Abstract
Liverworts are characterized by a high content of bioactive compounds reported to show antimicrobial, anticancer, and antioxidant properties. In this study, the biological effects of the methanol extract of the liverwort Pellia endiviifolia and its constituents, bis-bibenzyls perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E, were investigated using human peripheral blood cells as a model system. The assessment of the investigated compounds comprised testing their genotoxicity, apoptotic potential
Keywords
Introduction
Bryophytes produce a great number of secondary metabolites, such as terpenoids, phenolics, sterols, tannins, flavonoids, and aromatic compounds that are reported to have pharmacological potential displaying antibacterial, antifungal, antioxidant, and cytotoxic properties.1-3 These nonvascular plants are classified into three divisions: Bryophyta (mosses), Marchantiophyta (liverworts), and Anthocerotophyta (hornworts). 4 Liverworts are characterized by a high content of lipophilic mono-, sesqui-, and diterpenoid compounds, with bibenzyls and bis-bibenzyls as typical macrocyclic phytochemicals harvested from bryophytes. 5 These compounds have demonstrated anticancer, antimicrobial, and antioxidant properties, making them suitable for further studies related to potential therapeutic applications. 6 More than 60 macrocyclic and acyclic bis-bibenzyls have been isolated so far. 5 Among them, the marchantin type of bis-bibenzyls was reported to possess very promising bioactivities. For example, marchantin A, isolated from Marchantia species, is one of the well-studied bis-bibenzyls, whose antibacterial and antifungal activities have been extensively demonstrated. 7 Furthermore, the bis-bibenzyls of riccardin and perrottetin types isolated from the liverwort Lunularia cruciata exhibited cytotoxic activity against the A549 lung cancer cell line. 8 Also, perrottetin F and metabolites obtained by its biotransformation by Aspergillus niger showed promising antimicrobial activity and antiproliferative effects on human lung carcinoma. 9 Perrottetin E isolated from Conocephalum japonicum and Radula perrottetii exhibited cytotoxic effects against human nasopharyngeal epidermoid carcinoma (KB) cells10,11 and induced inhibition of thrombin activation. 12 The same compound, isolated from Bazzania trilobata, showed an inhibitory effect on Candida albicans. 13
Pellia endiviifolia (Dicks.) Dumort. (Pelliaceae) is a terrestrial liverwort that inhabits damp places growing by springs and watercourses, on shaded rocks and moist floors, and is widespread in the Northern Hemisphere. 14 Recently, different types of diterpenoids, such as sacculatal, isosacculatal, and sacculatanolide, and macrocyclic bis-bibenzyls, such as perrottetin E, perrottetin E-11′-methyl ether, 14-hydroxyperrottetin E-11′-methyl ether, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E, have been isolated from P endiviifolia.15-17 Importantly, sacculatal cytotoxic activity was reported against Lu1, KB, KB-V, LNCaP, and ZR-75-1 cell lines. 18 In addition, the results of our previous study showed promising cytotoxic activities against several cancer cell lines that were attributed to the bis-bibenzyls of the perrottetin type (perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E) isolated from the methylene chloride/methanol extract of P endiviifolia. 17 The possible application of these compounds in medicine led us to evaluate their biological effects in normal cells. Therefore, in this study, the genotoxicity, apoptotic potential, and redox modulating activities of the methanol extract of P endiviifolia and its constituents, perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E, were investigated using human peripheral blood cells as a model system. The genotoxicity testing of the investigated compounds was performed in human lymphocyte cultures using the cytokinesis-block micronucleus (CBMN) assay, which is one of the standard genotoxicity assays recommended by ISO 10993-3. 19 The CBMN assay is a reliable and precise test for assessing chromosome instability, mitotic dysfunction, cell proliferation, and cell death. 20 It enables the detection of micronuclei (MN) in the cytoplasm of interphase cells that originate from acentric chromosome fragments or whole chromosomes that are unable to migrate to the poles during the anaphase of cell division. 21 In addition, the CBMN assay enables measurement of the relative frequencies of mononucleate (cells where division does not occur), binucleate (BN), and polynucleate cells (cells where the nucleus is divided) in the culture, providing an accurate method of quantifying the effect of an agent on cell proliferation and the cytotoxic or cytostatic activity of treatment. Early toxicity and cell cycle delay can be evaluated by determining the ratio of polynucleated cells versus mononucleated cells for both untreated and treated cells. Accordingly, the cytokinesis-block proliferation index (CBPI) is used to estimate the cytotoxic and cytostatic activity of a treatment by comparing values in the treated and control cultures. 21 The investigated compounds were also evaluated for their redox modulating effects, which comprised measuring catalase activity and malondialdehyde (MDA) level in human blood cells as a measure of oxidative stress induction.
