Abstract
A series of novel betulin derivatives containing hydrazide-hydrazone moieties were synthesized. All compounds were evaluated for their cytotoxicity against four human carcinoma cell lines (HepG2, A549, MCF-7 and HCT-116) and a normal human gastric epithelial cell line (GES-1). Among them, compound
Nowadays, cancer has become the second leading cause of death worldwide.
1
The most effective therapies used in cancer treatment continue to be traditional cytotoxic agents.
2
It is worth noting that the discovery of potent anticancer drugs from natural sources is still one of the most important directions in the field of drug research.3–6 Betulin (lup-20(29)-ene-3β,28-diol, BE,

Structure of betulin (BE,
It is worth noting that there are three active positions in the betulin structure, namely the isopropenyl side chain at C-19, and two hydroxyl groups at C-3 and C-28.28–30 It is quite possible to make a chemical modification at these positions to obtain novel betulin derivatives with desired biological properties. According to the structure-activity relationship, a hydrogen donor group at C-28 can improve cytotoxic activity significantly.
Hydrazones have been demonstrated to possess antimicrobial, anti-inflammatory and antitumor activities.31–34 Hydrazones possessing an azometine -NHN = CH- proton constitute an important class of compounds for drug development. In addition, the pharmacophore ingredient with CO-NH-N = CH (hydrazide-hydrazones) has received considerable attention due to its broad spectrum of biological applications. 35 Thus, hydrazide-hydrazones are known as anti-diabetic, antileishmanial, antimalarial, and anti-tumor compounds.36–39 Hydrazide-hydrazones linked to a heterocyclic moiety such as pyrazole or pyrimidine are known to have a significant cytotoxic potential. 40 Hence, we tried to integrate the hydrazide-hydrazone moiety and heterocycles into a natural product in order to obtain new structures with remarkable cytotoxicity.
In the present work, as a part of our search for novel potential anticancer agents related to natural products, we report here on the synthesis of novel betulin derivatives containing a hydrazide-hydrazone unit as a side-chain attached to C28 of betulin. The anti-proliferative activities of the derivatives were evaluated against four human cancer cell lines in vitro.
Results and Discussion
The general procedure for the synthesis of betulin-28-hydrazide derivatives is shown in Figure 2. The 3-OH and 28-OH of betulin (

Reagents and conditions: (A) Ac2O, DMAP, pyridine, room temperature, 6 h, 80%; (B) Ti(i-PrOH)4, i-PrOH, 85°C, 5 h; 76%; (C) PCC, CH2Cl2, 35°C, 1 h, 80%; (D) NaOH, CH3OH, 80°C, 2 h, 65%; (E) CH3CH2OH, hydrazide substituent, 35°C, 5∼7 h, 76%∼85%.
