Abstract
A series of new hydroxamate derivatives of lupane triterpenoids has been designed and successfully synthesized. The synthesized compounds were evaluated for their in vitro antitumor activity using the 3-[4,5-dimethylthiazol-2-yl]−2,5-diphenyltetrazolium bromide-based assay against the human cancer cell lines KB and HepG2. Most of these derivatives possess at least moderate cytotoxic activity and the hydroxamate derivative compounds
Introduction
Cancer is a dangerous disease which caused about 9.6 million deaths in 2018. 1 In recent decades, there has been a lot of research and development on new anticancer drugs, but there is still an urgent need for the further search and development of new improved drugs to target the inhibition of cancer cells. Many studies have shown that the molecular pathways are related to the cellular epigenome, 2 -4 and their dysregulation has an effect on cancer cells. 5 -7
Histone deacetylases (HDACs) are enzymes that are known to be overexpressed in cancer cell lines, 8 -13 and hydroxamate has been recognized as a zinc-binding group in the molecular structure of HDAC inhibitors. Hydroxamic acids have also been considered in the design for cancer treatment. 14 -17 Lead compounds of this class were approved by the Food and Drug Administration for cancer treatment, such as Zolinza (SAHA) (Figure 1) for the treatment of cutaneous T-cell lymphoma, 18 Panobinostat for multiple myeloma 19 in 2006, and Belinostat (PXD101) for peripheral T-cell lymphoma 20 in 2014; CUDC-101, Trichostatin A, and Oxamflatin (Figure 1) are currently undergoing clinical trials against different cancer cell lines. 21 Therefore, hydroxamate derivatives have been designed and synthesized as a promising class of anticancer agents. 22,23

Structures of some histone deacetylase inhibitors.
Some lupane triterpenoids, such as betulin, betulinic acid, and its derivatives, have attracted considerable attention in recent years because of their activities against cancer cell lines. 24 -28 Reports have been rather limited so far on the combination of a triterpenoid and hydroxamate into single molecules, as well as their screening for anticancer activity. After considering the biological potential of such compounds, we decided to construct novel triterpenoid derived hydroxamates with the expectation of their potential use as anticancer agents. Therefore, in this study, a series of novel lupane derived hydroxamates were designed and synthesized at the C-28 position of the triterpenoid. The synthesized compounds were evaluated for their cytotoxic activity against 2 human cancer cell lines (KB and HepG2).
Results and Discussion
Chemistry
In this study, the conjugates of betulin derivatives with either hydroxylammonium chloride (NH2OH.HCl) or
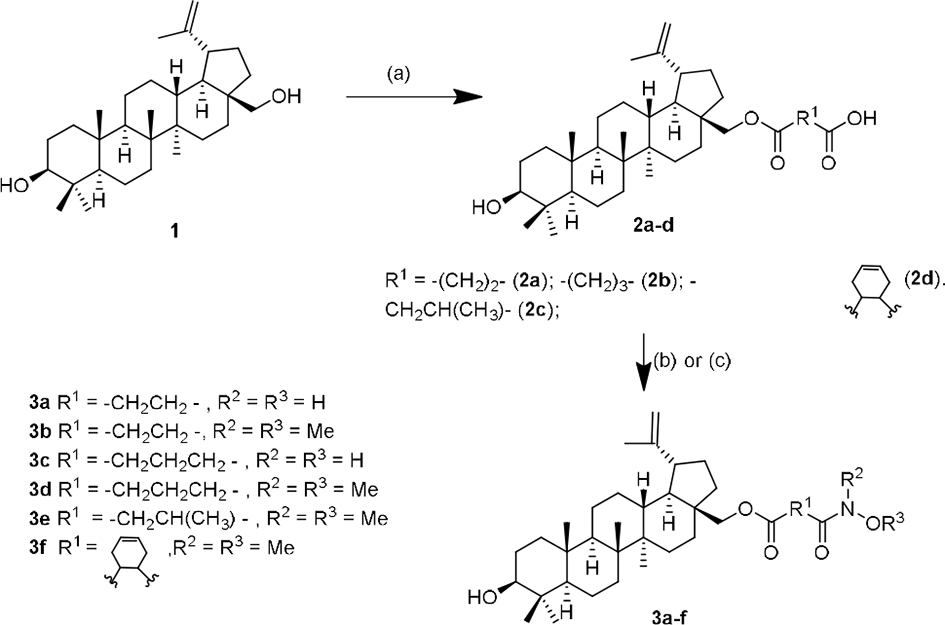
