller (VETA- Dar es Salaam) to obtain bioethanol. An electrically powered distillation unit equipped with a Delta Temperature Controller (Model DTA4848R0) was used to ensure precise and stable temperature regulation throughout the process. The vapors generated during distillation were condensed and collected in successive 100 mL fractions. Each distillate fraction was collected in sterile, airtight BOECO GERMANY containers to prevent contamination and evaporation. The alcohol content of each fraction was determined using a Gay-Lussac alcoholmeter (Type 2110), providing accurate readings of alcohol concentration. To further improve the purity and bioethanol concentration, redistillation was performed under the same temperature conditions. This step was essential to reduce residual water content and refine the final product quality. The redistilled bioethanol was again collected in 100 mL increments and subjected to the alcohol content analysis, ensuring consistency, quality, and suitability for potential biofuel or industrial applications. A schematic overview of the bioethanol production process, including fermentation and distillation steps, is illustrated in Figure 2.

Bioethanol production process.
Determination of Specific Gravity
The SG of bamboo juice was measured to estimate the concentration of fermentable sugars before fermentation, which correlates with potential bioethanol yield in both seasons, the dry and rainy. The SG, reflecting the liquid's density relative to water, indicates total dissolved solids, primarily sugars in the juice. A precision glass hydrometer with a range of 0.980 to 1.160 was used for measurements. Before testing, juice samples were filtered to remove suspended solids and maintained at approximately 25 °C for consistency. Each sample was placed in a 100 mL measuring cylinder, and the hydrometer was carefully immersed without touching the cylinder wall. Readings were taken at the lower meniscus of the liquid. Measurements were conducted on samples collected during rainy and dry seasons to compare seasonal variations in SG.
Determination of Total Soluble Solids for Bamboo Juice
To measure the concentration of sugars and other dissolved solids in bamboo juice, TSS was determined using a digital refractometer (Model: ERMA, TOKYO). Bamboo juice samples were collected during rainy and dry seasons to allow for seasonal comparison. Before measurement, the refractometer was calibrated using distilled water to ensure accuracy. A few drops of each juice sample were placed on the refractometer's prism, and the refractive index was measured. The TSS values were expressed in degrees Brix (°Brix), where 1 °Brix represents 1 g of sucrose per 100 g of solution (% w/w). 14 Although bamboo juice contains a mixture of sugars, amino acids, organic acids, and other solutes, the °Brix value provides a practical approximation of total solute concentration. The measurement serves as a reliable and rapid indicator of the available fermentable sugars and is used to monitor sugar depletion during the fermentation process.
Analytical Methods for Bioethanol Quantification
The quantification of bioethanol produced was carried out by measuring the total volume of bioethanol collected after the completion of both fermentation and distillation processes for all seasons. Following distillation, the bioethanol fractions were carefully collected in separate, clearly labeled bottles to avoid cross-contamination and ensure accurate tracking of yield from each batch. To determine the quantity of bioethanol, each sample was measured using a measuring cylinder (AXIOM, Germany), ensuring accurate volumetric readings. The cumulative volume of distillates from all fractions was recorded in milliliters (mL), representing the final bioethanol yield for each fermentation setup in each season. The method provided a straightforward yet reliable approach to assessing the efficiency of the fermentation and distillation processes in converting bamboo juice into bioethanol in both seasons.
pH Measurement
The pH of bamboo juice was monitored throughout the fermentation process to assess changes in acidity, which directly influences microbial activity and enzyme performance in the dry and rainy seasons. A calibrated digital pH meter (Model HI98129, HANNA Instruments, USA and Romania) was used for all measurements. Prior to use, the pH meter was calibrated with standard buffer solutions at pH 4.0 and 7.0 to ensure measurement accuracy and reliability. The measurement uncertainty of the resulting pH readings was estimated to be ±0.02 units, indicating high reliability and reproducibility of the measurements across all samples. The pH readings were taken every 12 h over a 312-h fermentation period. This regular monitoring allowed for observation of dynamic pH trends associated with sugar metabolism, organic acid formation, and microbial growth. Measurements were conducted on bamboo juice broths from rainy and dry seasons to facilitate seasonal comparison. The resulting data provided essential insights into the evolution of acidity and its impact on fermentation efficiency and microbial stability.
