Abstract
1-Hydroxymethyl goshuyuamide II (
Keywords
Tetradium ruticarpum (A. Jussieu) T. G. Hartley, known as “Wuzhuyu” in China, grows mainly in mainland China. The dried and nearly ripe fruits of T. ruticarpum are commonly used as a traditional Chinese medicine for the treatment of headache, epigastric distension, hyperbaropathy, dysentery, eczema, and rhinitis (Flora of China, 2004). Phytochemical studies have revealed that the main active constituents of T. ruticarpum are alkaloids, limonoids, coumarins, and flavonoids. 1 Evodiamine (EV), an indole alkaloid, and EV derivatives, have attracted much scientific attention because of their broad range of biological activities and unique structural characteristics. 2 Thus, the isolation of more EV derivatives from medicinal plants is a worthwhile objective.
In the course of screening for cytotoxic and anti-inflammatory EV derivatives from the fruits of T. ruticarpum, a new derivative, named 1-hydroxymethyl goshuyuamide II (

Chemical structures of
Results and Discussion
1-Hydroxymethyl goshuyuamide II (
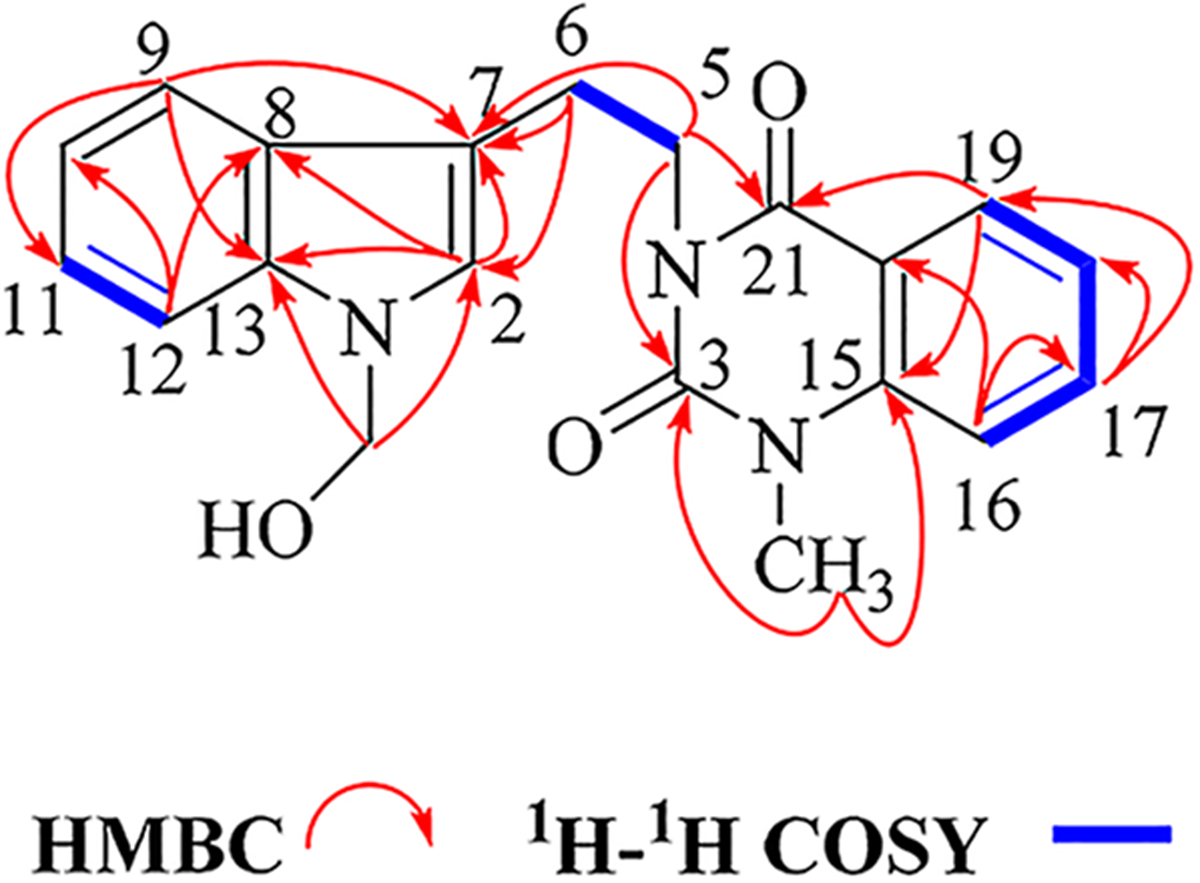
Key 2D-NMR correlations of compound
1H NMR (600 MHz) and 13C NMR (125 MHz) Data for

It is worth mentioning that hydroxymethyl goshuyuamide II (

Plausible biogenetic pathway of
The nine known compounds were identified as goshuyuamide II (
Compounds

Cell viability of compounds 1-10 on six human cancer cell lines at 40 μM.
The above results suggest that the closed ring between C2 and C3 maybe a functional group for cytotoxic activity. The cytotoxic activities of
Considering the traditional use of T. ruticarpum, all isolates were tested for their inhibitory activity against NO production in LPS-stimulated RAW264.7 cells. Compounds
Inhibitory Effects of Compounds From T. ruticarpum on LPS-Activated NO Production in RAW264.7 Cells.

