Abstract
The current investigation was intended to evaluate the antimyocardial ischemic effects of sesamol on lactate dehydrogenase (LDH) isoenzymes, DNA damage, and mitochondrial and lysosomal enzyme activities in isoproterenol (ISO)-induced myocardial infarction (MI) in male albino Wistar strain rats. Rats that received ISO (85 mg/kg body weight (B.W) subcutaneously) for the first 2 consecutive days showed significant reduction in the activities of tricarboxylic acid (TCA) cycle enzymes (isocitrate dehydrogenase, α-ketoglutarate dehydrogenase, malate dehydrogenase, and succinate dehydrogenase) and respiratory chain enzymes (cytochrome c oxidase and nicotinamide adenine dinucleotide hydrogen (NADH) dehydrogenase) in the heart mitochondria. The activities of the lysosomal enzymes (α-and β-glucosidases, α and β-galactosidases, β-glucuronidase and β-N-acetyl glucosaminidase and cathepsin-B and cathepsin-D) were increased significantly in the heart homogenate of ISO-induced MI rats. ISO injection also increased the % of tail DNA, tail length, and tail moment and decreased the % of head DNA. Pretreatment with sesamol (50 mg/kg B.W) every day for a period of 9 days prevented the above abnormalities induced by ISO. In conclusion, it can be inferred that administration of sesamol has a potent beneficial role against ISO-induced damage to the mitochondria, lysosomes, and DNA, thereby preventing MI.
Introduction
Cardiovascular diseases (CVDs) and their risk factors are important contributors to global morbidity and mortality. Among CVDs, myocardial infarction (MI) is the foremost cause of death and occurs as a result of prolonged myocardial ischemia that leads to irreversible injury and necrosis of myocardial tissue. Rat models of MI are widely used to design the diagnosis, prevention, and therapy of human MI. MI induced by isoproterenol (ISO) in rats is a widely accepted model to study the cardioprotective effects of various drugs. ISO, a synthetic β-adrenoceptor agonist, induces MI in animals as a result of its ability to disturb the physiological balance between the levels of free radicals and antioxidants. 1 Increased formation of free radicals causes an alteration in the membrane permeability leading to a loss of integrity and function of the myocardial membrane. Therapeutic intervention by amplifying endogenous antioxidants and repression of free radical production may reduce the infarction and inhibit myocardial dysfunction.
Lactate dehydrogenase (LDH) is a cytosolic enzyme, present in nearly every tissue, particularly in skeletal muscle, red blood cells (RBCs), heart, liver, lungs, brain, and kidneys. Increased serum LDH level occurs in connection with numerous diseases, and an abnormal elevation in the level of serum LDH 1 and LDH 2 is observed in MI. Therefore, detection of increased concentration of LDH 1 and LDH 2 in the blood has become a definitive prognostic and diagnostic measure for CVDs. In hypoxia, increased LDH activity enhances the conversion of pyruvate to lactate leading to the disturbance in the formation of adenosine triphosphate (ATP). 2
DNA damage is an essential biological marker for evaluating oxidative stress, and reactive oxigen species (ROS ) are important mediators of DNA damage. Damage occurs both in the mitochondrial DNA (mtDNA) and nuclear DNA in atherosclerosis. mtDNA is largely susceptible to ROS than nuclear DNA, mainly due to the absence of histone-like protein cores on the membrane and its poor DNA repairing activity. 3
In normal physiological circumstances, oxygen is essential for mitochondrial oxidative phosphorylation and for the production of ATP. The energy released from oxidative phosphorylation is an important source of energy for cardiac muscle contraction. Mitochondria consume about 90% of the oxygen in the cell and are the most vulnerable organelles to oxidative damage. The defect in oxygen supply disrupts electron transport chain in the mitochondria, resulting in the accumulation of toxic metabolites, ATP depletion, mitochondrial membrane depolarization, matrix enlargement, and cell death. 4 The potentially toxic metabolites formed in the cells during the mitochondrial electron transport chain are controlled by the cellular antioxidant defense system.
