Abstract
Chemical examination of Sinularia querciformis afforded one new cembranoid, querciformolide F (
Introduction
Octocorals belonging to the genus Sinularia (phylum Cnidaria, class Anthozoa, order Alcyonacea, family Alcyoniidae) have been found to be a rich source of cembranoid diterpenes1-7 and steroids8-9. Compounds of these two types from Sinularia are reported to exhibit anti-inflammatory,6-7 and cytotoxic3-6 activities. In order to discover marine natural products with new chemical structures and bioactive activities, we conducted research on the soft coral Sinularia querciformis collected from the waters of Southern Taiwan, leading to the isolation of one new cembranoid, querciformolide F (

Structures of compounds
Results and Discussion
Querciformolide F (1) was isolated as a colorless oil that showed a sodiated adduct ion peak in its HRESIMS at m/z 389.19354 (M + Na)+, accounted for the molecular formula, C20H30O6 (Calcd for C20H30O6 + Na, 389.19346), indicating six degrees of unsaturation in the compound. The IR spectrum revealed absorptions for hydroxyl (νmax 3391 cm−1) and ε-lactone (νmax 1710cm−1) groups. The 13C-NMR spectrum of 1 (Table 1), showed signals of 20 carbons, which were further identified by the assistance of the distortionless enhancement by polarization transfer (DEPT) spectrum as three methyls, six sp3 methylenes, three sp3 methines (including two oxymethines), one sp2 methylene, two sp2 methines, three sp3 quaternary carbons and two sp2 quaternary carbons (including one ester carbonyl). The NMR signals observed at δC 169.4 (C), 144.3 (C), 124.7 (CH2), 88.7 (C), 35.7 (CH), 33.4 (CH2), 32.2 (CH2) and δH 6.31 (s), 5.47 (s) showed the presence of an α-methylene-ε-lactone ring by comparing the very similar NMR data of the cembranoids with the same seven-membered lactone ring. Signals resonating at δC 61.1 (C), 61.3 (CH) and δH 2.89 (1H, dd, J = 10.4, 3.2 Hz) revealed the presence of a trisubstituted epoxide. The NMR signals at δC 83.6 (C) and δH 7.34 (1H, brs) showed the presence of a hydroperoxy group at a methine carbon. One 1,2-disubstituted double bond was also identified from NMR signals appearing at δC 125.9 (CH), 134.4 (CH), and δH 5.44 (1H, d, J = 16.0 Hz) and 5.68 (1H, ddd, J = 16.0, 10.0, 4.8 Hz).