Results and Discussion
The Cytokinesis-Block Micronucleus Assay
In the present study, human peripheral blood lymphocytes were treated with increasing concentrations of P endiviifolia extract and its constituents, perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E. CBMN analysis showed that all investigated compounds at medium and high concentrations induced a concentration-dependent increase in MN incidence and decrease in cell proliferation potential compared to the control (Table S1 in Supplemental material). As shown in Figure 1, treatment of lymphocyte cultures with the lowest concentration (5 µg/mL) of P endiviifolia extract induced a significant (P < .001) reduction in the incidence of MN and a decline in CBPI (P < .001) compared to the control. At medium (10 µg/mL) and high (50 µg/mL) concentrations, the P endiviifolia extract induced a significant enhancement of MN incidence and decrement of CBPI (P < .001). Similar effects were observed upon treatments with corresponding concentrations of P endiviifolia constituents. Namely, at medium (25 µM) and high (100 µM) concentrations, perrottetin E and 10′-hydroxyperrottetin E considerably increased the MN incidence and decreased CBPI in a concentration-dependent manner, being the most effective at the highest applied concentrations (P < .001). Accordingly, a negative correlation between these parameters was observed (r

Incidence of micronuclei (mean ± SD) and cytokinesis-block proliferation index (mean ± SD) in lymphocytes treated with (a) Pellia endiviifolia extract, (b) perrottetin E, (c) 10′-hydroxyperrottetin E, and (d) 10,10′-dihydroxyperrottetin E. Results are expressed as a percentage of control, set to 100%; * P < .001 in comparison with corresponding control, no label P > .05.

A representative micrograph showing mononucleated, binucleated, polynucleated cells and micronucleus in a binucleated cell following Giemsa staining.
Results of the study indicate that P endiviifolia extract and its constituents are genotoxic and display antiproliferative effects in human peripheral blood lymphocytes. The extent of their genotoxicity appears to be concentration-dependent. The obtained results are in accordance with a previously published report indicating a promising anticancer activity of the investigated compounds against several cancer cell lines, 17 namely, perrottetin E exhibited strong cytotoxic effects against HL-60 (half maximal inhibitory concentration (IC50) = 14.2 µM), chronic myeloid leukemia K-562 (IC50 = 37.2 µM), glioblastoma A-172 (IC50 = 8.8 µM), U-251 (IC50 = 15 µM), and human testicular embryonal carcinoma NT2/D1 cells (IC50 = 11.2 µM). 17 Similarly, 10′-hydroxyperrottetin E showed strong effects on the viability of acute myeloid leukemia U-937 (IC50 = 38.5 µM), A-172 (IC50 = 26.2 µM), and NT2/D1 cells (IC50 = 15.5 µM). The same study indicated that 10,10′-dihydroxyperrottetin E exhibited a moderate or weak cytotoxic effect against most of the investigated cancer cell lines, except for the NT2/D1 cells, where it displayed a strong cytotoxic effect, with an IC value of 6.8 µM. 17
Interestingly, in the current study, the medium concentration of 10,10′-dihydroxyperrottetin E did not promote significant micronuclei induction in normal cells. Since one of the major complications encountered during cancer treatment is therapy-associated normal tissue toxicity, 22 it is of great importance to discover a potential anticancer agent of high efficacy against cancer cells and low toxicity toward surrounding normal tissue. Further comprehensive studies of 10,10′-dihydroxyperrottetin E are needed to investigate the apparent selectivity of its cytotoxic activity.