Damaged lysosomal membrane leads to the release of lysosomal contents to the cytosol. Lysosomal hydrolases are more detrimental to the cells due to its hydrolytic action on biological molecules including the nucleic acids, proteins, and polysaccharides. Lysosomal enzymes are acid hydrolases and are significant mediators of MI. The released hydrolases into the cytosol stimulate the expressions of inflammatory mediators such as prostaglandins and oxygen radicals. 5 Inflammation has been known to play a key role in the initiation and development of MI.
Antioxidants play a major role in the prevention of CVDs. Some polyphenolic compounds show very potent antioxidant activities. Phenolic compounds are widely present in various plants, and recently, they have drawn more attention mainly due to their antioxidant properties. Sesamol is an important phenolic antioxidant present in the sesame oil. Sesame oil is extracted from the seeds of the flowering plant Sesamum indicum L. that grows in the tropical areas. Sesame oil is used as a cooking oil as well as a spice and traditionally; this oil is used for facial massage to prevent aging and remove wrinkles. 6 Sesamol has been reported to possess antioxidant, 7 antiaging, 6 antimutagenic, 8 antihepatotoxic, 9 chemopreventive, 10 and anticarcinogenic activity. 11 The major purpose of this study was to assess the preventive effects of sesamol on membrane damage due to ISO-induced β-adrenergic stimulation.
Results
Agarose gel electrophoretic pattern of serum LDH-isoenzymes in normal and MI rats is shown in Figure 1. ISO-administered MI rats showed an increase in the intensity of LDH 1 and LDH 2 isoenzyme bands. ISO rats treated with sesamol showed a significant reduction in the band intensity of LDH 1 and LDH 2.

Electrophoretogram showing the effect of sesamol on lactate dehydrogenase (LDH)-isoenzymes in the serum of control and ISO-induced rats. Lanes 1: control; 2: sesamol (50 mg/kg body weight (B.W); 3: ISO (85 mg/kg B.W); and 4: ISO (85 mg/kg B.W) + sesamol (50 mg/kg B.W).
Figure 2 reveals the activities of heart mitochondrial tricarboxylic acid (TCA) cycle enzymes, isocitrate dehydrogenase (ICDH), α-ketoglutarate dehydrogenase (α-KGDH), malate dehydrogenase (MDH), succinate dehydrogenase (SDH), the respiratory chain enzymes, NADH dehydrogenase, and cytochrome c oxidase in normal and experimental rats. The activities of mitochondrial TCA cycle dehydrogenases and respiratory chain enzymes were decreased significantly in ISO-injected MI rats when compared with control group. The activities of all these enzyme activities were optimized to near normal after sesamol treatment.

Effect of sesamol on heart mitochondrial tricarboxylic acid cycle enzyme activities in control and experimental rats.
The activities of lysosomal hydrolases such as α-and β-glucosidases and α-and β-galactosidase, β-glucuronidase, β-N-acetyl glucosaminidase, cathepsin-B, and cathepsin-D in the serum of normal and ISO rats are shown in Figure 3. These lysosomal hydrolase activities in serum were elevated significantly in ISO-injected rats. Sesamol treatment significantly reduced the activity of these enzymes to near those of the control rats.