Compounds
In Vitro Cytotoxicity Activity Data of Compounds
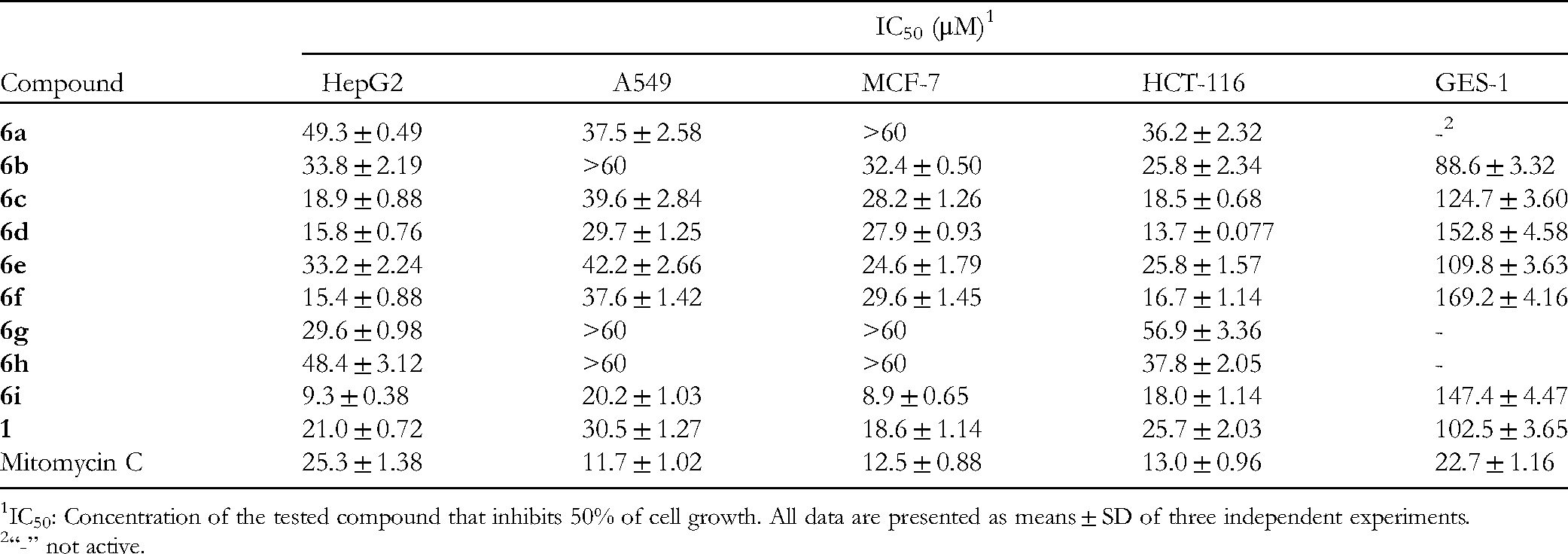
IC50: Concentration of the tested compound that inhibits 50% of cell growth. All data are presented as means ± SD of three independent experiments.
“-” not active.
For the HepG2 cell line, compounds
Experimental
General
Reagents were used without further purification. The 1H and 13C NMR spectra (δ, ppm, J, Hz) were recorded using a Bruker AVANCE NEO600 spectrometer (600 and 150 MHz, respectively) in either CDCl3 or DMSO-d6 (Bruker, Berlin, Germany). Tetramethylsilane was used as an internal standard. Melting temperatures were determined on an MP120 auto point apparatus (Haineng, Fujian, China). The mass spectra of all compounds were tested on an Esquire 6000 mass spectrometer in ESI mode (Bruker, Berlin, Germany). HRMS were obtained using an Agilent 6250 mass spectrometer (Agilient, San Francisco, USA). Flash chromatography was performed using silica gel (400 mesh) (Biohonor, Guangzhou, China).
Synthesis of 3-O,28-O-acetyl-betulin (2)
To a solution of betulin (
Synthesis of 3-O-acetyl-betulin (3 )
To a solution of intermediate (
Synthesis of 3-O-acetyl-betulinicaldehyde (4 )
To a solution of
Synthesis of Betulinicaldehyde (5 )
A solution of
General Procedure for the Synthesis of Novel Derivatives 6a-i
To a solution of
Betulin-28-(3-chloro)benzohydrazide (6a )
Yellowish powder. Yield 81%. Mp 205-207°C. 1H NMR (600 MHz, DMSO-d6) δ: 11.46 (1 H, s, NH), 7.98 (1 H, s, H28), 7.92 (1 H, s, Ar), 7.84 (1 H, d, J = 7.4 Hz, Ar), 7.65 (1 H, d, J = 7.5 Hz, Ar), 7.55 (1 H, d, J = 7.6 Hz, Ar), 4.71 (1 H, s, H29), 4.60 (1 H, s, H29), 4.26 (1 H, d, J = 3.3 Hz), 2.97 (1 H, s, OH), 1.98 (1 H, d, J = 11.9 Hz, H3), 1.86 (2 H, dd, J = 14.9, 8.9 Hz), 1.78 (2 H, d, J = 11.7 Hz), 1.68 (3 H, s, CH3), 1.66 to 1.23 (18 H, m), 1.15 (1 H, d, J = 12.0 Hz), 1.06 (1 H, d, J = 10.4 Hz), 0.97 (3 H, s, CH3), 0.93 (3 H, s, CH3), 0.87 (3 H, s, CH3), 0.75 (3 H, s, CH3), 0.65 (3 H, s, CH3). 13C NMR (150 MHz, DMSO-d6) δ: 161.5 (C = O), 157.1 (C28), 150.1 (C20), 136.0 (Ar), 133.7 (Ar), 131.9 (Ar), 130.9 (Ar), 127.6 (Ar), 126.8 (Ar), 110.5 (29), 77.2 (C3), 55.3 (C5), 50.8 (C9), 50.2 (C19), 49.3 (C17), 48.2 (C18), 42.9 (C14), 40.8 (C8), 38.9 (C4), 38.7 (C1), 38.6 (C13), 37.2 (C10), 36.8 (C7), 34.4 (C22), 32.2 (C21), 29.7 (C16), 28.6 (C23), 28.0 (C2), 27.6 (C15), 25.3 (C12), 20.8 (C11), 19.2 (C30), 18.4 (C6), 16.5 (C25), 16.4 (C26), 16.3 (C24), 14.9 (C27). HRMS (ESI): calcd for C37H54ClN2O2 [M + H]+: 593.3874, found: 593.3868.
