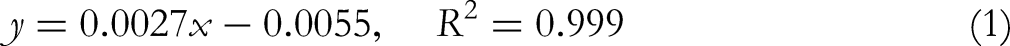Abstract
The aim of this study was to extract, identify and isolate the chemical constituents of the root and leaves of Cleome viscosa by liquid chromatography coupled to electrospray ionization mass spectrometry (LC-ESI-MS). The antioxidant activity and content of associated secondary metabolites in the extracts from different parts of the plant were also measured. The free radical scavenging activity of the methanol extracts of different parts of C viscosa was determined using DPPH assays. A wide range of polar and non- polar compounds were characterized by LC-ESI-MS. Based on the obtained results, 22 and 21 compounds were characterized in the extracts of leaf and root of C viscosa, respectively. Among these compounds, cleomeolide and 57,4′.trihydroxy-6,3.5-trimethoxyflavone, in the leaf extract, and nevirapine and vincetoxicoside A, in the root extract, were considered as main compounds. Phytochemical analysis also showed that the content of bioactive compounds varied remarkably in the extract of different parts of C viscosa. The leaf ethanol extract contained higher amounts of secondary metabolites, namely total phenols and flavonoids. The antioxidant activity of the extracts was also dependent on the plant parts, which was significantly correlated with the content of bioactive compounds studied. A strong free radical scavenging activity was observed for the methanol extract of the leaf (50.3%), followed by the flower (35.2%). In general, this study confirmed a wide range of compounds in different parts of the invasive weed C viscosa, especially in the leaf, with a high free radical scavenging activity.
Introduction
Cleome viscosa L. belongs to the family Cleomaceae, a small family of flowering plants in the order Brassicales, having approximately 300 species which belong to 9 genera. 1 C viscosa is a common weed found in tropical regions where it finds application in traditional medicine. C viscosa is a botanically erect, grooved, aromatic, glandular plant with sticky shoots. 2 A wide variety of constituents have been isolated from various parts of C viscosa, including triterpenoids, saponins, tannins, flavonoids and steroids. 3 The phenolic content of various parts of C viscosa varied, with 0.057% in leaves, 0.007% in roots, and 0.024% in stems, and the flavonoid content from 0.019% in leaves, 0.012% in roots, and 0.012% in stems. 4 Another study also found higher total phenolic contents in leaves (6.63%) and stems (5.85%) of C viscosa, while leaves (0.054%) and stems (0.048%) of this plant had lower flavonoid contents. 5 Quantitative evaluation of secondary metabolites in different parts of C viscosa also showed that the leaf extract contained the highest concentrations of alkaloids, cardiac glycosides, flavonoids, phenols, and saponins, but roots and stems had smaller amounts. 6
C viscosa is reported to have a wide range of traditional and medicinal applications. In traditional application, the whole plant and its parts (leaves, seeds, and roots) are claimed to possess effects as an antiseptic, anthelmintic, sudorific, carminative, antiscorbutic, febrifuge, and cardiac stimulant. Also, pharmacological studies have shown that C viscosa possesses various biological properties such as antimicrobial, anthelmintic, analgesic, immune modulatory, anti-inflammatory, antipyretic, antidiarrheal, psychopharmacological, and hepatoprotective activities. 7 C viscosa is currently widely distributed in farms in the east of Golestan province, Iran, and every year the biomass produced by them is destroyed by weeding and other agricultural activities. However, there are few reports on the antioxidant properties of the plant, and a detailed and comprehensive characterization of secondary metabolites by LC-ECI-MS has not been reported so far. Therefore, this study primarily aimed at the analysis and characterization of the metabolite profiles of the leaf and root extracts of C viscosa by LC-ESI/MS analysis. Also, the antioxidant activity and content of associated secondary metabolites in the extract of different parts of C viscosa were measured.
Materials and Methods
Plant Collection and Sample Preparation
C viscosa was collected from farms in Golestan province (northeast of Iran) in the 2020 crop year. Plant authentication was made using the colored flora of Iran 8 with the assistance of a systematic expert at Gonbad Kavous University. A herbarium specimen of C viscosa, with the herbarium code No. GKU/803897, was registered in the herbarium of Gonbad Kavous University.