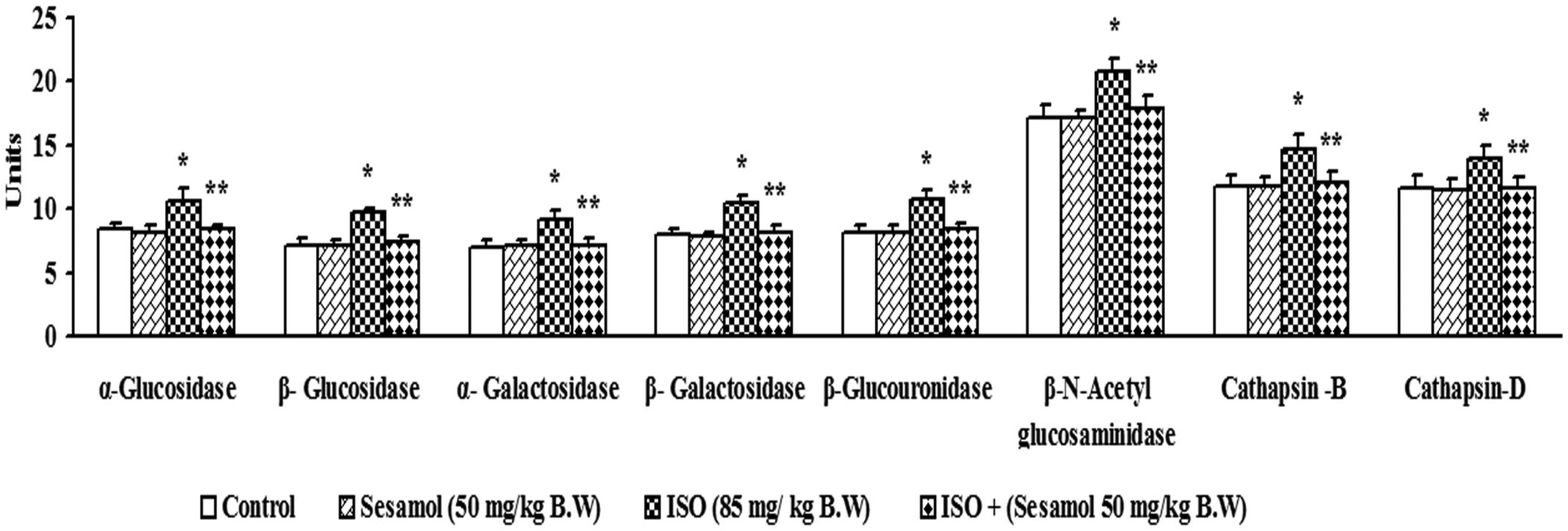
Effect of sesamol on the activities of serum α and β-glucosidases, α and β-galactosidases, β-glucuronidase, β-N-acetyl glucosaminidase, cathepsin B, and cathepsin D in control and ISO-induced MI rats.
Figure 4 represents the effect of sesamol on the activities of α-and β-glucosidases and α-and β-galactosidase, β-glucuronidase, β-N-acetyl glucosaminidase, cathepsin-B, and cathepsin-D in the heart homogenate of control and experimental rats. Compared with the control rats, ISO rats showed a significant increase in the activities of all the above lysosomal hydrolases and sesamol treatment normalized the activities of these enzymes in the heart.
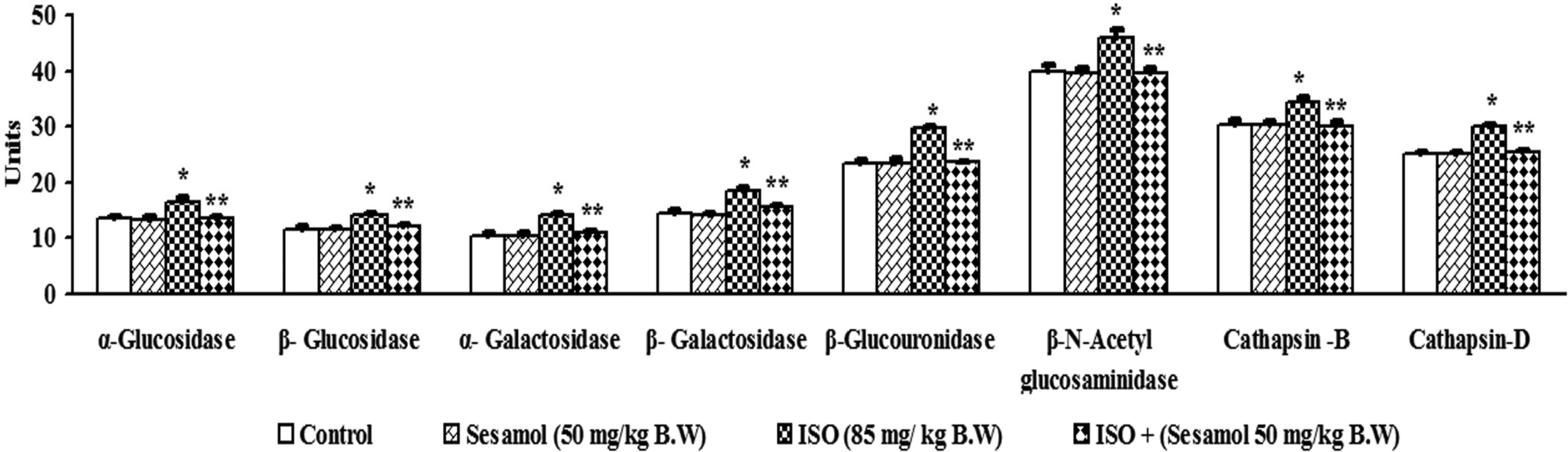
Effect of sesamol on the activities of α and β-glucosidases, α and β-galactosidases, β-glucuronidase, β-N-acetyl glucosaminidase, cathepsin B and cathepsin D in the heart of control and ISO-induced MI rats.