Positive control substance.
Analysis of the anti-inflammatory activity and the structures of the tested compounds
Experimental
General Experimental Procedures
Melting points, X-4 micro melting point apparatus; UV, Shimadzu UV-2401A spectrophotometer; IR, Bruker Tensor 27 spectrophotometer with KBr pellets; and optical rotations, JASCO-20C digital polarimeter. ESIMS, API QSTAR time-of-flight spectrometer. HREIMS, Waters AutoSpec Premier P776 spectrometer. 1D NMR and 2D NMR, Bruker AM-400, DRX-500 or AVANCE III-600 spectrometers with TMS as an internal standard. MPLC, Lisui EZ Purify III System including pump manager P03, detector modules P02, fraction collector P01, and columns packed with MCI gel. Semipreparative HPLC, Agilent 1260 apparatus equipped with an UV detector and a Zorbax SB-C-18 column. Silica gel and Sephadex LH-20 were used for column chromatography (CC). Fractions were monitored by TLC and compounds were visualized by heating the silica gel plates after spraying with 10% H2SO4 in EtOH.
Plant Material
The fruits of T. ruticarpum were purchased in December 2014 from the Herb Material Market of Juhuacun, Kunming, Yunnan Province, P. R. China, and identified by one of the authors (Jiang-Bo He). A voucher specimen (20141202) was deposited at the Phytochemistry Laboratory (Medicine in Kunming University).
Extraction and Isolation
The fruits of T. ruticarpum (10.0 kg) were extracted with 80% methanol (3 × 50 L, 24 h, each). The EtOH extracts were evaporated to dryness under reduced pressure, suspended in distilled H2O, and partitioned with EtOAc consecutively. The EtOAc fraction (500 g) was subjected to reversed-phase MPLC (MCI) (MeOH/H2O, 50%→100%) to give fractions A–F. Fr.D (52 g) was chromatographed on a silica gel column with a gradient elution of light petroleum–acetone (10:1, 8:2, 7:3, 6:4, 5:5, and 0:1) to give fractions D-I–D-IV. Fraction D-I was purified by recrystallization from light petroleum–acetone at 25°C to obtain
1-Hydroxymethyl goshuyuamide II (
Cytotoxicity Assay
The cytotoxicity of compounds
Anti-Inflammatory Assay
Murine monocytic RAW264.7 macrophages were dispensed into 96-well plates (2 × 105 cells / well) containing RPMI-1640 medium (Hyclone) with 10% FBS under a humidified atmosphere of 5% CO2 at 37°C. After 24 h preincubation, the cells were treated with serial dilutions of the compounds, with the maximum concentration of 25 μM in the presence of 1 μg/mL LPS for 18 h. Each compound was dissolved in DMSO and further diluted in medium to produce different concentrations. NO production in each well was assessed by adding 100 μL of Griess reagent (reagent A and reagent B, respectively, Sigma) to 100 μL of each supernatant from either LPS (Sigma)-treated or LPS- and compound-treated cells in triplicate. After 5 min incubation, the absorbance was measured at 570 nm with a 2104 Envision Multilabel Plate Reader (Perkin-Elmer Life Sciences, Inc., Boston, MA, USA). MG-132 was used as a positive control. 13
The cytotoxicity of the compounds was evaluated using an MTS assay. 14 Briefly, RAW264.7 cells, 2 × 105 cells/well, were seeded in 96- well plates. After 24 h incubation, the cells were treated either with or without test compounds at given concentrations for 18 h. Then, MTS was added to each well and the plates were kept for 4 h. Test compounds were dissolved in DMSO, and the absorbance was measured at 490 nm. The cytotoxicity was calculated by the cell viability of cells without test compounds as 100%.
Conclusions
From the fruits of T. ruticarpum, one new compound (
The cytotoxic activity of all isolates against HL-60, Hela, HepG-2, A-549, AGS, and MDA-MA-468 cell lines was evaluated. Compounds
Compounds
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211040342 - Supplemental material for A New Cytotoxic Evodiamine Derivative From Tetradium ruticarpum (A. Jussieu) T. G. Hartley
Supplemental material, sj-docx-1-npx-10.1177_1934578X211040342 for A New Cytotoxic Evodiamine Derivative From Tetradium ruticarpum (A. Jussieu) T. G. Hartley by Jing Qin, Da-Wei Kang, Xue-mei Zhang, Li-ling Mo, Ying-dong Zhang and Jiang-bo He in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by the National Natural Science Foundation of China (Nos. 82060668), the Science and Technology of Yunnan Province Fund project (No. 202001BA070001-034) (No. U09326024), the Yunnan Provincial Fund project (2021J0712), and the “Spring City Project"(C201914001).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