Betulin-28-(4-chloro)benzohydrazide (6b )
Yellowish powder. Yield 84%. Mp 208-210°C. 1H NMR (600 MHz, DMSO-d6) δ: 11.43 (1 H, s, NH), 7.98 (1 H, s, H28), 7.90 (2 H, d, J = 8.2 Hz, Ar), 7.59 (2 H, d, J = 8.2 Hz, Ar), 4.71 (1 H, s, H29), 4.60 (1 H, s, H29), 4.27 (1 H, d, J = 4.8 Hz), 3.17 (1 H, d, J = 4.2 Hz, OH), 3.04-2.93 (m, 1 H), 1.68 (3 H, s, CH3), 0.97 (3 H, s, CH3), 0.93 (3 H, s, CH3), 0.87 (3 H, s, CH3), 0.76 (3 H, s, CH3), 0.65 (3 H, s, CH3). 13C NMR (150 MHz, DMSO-d6) δ: 161.9 (C = O), 156.8 (C28), 150.1 (C20), 136.8 (Ar), 132.8 (Ar), 129.9 (Ar), 128.9 (Ar), 110.5 (C29), 77.2 (C3), 55.3 (C5), 50.8 (C9), 50.2 (C19), 49.3 (C17), 48.2 (C18), 42.9 (C14), 40.9 (C8), 39.0 (C4), 38.7 (C1), 38.6 (C13), 37.2 (C10), 36.8 (C7), 34.4 (C22), 32.2 (C21), 29.7 (C16), 28.6 (C23), 28.0 (C2), 27.6 (C15), 25.3 (C12), 20.8 (C11), 19.2 (C30), 18.4 (C6), 16.5 (C25), 16.4 (C26), 16.3 (C24), 14.9 (C27). HRMS (ESI): calcd for C37H54ClN2O2 [M + H]+: 593.3874, found: 593.3868.
Betulin-28-(4-bromo)benzohydrazide (6c )
Yellowish powder. Yield 76%. Mp 202-204°C. 1H NMR (600 MHz, DMSO-d6) δ: 11.43 (1 H, s, NH), 7.98 (1 H, s, H28), 7.82 (2 H, d, J = 8.4 Hz, Ar), 7.73 (2 H, d, J = 8.4 Hz, Ar), 4.71 (1 H, s, H29), 4.60 (s, 1 H, H29), 3.34 (1 H, s, OH), 1.68 (3 H, s, CH3), 0.97 (s, 3 H, CH3), 0.93 (3 H, s, CH3), 0.87 (3 H, s, CH3), 0.76 (3 H, s, CH3), 0.65 (3 H, s, CH3). 13C NMR (150 MHz, DMSO-d6) δ: 162.0 (C = O), 156.9 (C28), 150.1 (C20), 133.1 (Ar), 131.9 (Ar), 130.1 (Ar), 125.8 (Ar), 110.5 (C29), 77.2 (C3), 55.3 (C5), 50.8 (C9), 50.2 (C19), 49.3 (C17), 48.2 (C18), 42.9 (C14), 40.9 (C8), 39.0 (C4), 38.7 (C1), 38.6 (C13), 37.2 (C10), 36.8 (C7), 34.4 (C22), 32.2 (C21), 29.7 (C16), 28.6 (C23), 28.0 (C2), 27.6 (C15), 25.3 (C12), 20.8 (C11), 19.2 (C30), 18.4 (C6), 16.5 (C25), 16.4 (C26), 16.3 (C24), 14.9 (C27). HRMS (ESI): calcd for C37H54BrN2O2 [M + H]+: 637.3369, found: 637.3363.