Figure 5 (A) depicts the quantification of DNA damage in the myocytes of experimental animals. The level of DNA damage by means of percentage of DNA present in the comet head (% head DNA), tail (% tail DNA), tail moment, and tail length was revealed in Figure 5 (B). ISO administered rats showed an increased DNA damage in the myocyte, and the administration of sesamol significantly reduced the DNA damage caused by ISO.

Seasmol on ISO-induced DNA damage was analyzed by comet assay. Fluorescence microscopic images were recorded . The % head DNA, % tail length, and tail moment were calculated by computer aided product selection (CAPS) software. (i) control; (ii) sesamol (50 mg/kg body weight (B.W)); (iii) ISO (85 mg/kg B.W); and (iv) ISO (85 mg/kg B.W) + sesamol (50 mg/kg B.W).

Diagram showing the possible underlying mechanisms for the ISO-induced MI and the role of sesamol.
Discussion
This study reveals the protective effect of sesamol on myocardial damage induced by ISO in rats. Subcutaneous (s.c.) injection of ISO in rats causes damage to the DNA, to the lysosomal and mitochondrial membranes, and to the DNA in the myocardium by releasing the free radicals. Nonspecific raise of total LDH in the blood will take place following tissue injury, and the quantification of LDH isoenzymes is vital for the diagnosis of different types of diseases and disorders and also to identify the site of tissue damage. LDH is a glycolytic enzyme and exists in five different isoforms (LDH1–LDH5). Variation in the LDH1 and 2 isoenzymes in serum can be considered as the best diagnostic measure for evaluating myocardial damage; size of the infarction is indicated by the rate of appearance and disappearance of LDH in the blood. The results of the current study showed enhanced band intensity of LDH1 and LDH2 isoenzymes in the ISO group of rats; these results agree with those of Upaganlawar et al. 12 Decreased band intensity of LDH1 and 2 was absorbed in sesamol-treated ISO rats when compared with ISO alone administered rats; this might be due to minimal damage to the myocardium and thus the minimal leakage of enzymes.
Cardiomyocytes are rich in mitochondria, and the mitochondria are the most important organelles that contribute to myocardial injury. In the cell, mitochondria are the major oxygen consumers and are severely affected during hypoxia. Hypoxia is correlated with an impairment of mitochondrial respiration, diminished oxygen uptake, and myocardial energy production. Of all the biomolecules that are released from the damaged tissue, enzymes are considered as the principal markers of tissue injury due to their catalytic activity and tissue specificity. Mitochondrial play an important role in the synthesis ATP by oxidative phosphorylation. The inability to produce ATP in the mitochondria leads to apoptosis of the endocardium. Therefore, we decided to analyze the activities of mitochondrial enzymes in the myocardium. The mechanisms underlying the changes in mitochondrial functions are studied by evaluating the activities of tricarboxylic acid (TCA) cycle dehydrogenases (ICDH, α-KGDH, SDH, and MDH) and respiratory chain enzymes (cytochrome c oxidase and NADH dehydrogenase). ISO has been reported to induce hypoxia in the myocardium due to the increased free radical formation, and during hypoxia, the TCA cycle enzyme activities are known to be low. 13 The decreased activities TCA cycle enzymes affect pyruvate oxidation, leading to the inadequate formation of ATP. Cytochrome-C-oxidase and NADH dehydrogenase are also involved in the formation of ATP (Figure 6). These enzymes require cardiolipin for their activity, but increased lipid peroxidation decreases the level of cardiolipin. The deficiency of cardiolipin due to the increased phospholipid degradation lowers the activity of respiratory enzymes in ISO administered rats. 14 In this study, a remarkable decrease in the activities of the mitochondrial TCA cycle and respiratory chain enzymes was observed after administration of ISO, which is in line with other findings. 15 Sesamol administration restored the activities of the TCA cycle and respiratory chain enzymes in the heart mitochondria due to the ability of sesamol to scavenge the free radicals and prevent the membrane phospholipid degradation. The enhanced activities of the TCA cycle and respiratory chain enzymes might be a reason for the normal cardiac mitochondrial functions in ISO injected rats after treatment with sesamol.