1H and 13C NMR Spectroscopic Data for Querciformolide F.
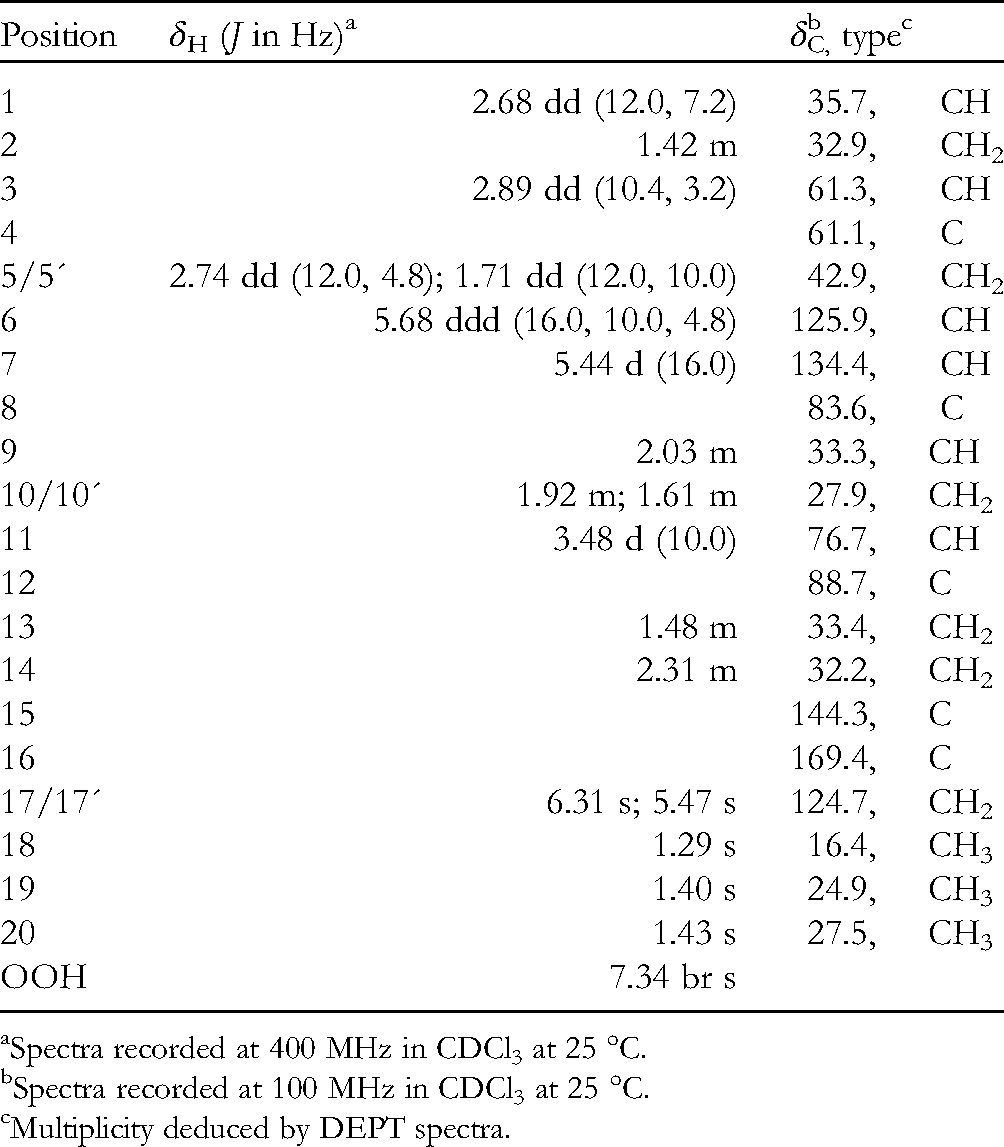
Spectra recorded at 400 MHz in CDCl3 at 25 °C.
Spectra recorded at 100 MHz in CDCl3 at 25 °C.
Multiplicity deduced by DEPT spectra.
From the 1H–1H COSY spectrum of 1, the separate spin systems of H2 to 13/H2 to 14/H-1/H2 to 2/H-3, H2 to 5/H-6/H-7 and H2 to 9/H2 to 10/H-11 enabled the identification of three different structural units, which were assembled with the assistance of an HMBC experiment. These data, together with the key HMBC correlations of H3 to 18 to C-3, C-4 and C-5; H3 to 19 to C-7, C-8 and C-9; H3 to 20 to C-11, C-12 and C-13 and H2 to 17 to C-1, C-15 and C-16 permitted the establishment of the carbon skeleton. The relative configurations of the six chiral centers at C-1, C-3, C-4, C-8, C-11, and C-12 in 1 were elucidated by detailed analysis of the nuclear Overhauser effect (NOE) correlations (Figure 2). H-1 (δH 2.68, dd, J = 12.0, 7.2 Hz) showed an NOE interaction with H-11 (δH 3.48, d, J = 10.0 Hz), and H3 to 18 (δH 1.29, s); therefore, assuming the α-orientation of H-1, H-11 and H3 to 18 this should also be positioned on the α-face. One of the methylene protons at C-10 (δH 1.92, m) exhibited NOE correlation with H-11 and was characterized as H-10α, while the other (δH 1.61, m) was assigned as H-10β. NOE correlations observed between H3 to 18 and H-6 (δH 5.68, ddd, J = 16.0, 10.0, 4.8 Hz), H-6 and H-5α (δH 2.74, dd, J = 12.0, 4.8 Hz), reflected the α-orientation of H-1, H-5α, H-6, H-10α, H-11, and H3 to 18. The J values for both H-6 and H-7 (16.0 Hz) further confirmed the E-configuration of the 6,7-double bond. Also, the NOE correlations observed for H-7 (δH 5.44, d, J = 16.0 Hz) with H3 to 19 (δH 1.40, s), and H3 to 20 (δH 1.43, s), H3 to 19, and H-10β, reflected the β-orientation of H-7, H-10β, H3 to 19, and H3 to 20. From the above observations and further analysis of other NOE interactions, the 1R*, 3S*, 4S*, 8R*, 11R* and 12R* relative configurations of 1 were established (Supplemental Materials, Figures S1–S10).