Determination of the Quality of Bioethanol
The quality of bioethanol was assessed by measuring the alcohol concentration of each sample using a Gay-Lussac alcoholmeter (Type 2110), which determines alcohol content based on liquid density. Alcohol levels were recorded as a percentage of alcohol by volume (v/v) for standardized comparison across samples in the dry and rainy seasons. Assessments were performed on fermented bamboo juice broths during the rainy and dry seasons. The approach allowed evaluation of seasonal effects on bioethanol quality, providing valuable insights into the suitability of bamboo juice as a feedstock under varying environmental conditions.
Determination of Percentage Alcohol by Volume Using Specific Gravity
The %ABV during fermentation was determined by measuring the change in SG of bamboo juice before and after fermentation in the dry and rainy seasons. The method is based on the principle that a decrease in SG reflects the conversion of sugars into bioethanol. Measurements were taken using a hydrometer, and %ABV was calculated using equation 1.
In the formula, the “Initial SG” represents the density of the juice before fermentation, corresponding to sugar content, while the “Final SG” indicates residual sugars post-fermentation. The constant 131.25 is an empirically derived factor that converts SG difference into alcohol concentration by volume, accounting for density differences between bioethanol and water. 15 This procedure was applied to samples collected during rainy and dry seasons, enabling seasonal comparison of fermentation efficiency and alcohol yield. The resulting %ABV values provided important insights into the potential of bamboo juice as a bioethanol feedstock under varying environmental conditions.
Bioethanol Analysis with Gas Chromatography-Flame Ionization Detector
Representative samples of bioethanol were analysed by the Gas Chromatography equipped with a Flame Ionization Detector (GC-FID) (8890 Series GC) to determine the quality of bioethanol produced. Bioethanol analysis was performed following the method as described in the previous study. 16 Samples were placed in 2 mL autosampler vials with 250 µL inserted and injected into the GC system (8890 Series GC, operated with OpenLab CDS 2.2 software). Separation was carried out using an 8890 GC equipped with a flame ionization detector (FID) set at 250 °C, with airflow of 400 mL/min, hydrogen at 30 mL/min, and nitrogen at 25 mL/min. A split/splitless inlet was operated at 250 °C in split mode with a 50:1 ratio, while helium served as the carrier gas in constant flow mode at 2.0 mL/min.
Data Analysis
Data analysis was performed using Microsoft Excel (2016) functions (average, standard deviation, max, min, etc), where descriptive statistics were computed and visualized through figures generated within the software. 17 To evaluate differences in bioethanol production between bamboo juice broths fermented during rainy and dry seasons, a one-way analysis of variance (ANOVA) was conducted. 17 The statistical test assessed whether there were significant mean differences in bioethanol yields across the two seasonal groups in the dry and rainy seasons. Measurements from bamboo juice samples collected in both seasons allowed for a comprehensive comparison of fermentation performance and bioethanol output under varying environmental conditions, with all data taken in triplicate to ensure precision and reproducibility. The standard repeatability was determined to be within ±5% of the mean value to ensure precision and reproducibility of the fermentation performance and bioethanol output reported for both seasons. To assess the quality of bioethanol, further analysis was performed using GC-FID.