Lysosomal compartment is the most important site of intracellular protein degradation. Lysosomal membranes are reported to contain large amounts of glycoproteins and phospholipids, which play an important role in maintaining lysosomal structure and function (Chen et al 16 ). The phospholipid rich lysosomal membrane is a principal site for free radical attack resulting in membrane damage. Lysosomal compartments are acidic and have many hydrolytic enzymes that can degrade almost all cellular components, which play an important role in cardiac damage. Oxygen-free radicals generated by ISO are responsible for the lysosomal mediated cardiac damage through the release of lysosomal enzymes in addition to direct myocardial damaging effect (Ganesan and Anandan 17 ). The damaged lysosomal membrane elevated the levels of lytic enzymes in the cytosol. The discharge of lysosomal enzymes in turn mediates cell death either directly or by activating complement pathway. In the present study, the activities of lysosomal enzymes (of α-& β-glucosidases, α-&β-galactosidases,β-glucuronidase, β-N-acetyl glucosaminidase and cathepsin-B and D) were enhanced in the serum and heart tissue of MI rats. The increased activities of these hydrolases in the myocardium of disease control rats are due to the lysosomal membrane damage caused by ISO. Mohamadin et al 18 have also reported an increase in the activities of lysosomal enzymes in the serum and heart tissues of ISO group rats. Sesamol administration diminished the activities of lysosomal hydrolases in the serum and myocardium by its ability to prevent lipid peroxidation and thus inhibited membrane damage induced by ISO.
Oxidative stress is the main reason for DNA damage, in all aerobic organisms. Oxidative DNA damage is an important reason for mutations and cell death. Several investigators have confirmed the ability of antioxidants in the prevention of cellular DNA damages induced by various toxic compounds.19,20 In this study, increased levels of DNA damage in the myocytes of ISO induced rats were observed, which is in consensus with previous reports. 21 Sesamol treatment decreased the DNA damage caused by ISO, which might be due to its ability to scavenge the radicals and inhibit oxidative stress. It has been also reported that sesamol scavenge free radicals within a fraction of a second (nanosecond) and inhibits DNA damage in calf thymus induced by Fenton. 22
In the present study, the effect of sesamol on lysosomal and mitochondrial membrane was determined in ISO-induced MI rats. ISO-induced myocardial damage was evidenced by a significant decrease in the activities of the TCA cycle and respiratory chain enzymes in the mitochondrial fraction and a significant increase in the activities of the lysosomal enzymes in the serum and heart tissue of MI rats. From the results of this study, we have found that the 50 mg/kg B.W dose of sesamol is significantly more effective against membrane damage caused by ISO than other two doses. Hence, we have chosen 50 mg/kg body weight (B.W) dose of sesamol for further study. We have observed “seizures” at 200 mg/kg B.W of sesamol administration with 25% mortality.
Conclusion
The results of the present study suggest that the sesamol displays cardioprotective effect by preventing DNA damage and restoring the activities of the mitochondrial and lysosomal enzymes to near normal by scavenging the free radicals thereby stabilizing the membranes. These effects might be due to the antioxidant and membrane stabilizing effects of sesamol, the principle component of sesame seed oil, and thus it would be a very effective and promising medicine for the treatment of MI.