Synthesis of hydroxamates 3a-f from betulin derivatives. Reagents and conditions: (a) 4 equiv anhydride, 1.2 equiv Et3N, dichloromethane, room temperature (rt), 24 hours; (b) 2 equiv hydroxylammonium chloride, 1 equiv BOP, 2 equiv 4-dimethylaminopyridine (DMAP), dimethylformamide (DMF), rt, 15 hours; (c) 2 equiv
Analogously, the reaction of 3

Synthesis of hydroxamates 5a,b and 7a,b. Reagents and conditions:(a) 2 equiv hydroxylammonium chloride, 1 equiv BOP, 2 equiv 4-dimethylaminopyridine (DMAP), dimethylformamide (DMF), room temperature (rt), 15 hours; (b) 2 equiv

Synthesis of hydroxamates 10a,b from betulinic acid. Reagents and conditions: (a) 2 equiv H2N-(CH2)5-COOH, 2 equiv 4-dimethylaminopyridine (DMAP), 1 equiv BOP, dimethylformamide (DMF), room temperature (rt), 24 hours; (b) 2 equiv hydroxylammonium chloride, 1 equiv BOP, 2 equiv DMAP, DMF, rt, 15 hours; (c) 2 equiv
Finally, the synthesis of hydoxamates

Synthesis of hydroxamates 15a,b. Reagents and conditions: (a) chromium trioxide, sulfuric acid, acetone, 0°C-room temperature (rt); (b) 4 equiv sodium borohydride, methanol, tetrahydrofuran, 0°C-rt, 10 hours; (c) 2 equiv H2N-(CH2)5-COOH, 1 equiv BOP, 2 equiv 4-dimethylaminopyridine (DMAP), dimethylformamide (DMF), rt, 24 hours; (d) 2 equiv hydroxylammonium chloride, 2 equiv DMAP, 1 equiv BOP, DMF, rt, 15 hours; (e) 2 equiv
Biology
The newly prepared compounds were subjected to cytotoxic evaluation in order to assess their biological relevance. Betulin derived hydroxamates
Synthesis and Cytotoxicity of Synthesized Compounds.

IC50, half-maximal inhibitory concentration.
aPositive control compound.
Conclusion
To sum up, 14 new hydroxamate derivatives of lupane triterpenoids have been designed and successfully synthesized. The synthesized compounds were evaluated using the MTT-based assay against the 2 human cancer cell lines (KB and HepG2). The results indicate that most of these derivatives possess at least moderate cytotoxic activity and compounds
Experimental
General: Chemistry
The reagents were commercially available and used without further purification. Analytical thin-layer chromatography (TLC) was performed on HSGF 254 (0.15, 0.2 mm thickness). Melting points were measured on a Boetius apparatus and are uncorrected. Column chromatography was performed using silica gel (60 Å, particle size 40-60 µm). NMR spectra were recorded on a BrukerAvance (500 MHz) spectrophotometer. Chemical shifts (
Syntheses
General procedure for preparation of conjugates between the triterpenoid and NH2OH.HCl and HNMeOMe.HCl 3a-f, 5a,b, 7a,b, 10a,b, and 15a,b
To a solution of the corresponding acid
((1R ,3aS ,5aR ,5bR ,9S ,11aR )-9-hydroxy-5a,5b,8,8,11a-pentame-thyl-1-(prop-1-en-2-yl)icosahydro-3aH -cyclopenta[a ]chrysen-3a-yl)methyl 4-(hydroxyamino)-4-oxobutanoate (3a )
White solid. Yield 57%.