Results and Discussion
In this study, fermenting bamboo juice was supplemented with scant mango fruit at the verge-stage (BM). The study compares bioethanol generation across the dry and rainy seasons to study the influence of seasonal variability on fermentation efficiency and the quality of bioethanol produced. The %ABV during the dry (%ABVD) and rainy (%ABVR) seasons, SG during the dry (SGD) and rainy (SGR)), temperature during the dry (T °C D) and rainy (T °C R)), and TSS during the dry (TSSD) and rainy (TSSR) seasons, respectively, were determined. A comparative analysis of seasonal variability was conducted to assess the effectiveness of fruit in enhancing bioethanol yield. The study optimized bioethanol production by supplementing bamboo juice with mango fruit at the verge-stage. The best results were achieved using 30 g of fruit per 1 L of juice, which provided efficient enzymatic hydrolysis and the highest %ABV. Lower amounts, 10 g and 20 g, resulted in reduced enzyme activity and lower yields, while 40 g caused excessive pulp content, which hindered fermentation efficiency and led to a slight decline in %ABV compared to the 30 g treatment. Thus, 240 g of fruit per 8 L of bamboo juice was established as the optimal ratio for maximizing bioethanol production without causing substrate inhibition in both seasons.
Bamboo Juice Supplemented with mango Fruit at the Verge Stage
Significant variations in fermentation and distillation were observed for the bamboo juice from the dry and rainy seasons, with distinct differences in %ABV, pH, SG, TSS, and temperature.
Total Soluble Solids
The trend in TSS serves as a critical indicator of fermentation dynamics and sugar utilization across the dry and rainy seasons. Variations in TSS profiles between the dry and rainy seasons reflect the influence of environmental conditions on microbial activity, fermentation efficiency, and substrate conversion. Results reveal that during the dry season, TSS declined from 10.01 °Brix to 2.13 °Brix during the 324 h, indicating a prolonged fermentation with steady microbial activity. However, a decline in TSS from 10.01 to around 2.1 °Brix was observed by day 6 (144 h) and remained constant thereafter. Figure 3 shows the TSS of bamboo juice with mango fruit during the rainy and dry seasons.
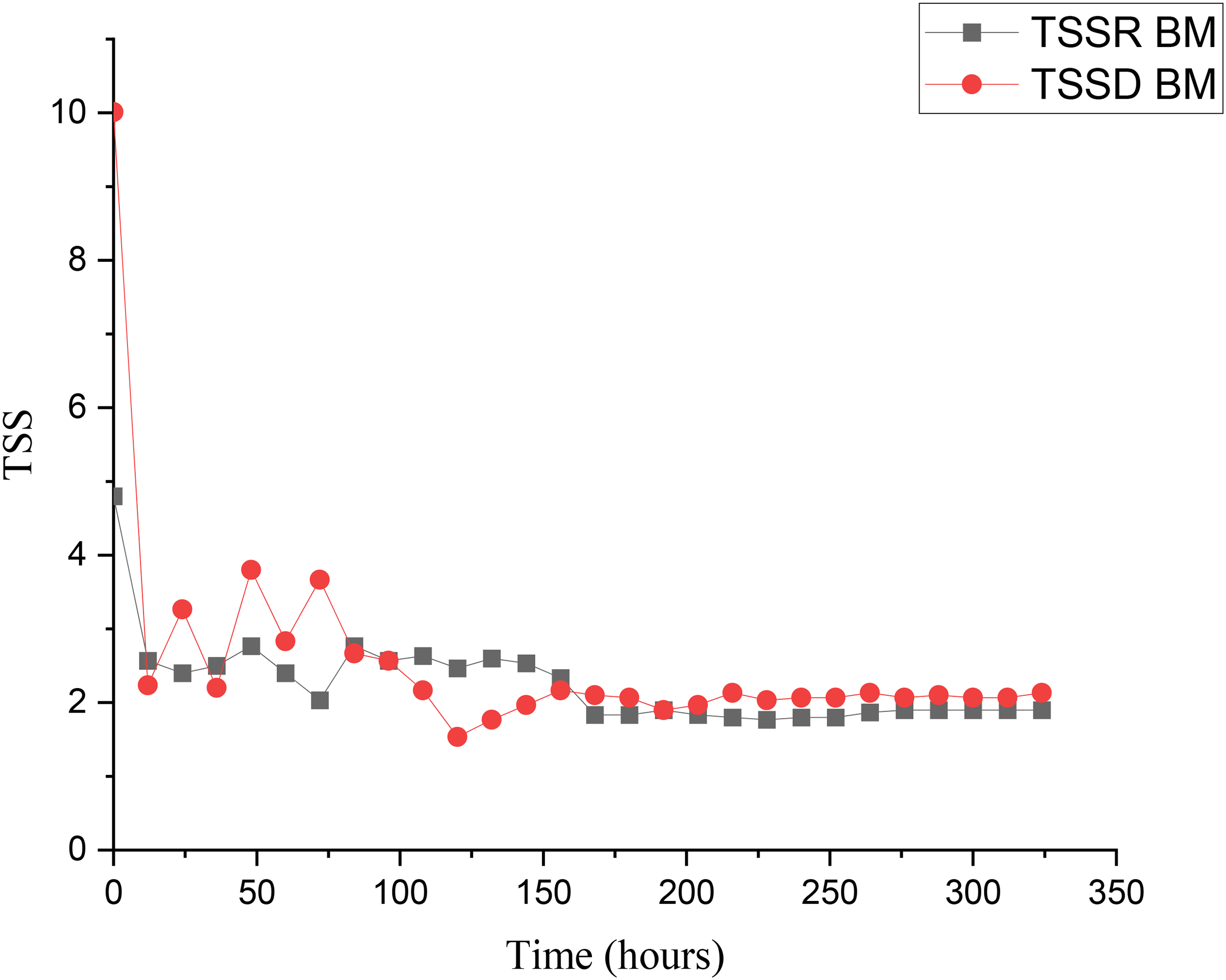
Total soluble solids variation of bamboo juice broths supplemented with mango fruit during rainy and dry seasons.