Betulin-28-(4-trifluoromethyl)benzohydrazide (6d )
White powder. Yield 80%. Mp 197-199°C. 1H NMR (600 MHz, DMSO-d6) δ: 11.57 (1 H, s, NH), 8.07 (2 H, d, J = 7.7 Hz, Ar-H), 8.00 (1 H, s, H28), 7.90 (2 H, d, J = 7.8 Hz, Ar-H), 4.72 (1 H, s, H29), 4.60 (1 H, s, H29), 4.27 (1 H, s), 2.97 (1 H, s), 1.69 (3 H, s, CH3), 0.98 (3 H, s, CH3), 0.93 (3 H, s, CH3), 0.87 (3 H, s, CH3), 0.76 (3 H, s, CH3), 0.65 (3 H, s, CH3). 13C NMR (150 MHz, DMSO-d6) δ: 161.8 (C = O), 157.4 (C28), 150.1 (C20), 137.9 (CF3), 131.9 (Ar), 123.0 (Ar), 128.9 (Ar), 125.3 (Ar), 123.5 (Ar), 110.6 (C29), 77.2 (C3), 55.3 (C5), 50.8 (C9), 50.2 (C19), 49.3 (C17), 48.2 (C18), 42.9 (C14), 40.9 (C8), 39.0 (C4), 38.7 (C1), 38.6 (C13), 37.2 (C10), 36.7 (C7), 34.4 (C22), 32.2 (C21), 29.7 (C16), 28.5 (C23), 28.0 (C2), 27.6 (C15), 25.3 (C12), 20.8 (C11), 19.2 (C30), 18.4 (C6), 16.5 (C25), 16.4 (C26), 16.3 (C24), 14.9 (C27). HRMS (ESI): calcd for C38H54F3N2O2 [M + H]+: 627.4137, found: 627.4131.
Betulin-28-(4-methoxy)benzohydrazide (6e )
White powder. Yield 77%. Mp 191-193°C. 1H NMR (600 MHz, DMSO-d6) δ: 11.25 (1 H, s, NH), 7.96 (1 H, s, H28), 7.87 (2 H, t, J = 14.7 Hz, Ar), 7.04 (2 H, d, J = 8.2 Hz, Ar), 4.71 (1 H, s, H29), 4.59 (1 H, s, H29), 4.27 (1 H, d, J = 5.1 Hz, OH), 3.83 (3 H, s, OCH3), 1.68 (3 H, s, CH3), 0.97 (3 H, s, CH3), 0.93 (3 H, s, CH3), 0.87 (3 H, s, CH3), 0.76 (3 H, s, CH3), 0.65 (3 H, s, CH3). 13C NMR (150 MHz, DMSO-d6) δ: 162.4 (C = O), 155.7 (C28), 150.2 (C20), 129.8 (Ar), 126.1 (Ar), 114.1 (Ar), 110.5 (C29), 77.2 (C3), 55.8 (OCH3), 55.3 (C5), 50.7 (C9), 50.2 (C19), 49.3 (C17), 48.2 (C18), 42.9 (C14), 40.9 (C8), 39.0 (C4), 38.7 (C1), 38.6 (C13), 37.2 (C10), 36.9 (C7), 34.4 (C22), 32.3 (C21), 29.8 (C16), 28.6 (C23), 28.0 (C2), 27.6 (C15), 25.3 (C12), 20.8 (C11), 19.2 (C30), 18.4 (C6), 16.5 (C25), 16.4 (C26), 16.3 (C24), 14.9 (C27). HRMS (ESI): calcd for C38H57N2O3 [M + H]+: 589.4369, found: 589.4378.
Betulin-28-(4-hydroxy)benzohydrazide (6f )
White powder. Yield 84%. Mp 213-215°C. 1H NMR (600 MHz, DMSO-d6) δ: 11.16 (1 H, s, NH), 10.05 (1 H, s, Ar-OH), 7.95 (1 H, s, H28), 7.75 (2 H, d, J = 7.0 Hz, Ar-H), 6.83 (2 H, d, J = 6.1 Hz, Ar-H), 4.71 (1 H, s, H29), 4.59 (1 H, s, H29), 4.22 (1 H, t, J = 6.0 Hz, 3-OH), 2.97 (1 H, m, H3), 1.98 (1 H, m), 1.81 (4 H, m), 1.66 (3 H, s, CH3), 1.64 to 1.16 (17 H, m), 0.97 (3 H, s, CH3), 0.91 (3 H, s, CH3), 0.89 (m, 2 H), 0.87 (3 H, s, CH3), 0.76 (3 H, s, CH3), 0.65 (3 H, s, CH3). 13C NMR (150 MHz, DMSO-d6) δ: 160.9 (C = O), 155.3 (C28), 150.2 (C20), 132.2 (Ar), 132.0 (Ar), 129.9 (Ar), 129.1 (Ar), 124.5 (Ar), 115.4 (Ar), 110.5 (C29), 77.2 (C3), 55.3 (C5), 50.6 (C9), 50.2 (C19), 49.3 (C17), 48.2 (C18), 42.9 (C14), 40.9 (C8), 38.9 (C4), 38.7 (C1), 38.6 (C13), 37.1 (C10), 34.4 (C7), 34.3 (C22), 30.5 (C21), 29.7 (C16), 28.5 (C23), 29.0 (C2), 27.6 (C15), 25.6 (C12), 20.8 (C11), 19.2 (C30), 18.4 (C6), 16.5 (C25), 16.4 (C26), 16.3 (C24), 14.8 (C27). HRMS (ESI): calcd for C37H55N2O3 [M + H]+: 575.4213, found: 575.4219.