Preparation of Plant Extract
The collected plant samples of C viscosa were divided into root, stem, leaf, and flower, cleaned with distilled water to remove soil and dust particles, and then dried in an oven at 60°C until they reached a fixed weight. The dried plant samples were first crushed in a mill and then passed through a sieve with a number 8 mesh. Finally, the powdered samples were stored in plastic bags until required. About 100 mg of powdered plant material was placed into 100 mL of different solvents, such as ethanol and methanol. Then the extracts were filtered using a refrigerated centrifuged at 1000 rpm for 10 min and used for phytochemical analysis.
Chemicals and Equipment
All chemicals and reagents used in this study were of analytical grade and purchased from Sigma Chemicals Company. The chemicals and reagents used included ethanol, gallic acid, Folin-Ciocalteau reagent, sodium carbonate, aluminum chloride, quercetin, sodium acetate, 2,2-diphenyl-1-picryl-hydrazyl-hydrate (DPPH), and methanol. The equipment used included a boiling water bath (Model WNB10) from Memmert Company, a drying oven (model ED 115) from Binder GmbH, Germany, a refrigerated centrifuge (model Universal 320 R) from Hettich-UK Company, and a Biochrom Libra S22 UV/visible Spectrophotometer.
Qualitative Analysis of the Chemical Composition Using LC-ESI-MS
LC-ECI-MS analyses were carried out using an HPLC system (Shimadzu, Tokyo, Japan) equipped with an AB SCIEX 3200 and a model ionization source from ESI Turbo V. A C18 reversed-phase column (Sigma-Aldrich, USA, SUPELCO Analytical HS-C18,4.6 × 150 mm, 3 μm) was used for separation. A gradient of water and methanol was applied from 20% to 100% methanol over 25 min with a flow rate of 0.5 mL/min. The MS was operated in positive ion mode with a capillary voltage of −10 V, a source temperature of 200°C, and nitrogen as an auxiliary gas. Ions were identified in a mass range of 100 to 1000 m/z. The LC-ECI-MS data were processed using MZmine analysis software, version 2.3. 9
Quantitative Analysis of the Chemical Constitutents in the Extract of Different Parts of C viscosa
Quantification of individual compounds was made from a calibration curve of the standard compound. 10
Determination of Total Phenolic Content
The total phenolic content of the extract was determined by the Folin–Ciocalteu method.
11
For this purpose, 2.5 mL of ethanol (80%) was added to 0.25 g of dried sample and centrifuged at 2°C for 10 min. The obtained supernatant was then preserved. The sample was re-extracted with 2.5 mL of ethanol (80%) and then centrifuged. The pooled supernatant was first evaporated and 3 mL of distilled water was added to the dry residue. Folin-Ciocalteu reagent (0.5 mL) and 2 mL of sodium carbonate (20%) were added to it and the mixture was kept in a boiling water bath for 1 min. The absorbance was measured at 650 nm with a UV/visible Spectrophotometer. The total phenolic content was calculated as mg GAE /100 g dry sample using the following equation based on the calibration curve of gallic acid:
Determination of Total Flavonoids Content
Total flavonoid contents in the various plant extracts were determined by the aluminum chloride colorimetric method.12,13 Stock quercetin solution was prepared by dissolving 5 mg quercetin in 1 mL methanol, then standard solutions were prepared by serial dilutions with methanol (5-200 μg/mL). Either 0.6 mL diluted standard quercetin solution or the extract was separately mixed with 0.6 mL of 2% aluminum chloride. After mixing, the solution was incubated for 60 min at room temperature. The absorbance of the reaction mixtures was measured at 420 nm with a UV/visible Spectrophotometer The content of total flavonoids in the test samples was calculated from the following calibration curve and expressed as mg quercetin equivalent per g dry weight of sample (mg QE/g).
Determining Total Flavonols Content
The total flavonols content of the extract of each plant part was determined using previously method.14,15 AlCl3 (2%)/ethanol (2 mL) and 3 mL (50 g/L) sodium acetate were added to 2 mL of the plant extract. The mixture was shaken and incubated at 20°C for 2.5 h. The absorbance of the reaction mixtures was measured at 440 nm with a UV/visible Spectrophotometer. Total flavonols content was calculated from the following calibration curve and expressed as mg quercetin equivalents per g dry weight of the sample (mg QE/g).
Determination of the Antioxidant Activity
The antioxidant activity was determined using the 2,2-diphenyl-1-picryl-hydrazyl-hydrate (DPPH) method as a free radical.
15
In order to conduct this experiment, 3.9 mL of DPPH made of stock solution (0.0004 g DPPH in 100 mL methanol) was poured into a tube and then 0.1 mL of methanol extract of each plant part was added. The mixture was kept in the dark for 30 min and then the absorbance of the solution was measured at 517 nm with a UV/visible Spectrophotometer. Radical inhibition of DPPH was calculated using the following equation.