Mp: 227-229oC.
IR (KBr) cm−1: 3367, 2958, 2879, 1706, 1698, 1567, 1454, 1367, 1251, 1138, 1093, 883.
1H NMR (500 MHz, dimethylsulfoxide [DMSO]-
13C NMR (125 MHz, DMSO-
LC–MS/MS: Found
((1R ,3aS ,5aR ,5bR ,9S ,11aR )-9-hydroxy-5a,5b,8,8,11a-pentame-thyl-1-(prop-1-en-2-yl)icosahydro-3aH -cyclopenta[a ]chrysen-3a-yl)methyl 4-(methoxy(methyl)amino)-4-oxobutanoate (3b )
White solid. Yield 63%.
Mp: 237-239°C.
IR (KBr) cm−1: 2940, 2869, 1733, 1667, 1534, 1455, 1360, 1278, 1174, 1045, 883.
1H NMR (500 MHz, deuterated chloroform [CDCl3])
13C NMR (125 MHz, CDCl3)
LC–MS/MS: Found
((1R ,3aS ,5aR ,5bR ,9S ,11aR )-9-hydroxy-5a,5b,8,8,11a-pentame-thyl-1-(prop-1-en-2-yl)icosahydro-3aH -cyclopenta[a ]chrysen-3a-yl)methyl 5-(hydroxyamino)-5-oxopentanoate (3c )
White solid. Yield 61%.
Mp: 217-219oC.
IR (KBr) cm−1: 3329, 2987, 2876, 1743,1697, 1561, 1455, 1389, 1238, 1109, 1087, 776.
1H NMR (500 MHz, DMSO-
13C NMR (125 MHz, DMSO-
LC–MS/MS: Found
((1R ,3aS ,5aR ,5bR ,9S ,11aR )-9-hydroxy-5a,5b,8,8,11a-pentame-thyl-1-(prop-1-en-2-yl)icosahydro-3aH -cyclopenta[a ]chrysen-3a-yl)methyl 5-(methoxy(methyl)amino)-5-oxopentanoate (3d )
White solid. Yield 61%.
Mp: 223-225oC.
IR (KBr) cm−1: 2939, 2868, 1723, 1641, 1557, 1403, 1359, 1260, 1193, 1087, 878.
1H NMR (500 MHz, CDCl3)
13C NMR (125 MHz, CDCl3)
LC–MS/MS: Found
((1R ,3aS ,5aR ,5bR ,9S ,11aR )-9-hydroxy-5a,5b,8,8,11a-pentame-thyl-1-(prop-1-en-2-yl)icosahydro-3aH -cyclopenta[a ]chrysen-3a-yl)methyl 4-(methoxy(methyl)amino)-3-methyl-4-oxobutanoate (3e )
White solid. Yield 63%.
Mp: 229-231oC.
IR (KBr) cm−1: 2939, 2869, 1732, 1668, 1553, 1460, 1384, 1224, 1176, 1046, 880.
1H NMR (500 MHz, CDCl3)
13C NMR (125 MHz, CDCl3)
LC–MS/MS: Found
((1R ,3aS ,5aR ,5bR ,9S ,11aR )-9-hydroxy-5a,5b,8,8,11a-pentame-thyl-1-(prop-1-en-2-yl)icosahydro-3aH -cyclopenta[a ]chrysen-3a-yl)methyl 6-(methoxy(methyl)carbamoyl)cyclohex-3-ene-1-carboxylate (3f )
White solid. Yield 62%.
Mp: 217-219oC.
IR (KBr) cm−1: 3074, 2963, 2866, 1733, 1711, 1510, 1454, 1384, 1291, 1185, 1020, 881.