Apoptosis of Leukocytes
In this study, leukocyte apoptosis induced by increasing concentrations of P. endiviifolia extract and its constituents perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E (Figure 3) was analyzed. The study results showed that the percentage of leukocyte apoptosis in control cultures was approximately 12%. Upon treatments of cell cultures with increasing concentrations of P endiviifolia extract, the percentages of apoptotic cells fluctuated between 18% and 80%. Upon treatments with perrottetin E, the percentages of apoptotic cells ranged from 16% to 41%. After treatment with 10′-hydroxyperrottetin E, the percentages of apoptotic cells fluctuated between 7% and 74%, while in the case of 10,10′-dihydroxyperrottetin E, the percentages of apoptotic cells ranged from 4% to 32%. The P endiviifolia extract and perrottetin E showed considerable proapoptotic potential, even at the lowest concentrations, 45% and 40%, respectively, while the same concentration of 10′-hydroxyperrottetin E and 10,10′-dihydroxyperrottetin E induced only 7% and 4% of apoptotic cells, respectively. Except for perrottetin E, which induced the maximal percentage of apoptotic cells at the highest employed concentration (41%), all other investigated compounds induced the maximal apoptosis at the medium (25 µM) concentration. The obtained results indicate that the investigated compounds have a significant proapoptotic potential, which is not concentration-dependent. According to the literature data, significant pro-apoptotic activity was reported for several bis-bibenzyls, such as marchantin M, isolated from Asterella angusta extract, which was found to be cytotoxic against prostate cancer PC3 cells and to induce upregulation of Bax expression.
23
Likewise, marchantin C was found to induce cell cycle arrest at the G(2)/M phase and trigger apoptosis in A172 and HeLa cells.
24
Riccardin C from Plagiochasma intermedium was reported to induce apoptosis in PC3 cells and modulate the expression levels of Bcl-2, Bax

A typical example of flow cytometric analysis of apoptosis in control cell culture and cell cultures treated with increasing concentrations of P endiviifolia extract. (AP, the percentage of apoptotic cells).
Catalase (CAT) Activity