Betulin-28-(2-furan)carbohydrazide (6 g )
Yellowish powder. Yield 75%. Mp 187-189°C. 1H NMR (600 MHz, CDCl3) δ: 9.19 (1 H, s, NH), 7.88 (1 H, s, H28), 7.47 (2 H, m, furan), 6.54 (1 H, dd, J = 3.3, 1.6 Hz, furan), 4.72 (1 H, s, H29), 4.62 (1 H, s, H29), 3.19 (1 H, dd, J = 11.5, 4.6 Hz, OH), 2.52 (1 H, s, H3), 2.19 (1 H, m), 2.05 (1 H, m), 2.02-1.75 (6 H, m), 1.720 (3 H, m, CH3), 1.66-1.04 (17 H, m), 1.00 (3 H, s, CH3), 0.99-0.92 (6 H, m, 2 × CH3), 0.92-0.87 (2 H, m), 0.81 (3 H, s, CH3), 0.75 (3 H, s, CH3), 0.68 (1 H, d, J = 10.8 Hz). 13C NMR (150 MHz, CDCl3) δ: 171.2 (C = O), 157.7 (C28), 149.8 (C20), 144.1 (C1’), 115. 7 (C2’, C4’), 112.4 (C3’), 110.1 (C29), 78.9 (C3), 55.3 (C5), 51.2 (C9), 50.3 (C19), 49.3 (C17), 48.1 (C18), 42.8 (C14), 40.9 (C8), 38.8 (C4), 38.7 (C1), 37.3 (C13), 37.2 (C10), 37.1 (C7), 34.2 (C22), 32.2 (C21), 29.8 (C16), 29.7 (C23), 28.0 (C2), 27.3 (C15), 25.3 (C12), 20.7 (C11), 19.1 (C30), 18.2 (C6), 16.4 (C25), 16.3 (C26), 15.4 (C24), 14.7 (C27). HRMS (ESI): calcd for C35H53N2O3 [M + H]+: 549.4056, found: 549.4051.
Betulin-28-(2-thiophene)carbohydrazide (6 h )
Yellowish powder. Yield 76%. Mp 176-178°C. 1H NMR (600 MHz, CDCl3) δ: 9.99 (1 H, s, NH), 8.18 (1 H, s, H28), 7.63 (1 H, d, J = 3.5 Hz, thio-), 7.50 (1 H, s, thio-), 7.18-7.10 (1 H, m, thio-), 4.74 (1 H, s, H29), 4.63 (1 H, s, H29), 3.18 (1 H, dd, J = 11.5, 4.6 Hz, 3-OH), 2.60 (1 H, d, J = 4.6 Hz, H3), 2.16-1.85 (7 H, m), 1.72 (3 H, s, CH3), 1.67-1.08 (18 H, m), 1.01 (3 H, s, CH3), 0.96 (3 H, s, CH3), 0.90 (3 H, s, CH3), 0.87 (2 H, m), 0.78 (3 H, s, CH3), 0.75 (3 H, s, CH3), 0.67 (1 H, d, J = 11.1 Hz). 13C NMR (150 MHz, CDCl3) δ: 162.6 (C = O), 152.5 (C28), 149.8 (C20), 135.1 (C1’), 134.1 (C2’), 133.0 (C3’), 128.8 (C4’), 110.1 (C29), 78.9 (C3), 55.3 (C5), 51.1 (C9), 50.4 (C19), 49.4 (C17), 48.0 (C18), 42.9 (C14), 40.9 (C8), 38.8 (C4), 38.7 (C1), 38.6 (C13), 37.1 (C10), 37.0 (C7), 34.2 (C22), 32.6 (C21), 29.9 (C16), 29.8 (C23), 28.0 (C2), 27.4 (C15), 25.3 (C12), 20.7 (C11), 19.1 (C30), 18.2 (C6), 16.1 (C25), 16.0 (C26), 15.4 (C24), 14.7 (C27). HRMS (ESI): calcd for C35H53N2O2S [M + H]+: 565.3828, found: 565.3822.