The TSSD batch commenced at 10.01 °Brix, dropped sharply to 3.30 °Brix within 12 h, followed by a slower decline to 1.9–3.8 °Brix, reflecting rapid early sugar metabolism and later slowdown due to nutrient depletion and bioethanol build-up. A p-value of 0.0061 (p < 0.05) indicates a statistically significant difference during the dry and rainy seasons, confirming a strong correlation between sugar consumption and the fermentation timeline, and underscoring the critical role of monitoring TSS in bamboo juice to enhance fermentation efficiency. Findings align with previous results on a related study. 18 Besides, in the rainy season, TSS declined from 5.30 °Brix to 1.7 °Brix, indicating efficient and consistent fermentation under moderate temperatures and higher moisture, which is consistent with the observations reported earlier. 6 These findings provide practical insights for improving the fermentation processes, particularly through the application of amylase and invertase enzymes from mango fruit at the verge stage to prolong the active fermentation phase in both dry and rainy seasons.
Specific Gravity
The SG serves as a vital parameter for monitoring sugar concentration and fermentation progress throughout the process. Changes in SG reflect the extent of sugar conversion into bioethanol and reveal inconsistencies in microbial performance under varying environmental conditions. During the dry season, SG fluctuated between 1.04 and 1.0, indicating variable sugar content due to incomplete fermentation and microbial stress as reported earlier. 19 Fluctuation in SG suggests irregular sugar consumption. In contrast, the rainy season shows fluctuation in SG between 1.03 and 1.0 throughout fermentation, reflecting efficient sugar conversion by the microbes. Generally, the SG value of 1.0 for both seasons was reached on day 5 (120 h) and remained constant thereafter. Stable SG implies minimal ongoing sugar-to-bioethanol conversion due to early substrate exhaustion or limited microbial activity. 20 This differs from typical fermentation, where SG progressively declines as sugars convert to bioethanol. 19 The stable SG during the rainy season aligns with the previously reported findings. 21
Percentage Alcohol by Volume
The %ABV is a key indicator of fermentation performance, reflecting the efficiency of sugar conversion into bioethanol over time. Seasonal environmental conditions, particularly temperature and humidity, play a significant role in influencing the %ABV trends by affecting microbial activity and fermentation stability. During the dry season, fermentation began with 5.15% ABV at hour 0, followed by a sharp decline to 0.20% after 12 h. A steep drop suggests rapid initial sugar utilization due to high microbial activity at the start of fermentation. Besides, a sudden decrease indicates early microbial stress or an imbalance in fermentation conditions. 22 After the initial drop, %ABV gradually increased, reaching 0.53% between hours 120 and 144, suggesting partial recovery of microbial activity. The levels then stabilized between 0.34% and 0.49% up to hour 264, followed by a slight rise to 0.99% at hour 324. Figure 4 presents the %ABV of bamboo juice with mango fruit during the rainy and dry seasons.