Oxidative stress appears to be due to increased production of reactive oxygen species (ROS) and/or a decrease in the antioxidant defense system. As the first-line defense antioxidant enzyme, catalase plays an essential role in appraising cellular oxidative status. Therefore, we analyzed erythrocyte catalase activity in blood cultures treated with increasing concentrations of the investigated compounds to assess whether the P endiviifolia extract and its constituents affect the antioxidant defense system (Table S2 in Supplemental material). As shown in Figure 4, the P endiviifolia extract and its constituents, perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E, did not induce significant changes in catalase activity compared to the control.
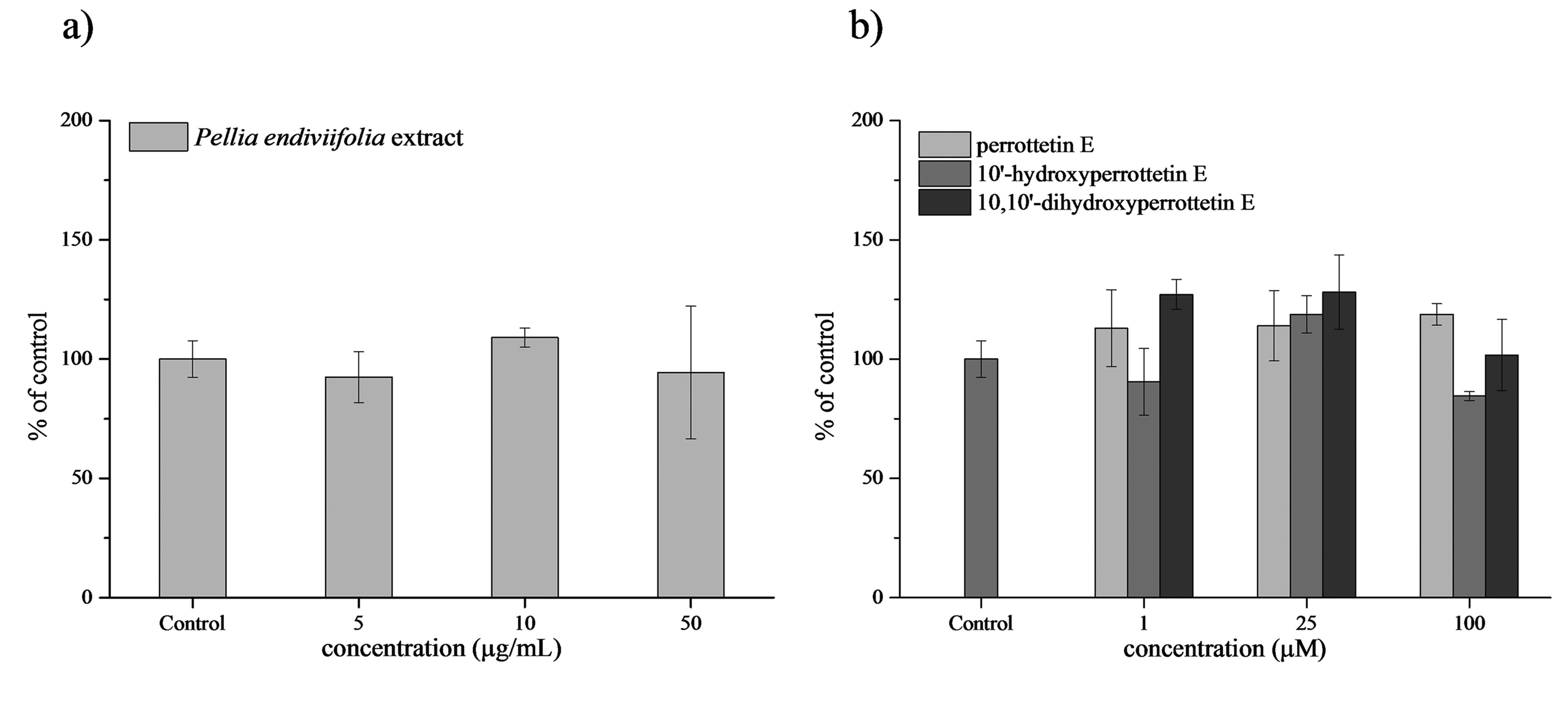
Catalase activity (mean ± SD) in erythrocytes treated with (a) P endiviifolia extract, (b) perrottetin E, 10′-hydroxyperrottetin E
According to literature data, the methanol extract of P endiviifolia showed promising antioxidant potential, as revealed by investigation of its DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity, and total phenolic and flavonoid estimation. 1 In the current study, P endiviifolia extract and its constituents did not affect erythrocyte catalase activity in the investigated concentration range.
Thiobarbituric Acid (TBA) Analysis
The potential of the investigated compounds to induce lipid peroxidation in the cell membrane was assessed by measuring the level of MDA in lymphocytes. Lipid peroxidation represents an indicator of ROS-mediated damage to cell membranes. Therefore, as the end product of lipid peroxidation, MDA is commonly used to evaluate oxidative stress conditions.
Similar to the results obtained for catalase activity, in cultures treated with increasing concentrations of P endiviifolia extract and its constituents, perrottetin E, 10′-hydroxyperrottetin E

Level of malondialdehyde (mean ± SD) in lymphocytes treated with (a) P endiviifolia extract, (b) perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E. Results are expressed as a percentage of control, set to 100%.
Conclusion
Biological effects of P. endiviifolia extract and its constituents, perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E, were examined using human peripheral blood cells as a model system. Results of the study demonstrate that the investigated compounds induced a concentration-dependent enhancement of MN incidence and suppression of cell proliferation. The apoptosis assessment showed their substantial proapoptotic effects, which were not concentration-dependent. The P. endiviifolia extract and its constituents did not induce oxidative stress in human peripheral blood cells, as shown by their insignificant effects on catalase activity and MDA production. The obtained results suggest that apoptosis induced by the investigated compounds was not oxidative stress-mediated.