1H NMR (500 MHz, CDCl3)
13C NMR (125 MHz, CDCl3)
LC–MS/MS: Found
(3aR ,5aR ,5bR ,11aR )-3a-(hydroxycarbamoyl)-1-isopropyl-5a,5b,8,8,11a-pentamethyl-2-oxo-3,3a,4,5,5a,5b,6,7,7a,8,9, 10,11,11a,11b,12,13,13a-octadecahydro-2H -cyclopenta[a ]- chrysen-9-yl acetate (5a )
White solid. Yield 59%.
Mp: 203-205 oC.
IR (KBr) cm−1: 3435, 2948, 2856, 1735, 1647,1673, 1381, 1246, 1025, 825.
1H NMR (500 MHz, DMSO-
13C NMR (125 MHz, DMSO-
LC–MS/MS: Found
(3aR ,5aR ,5bR ,11aR )-1-isopropyl-3a(methoxy(methyl)carbamoyl)-5a,5b,8,8,11a-pentamethyl-2-oxo-3,3a,4,5,5a,5b,6,7,7a,8,9,10,11,11a,11b,12,13,13a-octadecahydro-2H -cyclopenta[a ]chrysen-9-yl acetate (5b )
White solid. Yield 60%.
Mp: 237-239oC.
IR (KBr) cm−1: 2940, 2876, 1731, 1698, 1660, 1455, 1365, 1246, 1106, 1024, 869.
1H NMR (500 MHz, CDCl3)
13C NMR (125 MHz, CDCl3)
LC–MS/MS: Found
(3aR ,5aR ,5bR ,9S ,11aR )-3a-((6-(hydroxyamino)−6-oxohexyl)carbamoyl)−1-isopropyl-5a,5b,8,8,11ª-pentamethyl-2-oxo-3,3a,4,5,5a,5b,6,7,7a,8,9,10,11,11a,11b,12,13,13a-octadecahydro-2H -cyclopen-ta[a ]chrysen-9-yl acetate (7a )
White solid. Yield 53%.
Mp: 214-216oC.
IR (KBr) cm−1: 3259, 2946, 2872, 1735, 1693, 1685, 1674, 1527, 1456, 1378, 1245, 1106, 1027, 731.
1H NMR (500 MHz, DMSO-
13C NMR (125 MHz, DMSO-
LC–MS/MS: Found
(3aR ,5aR ,5bR ,11aR )-1-isopropyl-3a-((6-(methoxy(methyl)ami-no)−6-oxohexyl)carbamoyl)−5a,5b, 8,8,11a-pentamethyl-2-oxo-3,3a,4,5,5a,5b,6,7,7a,8,9,10,11,11a,11b,12,13,13a-octadecahydro-2H -cyclopenta[a ]chrysen-9-yl acetate (7b )
White solid. Yield 64%.
Mp: 223-225oC.
IR (KBr) cm−1: 3328, 2927, 2853, 1719, 1669, 1695, 1628, 1512, 1452, 1378, 1252, 1186, 1086, 894.
1H NMR (500 MHz, CDCl3)
13C NMR (125 MHz, CDCl3)
LC–MS/MS: Found
(1R ,3aS ,5aR ,5bR ,9S ,11aR )-9-hydroxy-N -(6-(hydroxyamino)-6-oxohexyl)−5a,5b,8,8,11a-pentame-thyl-1-(prop-1-en-2-yl)icosa-hydro-3aH -cyclopenta[a ]chrysene-3a-carboxamide (10a )
White solid. Yield 61%.
Mp: 276-278 oC.
IR (KBr) cm−1: 3386, 3081, 2940, 2866, 1707, 1636, 1530, 1452, 1375, 1247, 1194, 1043, 847.
1H NMR (500 MHz, DMSO-
13C NMR (125 MHz, DMSO-
LC–MS/MS: Found
(1R ,3aS ,5aR ,5bR ,9S ,11aR )-9-hydroxy-N -(6-(methoxy(methyl)-amino)−6-oxohexyl)−5a,5b,8,8,11a-pentamethyl-1-(prop-1-en-2-yl)icosahydro-3aH -cyclopenta[a ]chrysene-3a-carboxamide (10b )
White solid. Yield 65%.