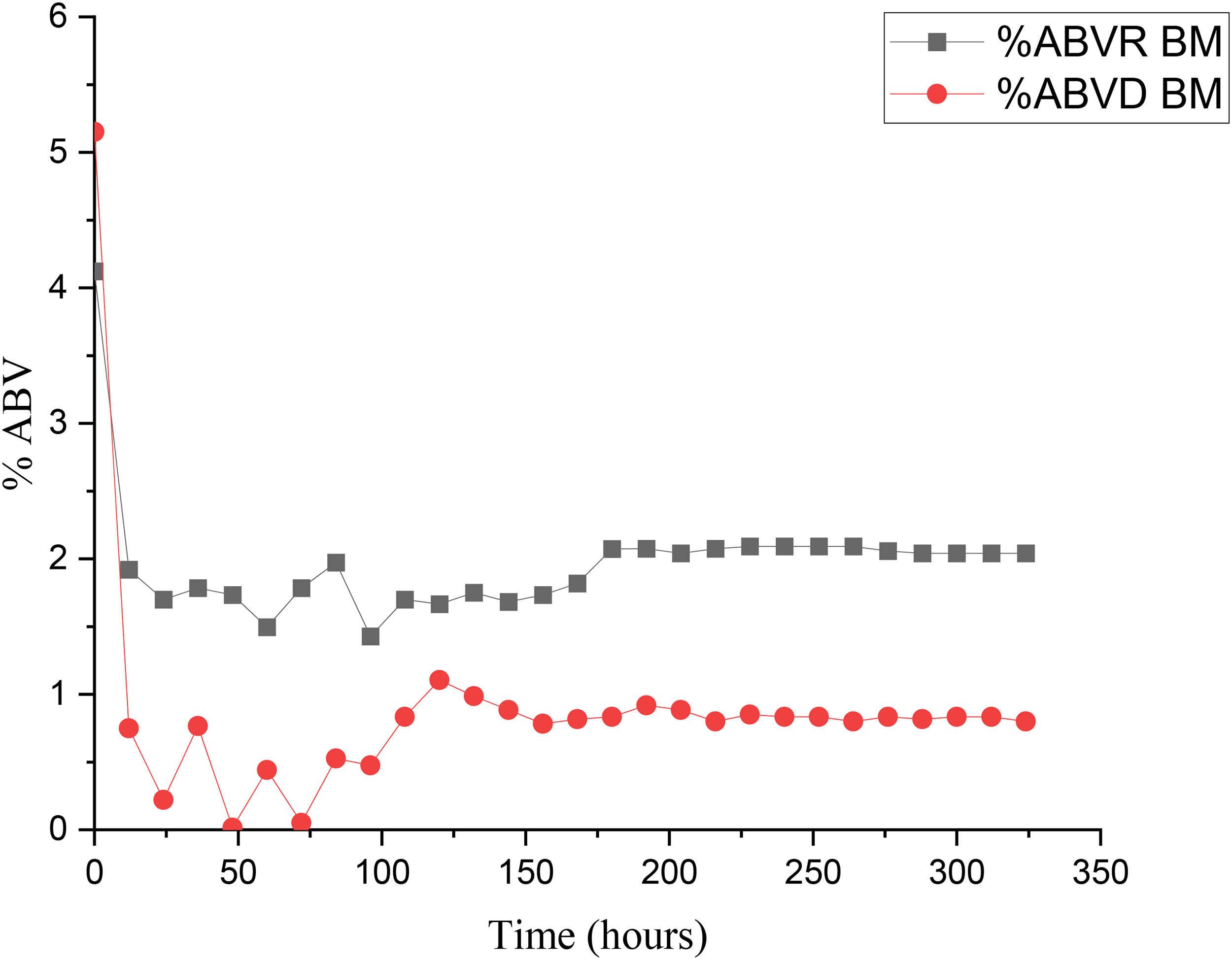
Percentage alcohol by volume of bamboo juice broths with mango fruit during the dry and the rainy season.