Taken together, the results of the study indicate genotoxic, antiproliferative
Material and Methods
Chemicals
All chemicals were of analytical grade. For isolation of the pure bis-bibenzyls: methylene-chloride, methanol, n-hexane, and ethyl acetate (EtOAc) were purchased from J.T. Baker. Deionized water was used throughout.
For the cell culture: Panserin 701 was purchased from Pan-Biotech, lymphocyte separation medium and RPMI-1640 from Capricorn Scientific, cytochalasin B, propidium iodide (PI), ribonuclease A (RNase A), and dimethylsulfoxide (DMSO) from Sigma-Aldrich, TBA and Giemsa stain from Merck, phosphate buffer (PBS) from Bioatlas, and methanol and acetic acid from VWR.
Plant Material
P endiviifolia Extract and bis-Bibenzyls
Methanolic extract of P endiviifolia was freshly prepared from dried plant material. Upto 20 g of dried plant material was extracted three times with 100 mL of methanol, each time for 24 h. Extracts were combined, evaporated in a rotary vacuum evaporator (Laborota 4001), and 1.22 g residue was obtained (yield 6.1%). Since we used the same plant material in our previous work, we also used previously isolated bis-bibenzyls perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E for investigation of their biological activity. The complete isolation process of pure bis-bibenzyls has been described in our previous work. 17
Blood Sample Preparation
A blood sample was obtained from a healthy, non-smoking, young volunteer donor. The blood sampling was performed in compliance with the relevant laws and institutional guidelines, following the standards of the Ethics Committee of the Vinca Institute—National Institute of the Republic of Serbia,
31
the Declaration of Helsinki,
32
Good Clinical Practice,
33
and meeting the requirements specified by the current Health and Ethical regulations in Serbia.
34
Aliquots of heparinized whole blood (0.5 mL) were added to culture tubes containing 4.5 mL of Panserin 701. An adequate number of blood cultures was established to enable examination of micronuclei, CBPI, catalase activity, and MDA level. All cultures were incubated for 72 h. After 24 h of initiation, the blood cultures were treated with increasing concentrations of P endiviifolia extract (final concentrations in blood cultures 5 µg/mL, 10 µg/mL, and 50 µg/mL), and its pure constituents perrottetin E, 10′-hydroxyperrottetin E, and 10,10′-dihydroxyperrottetin E (final concentrations in blood cultures were 1 µM, 25 µM
For each analysis performed, the blood cultures were set up in triplicate. The obtained data were pooled, and the results are expressed as the mean and standard deviation (SD) of the mean.
Cytokinesis-Block Micronucleus Assay
For MN preparation, the CBMN assay of Fenech
35
was used. Cytochalasin B at a final concentration of 4 µg/mL was added to each culture 44 h after incubation to inhibit cytokinesis. The lymphocyte cultures were incubated for a further 28 h. Cells were collected by centrifugation and treated with hypotonic solution (0.56% KCl + 0.90% NaCl, mixed in equal volumes) at 37 °C. The cell suspension was fixed in methanol/acetic acid (3:1), washed three times with fixative
A CBPI was calculated according to a method of Surrales, 36 as follows: CBPI = (MI + 2MII + 3[MIII + MIV])/N, where MI–MIV represent the number of cells with one to four nuclei, respectively, and N is the number of cells scored. For its determination, at least 500 cells per slide were counted.
Apoptosis of Leukocytes
Aliquots of heparinized whole blood (0.5 mL) were added to culture tubes containing 4.5 mL of RPMI. One hour after initiation, the blood cultures were treated with increasing concentrations of P endiviifolia extract and its constituents. After 24 h of incubation, cells were gently washed with physiological saline (0.9% NaCl) at 37 °C and fixed in methanol/acetic acid (3:1). Afterward, the pellet was fixed in 96% ethanol and stored at + 4 °C. Samples were incubated at room temperature for 30 min in phosphate buffer with propidium iodide and ribonuclease A. Apoptosis was assessed by flow cytometric (CyFlow Cube 6) identification of cells displaying apoptosis-associated DNA condensation. DNA content was assessed by measuring the ultraviolet fluorescence of propidium iodide-stained DNA. The apoptotic population was calculated using CyFlow software. At least 10 000 cells per sample were analyzed.