Materials and Methods
Animals
Male albino rats of Wistar strain with a body weight ranging from 180 to 200 g were obtained from the Central Animal House, Department of Experimental Medicine, Rajah Muthiah Medical College and Hospital, Annamalai University, and maintained in an air-conditioned room (25 ± 30 °C) with a 12 hour light/12 hour dark cycle. Feed and water were provided ad libitum. All experimental studies were conducted in the Department of Biochemistry, Faculty of Science, Annamalai University. The experimental study protocol was approved by the Animal Ethics Committee of Rajah Muthiah Medical College and Hospital (Reg No.160/ 1999/CPCSEA, Pro. No.579), Annamalainagar.
Chemicals
ISO hydrochloride and sesamol were purchased from Sigma-Aldrich Chemical Co., St. Louis, Missouri, USA. The purity of sesamol is ≥ 99% thin layer chromatography (TLC). All other chemicals and reagents used were of analytical grade from E. Merck, Mumbai, India.
Experimental Induction of Cardiotoxicity
MI was induced by s.c. injection of ISO hydrochloride (ISO, 85 mg/kg B.W, dissolved in physiological saline) in the right thigh of the rat for 2 consecutive days, at an interval of 24 hours.
Experimental Design
The rats were randomly divided in to four groups of eight rats each. Sesamol, dissolved in saline (0.9% NaCl) was administered intraperitoneally (i.p.) once a day in the morning for 7 days to the rats. Saline alone acted as a vehicle control.
Group I: Control rats (0.9% saline i.p. from third to ninth day). Group II: Control + sesamol (50 mg/kg B.W. i.p. from third to ninth day). Group III: ISO (85 mg/kg B.W. s.c. on first and second day). Group IV: ISO (85 mg/kg B.W. s.c. on first and second day + sesamol [50 mg/kg B.W., i.p. from third to ninth day]).
The total experimental duration was 9 days. On 10th day morning, the animals were anesthetized using ketamine (24 mg/kg B.W, intramuscular injection) and sacrificed between 8:00 am and 9:00 am by cervical dislocation. Blood samples were collected in a dry test tube, and serum samples were separated by centrifugation at 2000 rpm for 10 minutes. The heart tissue was excised immediately, washed with chilled isotonic saline, and used for the analysis of various parameters.
Myocyte Preparation
Cardiomyocytes were isolated by the method of Van der et al
23
. The heart tissues after excision from the rats were immediately placed into chilled dissociation buffer, which was prepared my mixing 5.4
Separation of Serum LDH Isoenzymes by Agarose Gel Electrophoresis
Agarose gel electrophoresis was used to separate the serum LDH isoenzyme
Isolation of Mitochondria
The heart mitochondria were isolated from the cell debris, nuclei, soluble components, microsomes, and RBCs by differential centrifugation.
25
0.25
Assay of Lysosomal Enzymes in the Serum and Heart Tissue
β-glucuronidase activity was assayed by the method of Kawai and Anno (1971) 32 in the serum, heart tissue homogenate. Β-N-acetyl glucosaminidase, β-galactosidase, and cathepsin-B activities in the serum and heart tissue homogenate were analyzed according to the method of Moore and Morris, 33 Conchie and Levvy, 34 and Barrett, 35 respectively. Sapolsky et al 36 method was used for the estimation of cathepsin-D activity in the serum and heart tissue. The total protein content in the tissues was determined by Lowry et al 37 method.
Statistical Analysis
The data was analyzed using one way analysis of variance on Statistical Package for Social Sciences, personal computer Version 10, and the group means were compared using Duncan's Multiple Range Test. All quantitative measurements were expressed as means ± SD for control and experimental rats. The results were considered statistically significant if P value was less than .05.
Ethical Approval
This study was approved by the Animal Ethics Committee of Rajah Muthiah Medical College and Hospital, Annamalainagar (Reg No.160/ 1999/CPCSEA, Pro. No.579).
Statement of Human and Animal Rights
All of the experimental procedures in this study involving animals were conducted in accordance with the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), New Delhi, India (Reg No.160/ 1999/CPCSEA, Pro. No.579) and approved by the Animal Ethics Committee of Rajah Muthiah Medical College and Hospital, Annamalainagar.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Footnotes
Author contributions
Study conception and design Dr. Kodukkur Viswanathan Pugalendi. Data collection, analysis and interpretation of results Dr. Lakshmanan Vennila. Manuscript preparation Dr. Thangaiyan Radhiga.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