Besides, fermentation during the rainy season began at a %ABV of 4.14% and declined gradually to 2.04% at hour 324. Steady trend reflects more efficient and sustained fermentation under relatively stable environmental conditions. Throughout the fermentation period, bioethanol levels remained consistently above 1.8%, indicating continuous microbial activity and effective sugar conversion. A p-value of 0.0053 (p < 0.05) was obtained, indicating a statistically significant difference between the dry and rainy seasons. This confirms that the observed increase in %ABV of BM is not due to random variation but follows a consistent and measurable trend, thereby validating the reliability of the fermentation pattern. The absence of sharp fluctuations suggests that the fermentation process was less affected by stress factors, including temperature variability or nutrient imbalances, which are more common in the dry season. These findings are in agreement with a previous report. 23 The contrast in bioethanol retention between the two seasons supports the claim that rapid sugar consumption in the dry season, followed by a decline in microbial activity due to nutrient depletion and environmental stress, limits overall fermentation efficiency. The observed patterns further reinforce the role of seasonal variation in shaping fermentation performance and final bioethanol quality.
pH Trend
The pH is an essential parameter in fermentation as it directly influences enzyme activity, microbial growth, and overall metabolic environment. Monitoring pH variations throughout the fermentation process helps reveal the stability and efficiency of microbial activity under different seasonal conditions. During the dry season, pH increased from 3.84 to 4.33 by hour 324, revealing greater fluctuations compared to the rainy season. The variations, especially between hours 12 and 48, suggest the interruptions in microbial activity caused by environmental stress. Figure 5 shows the pH trend of bamboo juice broths with mango fruit during the rainy and dry seasons.

pH variation of bamboo juice with mango fruit during dry and rainy seasons.
In contrast, the rainy season's pH declined steadily from 3.79 to 3.7, indicating stable microbial activity and consistent organic acid production. These findings are consistent with those reported earlier. 24 Generally, the maximum pH value was reached on day 6 (144 h) and remained constant
Temperature
Temperature is a critical environmental factor that influences the rate of microbial metabolism, enzyme activity, and overall fermentation efficiency. Tracking temperature variations during fermentation helps assess the stability of the process and the adaptability of microbial communities under seasonal conditions. During the dry season, temperatures fluctuated between 25.03 and 26.6 °C, averaging at about 25.3 °C. The moderate fluctuations supported adequate fermentation as microbes adapt metabolically to variable conditions, enhancing efficiency and bioethanol yield. These were significantly warmer than the rainy season's stable range from 21.2 to 22.8 °C. Prolonged exposure above the optimal 20–25 °C range can reduce microbial activity viability, cause early microbial deactivation, increase bioethanol loss, and raise contamination risk. 25 Besides, the rainy season's stable temperature at around 22.4 °C created ideal conditions for microbial activity, promoting steady and efficient fermentation. Likewise, it was observed that stable temperature conditions enhance microbial metabolic activity, leading to increased fermentation efficiency and improved bioethanol quality. 26 Figure 6 illustrates the temperature variation trends observed during fermentation across both the dry and rainy seasons.