16
Statistical Analysis
The data of secondary metabolites and antioxidant activity of the plant parts were analyzed using SAS statistical software, version 9.3, 17 and the means were compared using the Least Significant Difference (LSD) test at a 5% probability level.
Results
The Chemical Composition of Hydroalcoholic Extracts of the Leaf and Root of C viscosa
LC- ESI-MS was used to separate and characterize the chemical composition of the extracts of root and leaf of C viscosa. Comparison of the MS data showed 22 and 21 compounds in the extracts of the leaf and root samples, respectively (Table 1). Different types of compounds such as phenols, flavonoids and terpenes were identified in the extracts of both leaf and root of C viscosa. Major groups of phenolic compounds found in the leaf and root of C viscosa were coumarinolignoid, methylated flavonoids, flavone glycosides and flavonol glycosides. However, the major compounds identified in the hydroalcoholic extract of the leaf included cleomeolide, 5,7,4′.trihydroxy-6,3.5-trimethoxyflavone, astragalin, kaempferitrin, kaempferol 3-O-(4-O-acetyl)-α-l-rhamnopyranoside, nevirapine and (1 R*,3 E,7 Z, 12 R*)-20-hydroxycembra-3,7,15-trien1 to 9 -oic acid at m/z 318, 360, 448, 578, 474, 267 and 320, respectively. In the root extract, the major compounds were nevirapine, vincetoxicoside A, cleomeolide, quercetin 3-O-(2″-acetyl)-glucosid, (3 E,7 Z, 11 Z)-17,20-dihydroxycembra-3,7,11,15-tetraenoic acid, chrysosplenetin and cleomiscosin A or B at m/z 267, 610, 318, 302, 334, 374 and 387, respectively.
Chemical Composition of Hydroalcoholic Extracts From the Leaf and Root of C. viscosa.

The total HPLC-ESI-MS analyses of C viscosa leaf and root extracts are shown in Figure 1.

The HPLC- ESI-MS analysis of C viscosa Extracts. (a) Total Chromatogram of Leaf Extract; (b) Total Chromatogram of Root Extract.
Quantitative Analysis of the Chemical Constituents of the Extract of Different Parts of C viscosa at Full Flowering Stage
Total Phenolic Content
Total phenolic content of the extracts from different plant parts of C viscosa at full flowering stage are presented in Table 2. According to the results, ethanolic extracts obtained from all parts of the plant were varied in terms of total phenolic content. Ethanolic extract of the leaf had the highest total phenolic content, with a value of 13.10 ± 0.53 mg GAE/100 g dry weight of the sample. The lowest total amount of phenolic compounds was observed in the root extract (7.01 ± 0.55 mg GAE/100 g dry weight of sample).
Quantification of Total Phenols, Flavonoids and Antioxidant Activity of the Extracts of Different Parts of C viscosa at Full Flowering Stage.

Data with different letters in the same column indicate mean ± standard error of three replications and groups based on the least significant difference (LSD) comparisons with 95% confidence level.
Total Flavonoids Content
As shown in Table 2, the highest flavonoids content was observed in the leaf extract of C villosa (2.57 ± 1.03 mg QE/g dry weight of the sample), whereas the lowest amount was observed in the root extract, with a value of 1.18 ± 0.06 mg QE/g dry weight of sample.
Total Flavonols Content
Total flavonols content in ethanol extracts from different plant parts of C viscosa ranged from 1.05 ± 0.05 to 0.20 ± 0.03 mg QE/g dry weight of the sample. The greatest content was recorded in the flower extract, followed by the leaf (1.03 ± 0.09 mg QE/g dry weight of the sample) and the lowest was found in the root extract (Table 2).
Antioxidant Activity
The antioxidant activity of C viscosa was dependent on the plant part. For methanol extracts of different plant parts, the leaf extract had the greatest antioxidant activity (50.3%) for free radical scavenging, followed by the flower (35.2%). Compared to other parts of C viscosa, the root extract had the lowest radical scavenging activity, with a value of 11.3% (Table 2).