Mp: 229-231oC.
IR (KBr) cm−1: 3371, 2934, 2864, 1697, 1638, 1530, 1463, 1388, 1247, 1193, 1082, 882.
1H NMR (500 MHz, CDCl3)
13C NMR (125 MHz, CDCl3)
LC–MS/MS: Found
(1R ,3aS ,5aR ,5bR ,8R ,9S ,11aS )-9-hydroxy-N -(6-(hydroxyamino)-6-oxohexyl)-5a,5b,8,11a-tetrame-thyl-1-(prop-1-en-2-yl)icosa-hydro-3aH -cyclopenta[a ]chrysene-3a-carboxamide (15a )
White solid. Yield 54%.
Mp: 205-207oC.
IR (KBr) cm−1: 3362, 2976, 2953, 1714, 1687, 1538, 1475, 1321, 1269, 1108, 1089, 765.
1H NMR (500 MHz, DMSO-
13C NMR (125 MHz, DMSO-
LC–MS/MS: Found
(1R ,3aS ,5aR ,5bR ,8R ,9S ,11aS )-9-hydroxy-N -(6-(methoxy(me-thyl)amino)−6-oxohexyl)−5a,5b,8,11a-tetramethyl-1-(prop-1-en-2-yl)icosahydro-3aH -cyclopenta[a ]chrysene-3a-carboxamide (15b )
White solid. Yield 71%.
Mp: 224-226oC.
IR (KBr) cm−1: 3374, 2933, 2866, 1697, 1637, 1530, 1452, 1375, 1296, 1198, 1040, 882.
1H NMR (500 MHz, CDCl3)
13C NMR (125 MHz, CDCl3)
LC–MS/MS: Found
General: Biological Screening
Two human cancer cell lines (epidermoid carcinoma cell line KB-ATCC CCL-17 and hepatoma carcinoma cell line HepG2-ATCC HB-8065), obtained from the American Type Culture Collection (USA) ATCC, were used for cytotoxic evaluation. The cells were grown in Roswell Park Memorial Institute Medium 1640 medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 µg/mL streptomycin at 37°C in a humidified atmosphere (95% air and 5% carbon dioxide). The exponentially growing cells were used throughout the experiments. The inhibitory effects of the compounds on the growth of the human cancer cell lines were determined by measuring the metabolic activity using an MTT assay. Briefly, human cancer cell lines (1 × 105 cells/mL) were treated for 3 days with a series of concentrations of the compounds (in DMSO): 0.125, 0.5, 2.0, 8.0, 32.0, and 128.0 µg/mL. After incubation, 0.1 mg MTT solution (50 µL of a 2 mg/mL solution) was added to each well, and the cells were then incubated at 37°C for 4 hours. The plates were centrifuged at 1000 rpm for 10 minutes at room temperature, and the media were then carefully aspirated. DMSO (150 µL) was added to each well to dissolve the formazan crystals. The plates were read immediately at 540 nm on a microplate reader (TECAN GENIOUS). All the experiments were performed 3 times, and the mean absorbance values were calculated. The results are expressed as the percentage of inhibition that produced a reduction in the absorbance by the treatment of the compounds compared with the untreated controls. A dose–response curve was generated, and the IC50 value was determined for each compound as well as each cell line.
Supplemental Material
Figure S1 - Supplemental material for Design, Synthesis, and Cytotoxic Evaluation of Novel Lupane Triterpenoid Derived Hydroxamates
Supplemental material, Figure S1, for Design, Synthesis, and Cytotoxic Evaluation of Novel Lupane Triterpenoid Derived Hydroxamates by Dang Thi Tuyet Anh, Dinh Thi Cuc, Le Nhat Thuy Giang, Nguyen Thi Hien, Vu Ngoc Doan, Nguyen Ha Thanh, Nguyen Van Tuyen and Phan Van Kiem in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are indebted to Vietnam Academy of Science and Technology (VAST, code: NVCC06.01/20-20) for financial support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