Trends in temperature variations during fermentation.
Distillation
Results indicate that bioethanol production from bamboo juice supplemented with mango fruit at the verge stage is significantly more efficient during the dry season compared to the rainy season. Enhanced efficiency aligns with higher TSS recorded in the dry season, where the BM sample had 10.01 °Brix, resulting in a higher bioethanol concentration of 39% v/v in the first aliquot of 100 mL. In contrast, the rainy season sample exhibited a lower TSS of 5.3 °Brix, producing 35% v/v bioethanol of similar volume. Thus, from the dry season, the BM sample yielded 39%, 33%, and 25% v/v bioethanol across the first, second, and third 100 mL aliquots, respectively, while the rainy season yielded 35%, 30%, and 22% v/v, respectively. Redistillation of collected distillates further concentrated bioethanol to 87% v/v, meeting the standard for acceptable biofuel and industrial applications. Findings indicate that the dry season conditions, characterized by higher sugar content and more favorable microbial activity, support improved fermentation efficiency and greater bioethanol output from bamboo juice.
Gas Chromatography-Flame Ionization Detector (GC-FID) Analysis
Samples were submitted to the Tanzania Bureau of Standards (TBS) for GC-FID analysis. The results confirmed that ethanol was present at high concentrations in all bamboo juice distillate samples relative to other trace impurities. Notably, the ethanol concentrations determined by GC-FID were consistent with those obtained using the alcoholmeter. Figures 7 and 8 show the GC-FID chromatograms for the two representative samples.

GC-FID Chromatogram of bioethanol (35%) from Bamboo juice (E2) sample.

GC-FID Chromatogram of bioethanol (87%) from Bamboo juice (E1) sample.
Limitations of the Study
The study's limited geographic coverage and restriction to two seasons may constrain the generalizability of the findings to other regions, climates, and bamboo species. Fermentation was conducted under small-scale laboratory conditions, which may not fully reflect industrial-scale performance or operational variability. Moreover, the use of spontaneous fermentation could have introduced variability in microbial populations, potentially affecting fermentation efficiency and bioethanol yield. The distillation process was limited to conventional distillation and redistillation, without advanced dehydration steps, which may have restricted bioethanol purity compared with industrial fuel-grade standards. Finally, the study did not incorporate an economic or environmental analysis, limiting conclusions regarding commercial feasibility and sustainability.
Conclusions and Recommendations
The study demonstrates that supplementing bamboo juice with mango fruit at the verge-stage produces significantly higher bioethanol concentrations from the dry season compared to the rainy season. The increased yield is primarily attributed to a higher TSS of 10.01 °Brix recorded in the dry season versus 5.3 °Brix from the rainy season. The maximum concentration of 39% v/v bioethanol was obtained from dry as compared to 35% v/v in the rainy season from the first 100 mL of distillate. Redistillation of the concentrated bioethanol achieved 87% v/v, meeting the standard required for biofuel and industrial applications. Findings indicate that bamboo juice harvested during the dry season, when supplemented with mango fruit at the verge stage, has greater potential for efficient bioethanol production. It is therefore recommended that large-scale bioethanol production prioritize bamboo harvesting during the dry season with mango supplementation. Further studies should explore pilot-scale fermentation and establish the consistency and efficiency under industrial processing conditions.
Footnotes
Acknowledgements
Authors are grateful to Mkwawa University College of Education for materials support.
Ethical Approval
Ethical approval was not required for this study as it did not involve human participants, animals, or sensitive personal data.
Author Contribution
JE participated in the conception, design, drafting and revising the paper critically for intellectual content, LM participated in the conception, design, drafting and revising the paper critically for intellectual content and OM participated in data collection, data analysis and writing the draft of the manuscript. All authors approved the final manuscript for publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
This study did not involve the generation or use of any datasets, and therefore, no data is available.