Catalase Activity Analysis
Catalase activity was measured in erythrocytes using the method of Aebi,37,38 with minor modifications, by following the catalytic reduction of hydrogen peroxide (H2O2). The thawed hemolyzed erythrocytes were diluted 1:500 with phosphate buffer. The enzyme activity measurement was performed by monitoring the decrease in the decomposition of the substrate (H2O2) using a Perkin Elmer Lambda 25 Spectrophotometer at 240 nm. The blank contained enzyme hemolysate and substrate-free phosphate buffer. Catalase activity was measured by adding H2O2 in phosphate buffer to diluted hemolysate. The activity was expressed as K: the rate constant of the first-order reaction per minute per mg of Hb. The rate constant was calculated using the following formula: K = (2,3/Δt)(logA1/A2) (min−1mgHb−1), in which A1 and A2 were the absorbances of the samples at the beginning and the end of the measurement, respectively, and t was the time interval of the measurement. The K value was normalized to the hemoglobin concentration of each sample. Hemoglobin concentration was determined spectrophotometrically at 540 nm by using Drabkin's method. 39
Thiobarbituric Acid Analysis
A thawed lymphocyte suspension was treated with TBA and used to determine MDA levels spectrophotometrically at 532 nm. 40 Values are expressed as nmol TBA-reactive substance (MDA equivalent)/mg protein, using a standard curve of 1,1,3,3-tetramethoxypropane. Protein concentration was determined according to the method of Lowry. 41
Statistics
Statistical analysis was performed using the Product-Moment and partial correlations and One-way ANOVA followed by post-hoc Tukey test using the statistical software package Statistics, version 8 for Microsoft Windows and OriginPro 8.5.1. P values <.05 were considered significant.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X211056422 - Supplemental material for Assessment of the Biological Effects of Pellia endiviifolia and its Constituents in Vitro
Supplemental material, sj-doc-1-npx-10.1177_1934578X211056422 for Assessment of the Biological Effects of Pellia endiviifolia and its Constituents in Vitro by Ivana Ivković, Danka Bukvički, Miroslav Novaković, Ivana Majstorović, Andreja Leskovac, Sandra Petrović and Milan Veljić in Natural Product Communications
Footnotes
Author Contributions
Collection: Milan Veljić; Plant preparation and extraction: Miroslav Novaković, Ivana Ivković and Danka Bukvički; Conceptualization and methodology: Milan Veljić, Danka Bukvički and Andreja Leskovac; Experimental work: Ivana Ivković, Miroslav Novaković, Ivana Majstorović, Andreja Leskovac and Sandra Petrović; Data analysis, writing, editing, and review of the manuscript: Sandra Petrović, Andreja Leskovac and Ivana Ivković; Overall supervision: Sandra Petrović and Andreja Leskovac. All authors contributed to the preparation of the manuscript and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Ministry of Education, Science and Technological Development of the Republic of Serbia, under the contract numbers 451-03-9/2021-14/200017, 451-03-9/2021-14/200178
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the relevant laws and institutional guidelines, following the standards of the Ethics Committee of the Vinca Institute—National Institute of the Republic of Serbia (2019), the Declaration of Helsinki (WMA Declaration of Helsinki—Ethical Principles for Medical Research Involving Human Subjects, 2013), the Good Clinical Practice (EMA Guideline for Good Clinical Practice GCMP/ICH/135/95, 2015) and meeting the requirements specified by the current Health and Ethical regulations in Serbia (Law on Health Care, Official Gazette of the Republic of Serbia, no. 25/2019-40, 2019).
Statement of Informed Consent
Verbal informed consent was obtained from the healthy volunteer for his anonymized information to be published in this article.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