Key COSY (Fx1), HMBC (Fx2), and protons with NOESY (Fx3) correlations of
The in vitro anti-inflammatory activities of compounds
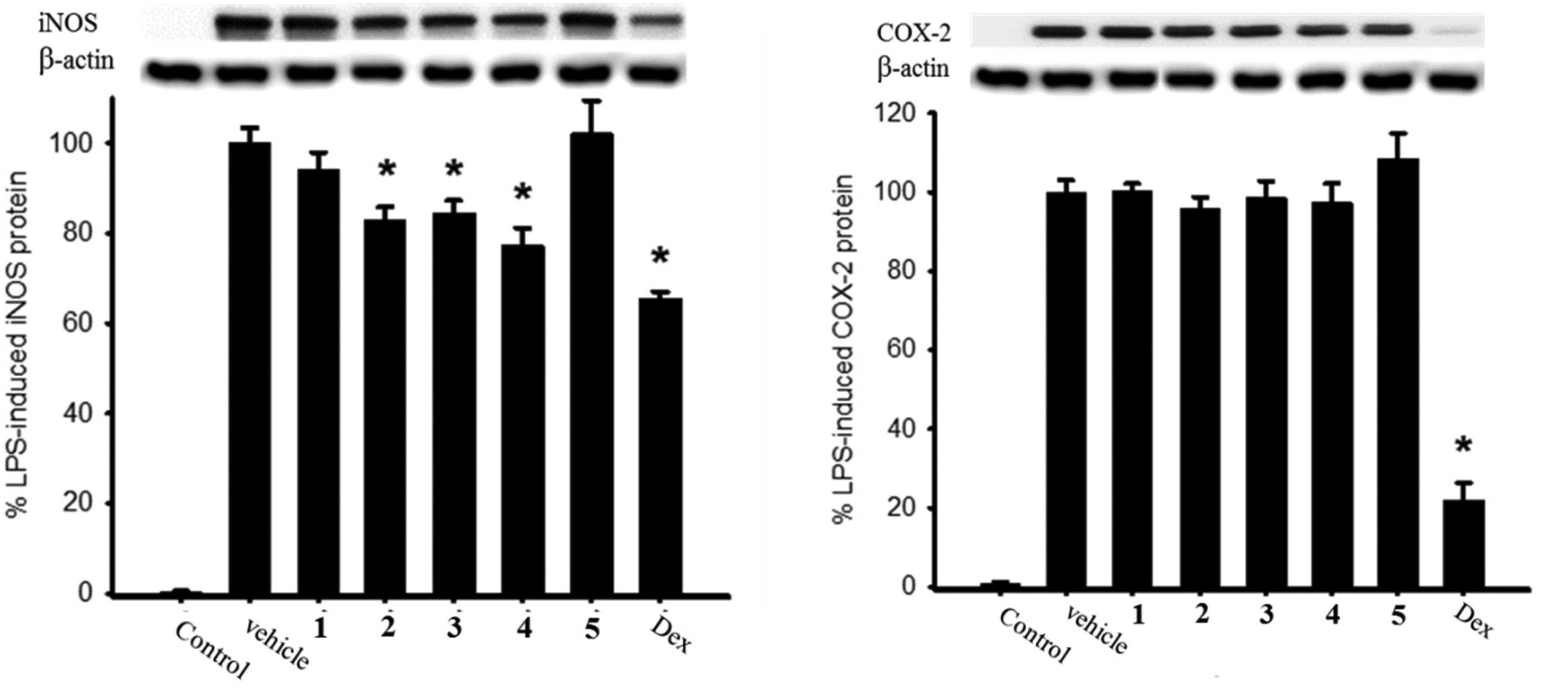
Effect of compounds
Effects of Compounds

Dexamethasone (DEX, 10 μM) was used as a positive control.