Betulin-28-isonicotinohydrazide (6i )
Yellowish powder. Yield 78%. Mp 221-223°C. 1H NMR (600 MHz, DMSO-d6) δ: 11.58 (1 H, s, NH), 8.76 (2 H, d, J = 5.6 Hz, Py), 8.00 (1 H, s, H28), 7.78 (2 H, d, J = 5.7 Hz, Py), 4.71 (1 H, s, H29), 4.60 (1 H, s, H29), 4.27 (1 H, m, 3-OH), 3.04-2.93 (1 H, m, H3), 1.98 (1 H, d, J = 12.2 Hz), 1.86 (2 H, d, J = 9.0 Hz), 1.78 (2 H, m), 1.68 (3 H, s, CH3), 1.67-1.00 (17 H, m), 0.98 (3 H, s, CH3), 0.93 (3 H, s, CH3), 0.87 (3 H, s, CH3), 0.85 (2 H, m), 0.76 (3 H, s, CH3), 0.74 (1 H, s), 0.65 (3 H, s, CH3), 0.63 (1 H, s). 13C NMR (150 MHz, DMSO-d6) δ: 161.4 (C = O), 157.9 (C28), 150.7 (C20), 150.1 (C1’), 141.1 (C2’), 121.9 (C3’), 110.6 (C29), 77.2 (C3), 55.3 (C5), 50.8 (C9), 50.2 (C19), 49.3 (C17), 48.2 (C18), 42.9 (C14), 40.9 (C8), 39.0 (C4), 38.7 (C1), 38.6 (C13), 37.2 (C10), 36.7 (C7), 34.5 (C22), 32.2 (C21), 29.7 (C16), 28.6 (C23), 28.0 (C2), 27.6 (C15), 25.3 (C12), 20.8 (C11), 19.2 (C30), 18.4 (C6), 16.4 (C25), 16.3 (C26), 16.2 (C24), 14.9 (C27). HRMS (ESI): calcd for C36H54N3O2 [M + H]+: 560.4216, found: 560.4211.
In Vitro Cytotoxic Activity
All cell lines were obtained from the Shanghai Cell Bank of the Chinese Academy of Science. Cells were cultured in RPMI-1640 medium supplemented with 10% FBS, 100 units/mL of penicillin and 100 μg/mL streptomycin at 37 °C in a humidified atmosphere of 5% CO2. Cytotoxic activities of all tested compounds against four cancer cell lines were evaluated by MTT assay. Cells were seeded into 96-well plates (1 × 104 cells/well) for 24 h. Then the cells were treated with compounds at gradient concentrations from 5 to 60 μM for 48 h and then 10 μL MTT (Sigma Chemical Co., Ltd, USA) solution (5 mg/mL in PBS) was added for 2 h. The solution was replaced by 100 μL DMSO, and the absorbance was measured at 490 nm on a Spectra Max 340 microplate reader. The IC50 values were derived by SPSS nonlinear regression analysis.
Conclusions
In summary, a series of novel betulin derivatives containing a hydrazide-hydrazone side-chain were synthesized by five steps. The yields of these derivatives were ideal from 76% to 85%, which indicated that the synthetic route was designed reasonably. All compounds were evaluated for their in vitro antiproliferative activities against four human carcinoma cell lines (HepG2, A549, MCF-7 and HCT-116). Among the compounds under biological study, compound
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211055345 - Supplemental material for Synthesis and Cytotoxic Activity of Novel Betulin Derivatives Containing Hydrazide-Hydrazone Moieties
Supplemental material, sj-docx-1-npx-10.1177_1934578X211055345 for Synthesis and Cytotoxic Activity of Novel Betulin Derivatives Containing Hydrazide-Hydrazone Moieties by Jiale Wu, Jiafeng Wang, Yinglong Han, Yu Lin, Jing Wang and Ming Bu in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Central Government Support Fund for the Reform and Development of Local Universities-Talent Training Support Program Project (grant number ZYZX2019).
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