Discussion
Different types of bioactive compounds such as phenols, flavonoids and terpenes in the extract of both leaf and root of C viscosa were identified by HPLC-ESI-MS analysis. Cleomeolide and 5,7,4′.trihydroxy-6,3.5-trimethoxyflavone, in the leaf extract, and nevirapine and vincetoxicoside A, in the root extract, were considered as the main chemical compounds. In this study, compounds such as 5-hydroxy-3,6,7,3′,4,5′-hexamethoxyflavone, kaempferol 7-O-α-l-rhamnopyranoside and quercetin 3-O-(2″-acetyl)-glucoside in leaf extract, and 3 E,7 Z, 11 Z-17,20-dihydroxycembra-3,7,11,15-tetraen 1n9 oic acid and vincetoxicoside A, in root extract, have been detected. For the first time, our results showed that compounds with a m/z of 267, 387, 416 and 506, which were reported in previous studies of the seed and flower of C viscosa, were also identified in the leaf and root extracts.16,18,22,24 Some compounds with molecular masses of 300, 344, 360, 374, 404 and 418, which were found only in the leaf and stem of some species such as C droserifolia, C amplyocarpa, C brachycarpa and C chrysantha, were also detected in the root and leaf of C viscosa under study. 27 Also, compounds with m/z values of 318, 448, 474, 478, 578, 594, 611, and 756 were characterized in both root and leaf extracts.20,24,26 According to the HPLC- ESI-MS analysis, it can be concluded that C viscosa leaf had approximately the same chemical composition as that of the root.
According to the results, different parts of C viscosa were also varied in terms of total phenols, flavonoids and flavonols. Analysis has revealed that the Cleome genus is a rich source of phenolic acids, flavonoids, alkaloids, terpenoids, fatty acids, coumarino-lignan, nutrients, especially vitamins A and C, protein, gallotannins, saponins, iridoids, hexacosanol, and kaempferol.27-32 Compared to other parts, the leaf extract of C viscosa contained the highest amounts of these compounds. This is not unexpected because leaves are the most important source of photosynthesis. These results are in agreement with previous studies 33 which reported that a wide range of compounds including alkaloid, flavonoid, phenol, saponins, tannin and hydrogen cyanide, as well as proteins, fats and carbohydrate were found in different parts of C viscosa. The highest percentage of flavonoids (0.63%), saponins (0.44%), tannins (0.35%), proteins (11.84%) and fats (2.16%) were observed in the leaf extract.
In general, total phenols, flavonoids and flavonols in different parts of C viscosa, especially the leaves, were significantly high; these compounds are very important due to their radical scavenging properties 34 and could be responsible for the strong antioxidant activity of the C viscosa extracts. In this study, the highest antioxidant activity was related to C viscosa leaf extract. The findings of this study are in agreement with the results of other researchers 35 who reported that the strong free radical scavenging activity exhibited by C viscosa leaf alcoholic extracts could be attributed to phenols and flavonoids. These researchers reported that free radical scavenging can be due to their electron transfer or hydrogen donating ability. In another study on the antioxidant activity of two species of Cleome, it was reported that methanol extracts of C viscosa and C burmanni had strong antioxidant activities against different reactive oxygen species (ROS). 36 It was also reported that different extracts from various parts of C coluteoides had different antioxidant and antimicrobial effects. The methanol extract of the leaf and the ethyl acetate extract of flowers had the highest antioxidant and antimicrobial activities, respectively, which are probably related to the total phenolics content. 37
Conclusion
HPLC-ESI- MS results showed that the extracts of root and leaf parts of C viscosa contain a wide range of polar and non-polar chemical compounds. In this study, the compounds with molecular masses of 267, 387, 416 and 506 have been reported in the root and leaves of C viscosa for the first time. Also, the antioxidant activity of C viscosa against free radical scavenging was dependent on the plant parts, which were significantly correlated with the content of total phenols, flavonoids and flavonols. In general, due to the suitable amounts of secondary metabolites such as total phenols and flavonoids in different parts of C viscosa, especially in the leaf, and their free radical scavenging activity, exploitation is suggested of the huge biomass generated by these luxuriantly growing weeds as a natural antioxidant and medicine, edible plant and bio-herbicide in sustainable agriculture.
Footnotes
Acknowledgments
The authors are grateful to the expert at the Herbarium of Gonbad Kavoos University for identification of the sample. In addition, the authors express special gratitude to experts at Mashhad University of Medical Sciences for their technical assistance.
Author Contributions
Conception or design of the study by Ebrahim Gholamalipour Alamdari, Zinab Avarseji and Akram Taleghani. Ebrahim Gholamalipour Alamdari performed the experiments and analyzed the data. All authors have read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.