Materials and Methods
General Experimental Procedures
A digital polarimeter (model P-1010; JASCO Corp., Tokyo, Japan) was used to determine optical rotations of the samples. IR spectra were collected using a Nicolet iS5 FT-IR spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). NMR spectra were taken on a Varian NMR Mercury Plus spectrometer operating at 400 MHz for 1H and 100 MHz for 13C in CDCl3 using the residual CHCl3 signal (δH 7.26 ppm) and CDCl3 (δC 77.1 ppm) as the internal standard for 1H and 13C NMR, respectively; coupling constants (J) are given in Hz. For ESIMS and HRESIMS, the results were obtained using a SolariX FTMS mass spectrometer (7 Tesla; Bruker, Bremen, Germany). Gravity column chromatography was performed on silica gel (range, 230 to 400 mesh, Merck). TLC was carried out on precoated Kieselgel 60 F254 (0.2 mm, Merck) and compounds were visualized by spraying with 10% H2SO4 solution followed by heating. High-performance liquid chromatography (HPLC) was performed using a system comprised of a Hitachi L-7100 pump and a Rheodyne 7725 injection port. A semi-preparative normal phase column (Hibar 250 × 10 mm, Supelco, silica gel 60, 5 μm) was used.
Animal Material
Specimens of S. querciformis were manually collected by an underwater diver at a depth of 10 to 15 m from the sea around southern Taiwan on third June 2016. A representative sample of the soft coral (voucher no.: NMMBA-TW-SC-2015-0603) is stored in the National Museum of Marine Biology and Aquarium, Taiwan.
Extraction and Isolation
S. querciformis (wet/dry weight = 1870/570 g) was sliced and extracted with a mixture of methanol: dichloromethane (1:1). The extract was partitioned between ethyl acetate (EtOAc) and H2O. The EtOAc layer (7.60 g) was applied to a silica gel column and eluted with a gradient of n-hexane : EtOAc : acetone (from 100% n-hexane to 100% acetone) to yield 13 fractions, A–M. Fraction I was chromatographed using Si gel CC using n-hexane : EtOAc (1:1) to obtain fractions I1-I19. Fraction I11 was separated by NP-HPLC using a mixture of n-hexane : acetone (7:3) to yield querciformolide F (
Querciformolide F (
Anti-Inflammatory Test
A macrophage (RAW264.7) cell line was purchased from ATCC. We measured the in vitro anti-inflammatory activities of
Conclusion
One new cembranoid, querciformolide F (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211059299 - Supplemental material for Natural Cembrane Diterpenoids From the Soft Coral Sinularia querciformis
Supplemental material, sj-docx-1-npx-10.1177_1934578X211059299 for Natural Cembrane Diterpenoids From the Soft Coral Sinularia querciformis by Yi-Ying Wu, Chia-Ching Hsieh, Chia-Ying Li and Wen-Huei Chang, Jih-Jung Chen, Kuei-Hung Lai, Zhi-Hong Wen, Hsu-Ming Chung in Natural Product Communications
Footnotes
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology, Taiwan (grant number MOST 110-2320-B-153 -001).
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
