Abstract
Gastrodin (GAS) and its aglycone,
Gastrodin (GAS) is a major bioactive ingredient in the Traditional Chinese herbal medicine
Following either oral or intravascular administration, GAS is metabolized to HBA.8,9 Both compounds can traverse the blood–brain barrier to act on the central nervous system.10,11 Though many past pharmacokinetic studies have investigated the effects of GAS and GEB extracts, very few focus on HBA. 12 Some studies report that the GAS plasma concentration–time curve has 2 peaks, and speculate that there is also a presence of enterohepatic circulation.13,14 This phenomenon might be further explained by the in vivo biotransformation of HBA to GAS.
In this study, a high-performance liquid chromatography-diode array detector (HPLC-DAD) method was used to measure the plasma concentrations of GAS and HBA, following oral ingestion, in rats. The sedative effect of these compounds was then analyzed at different administration durations in larval zebrafish (
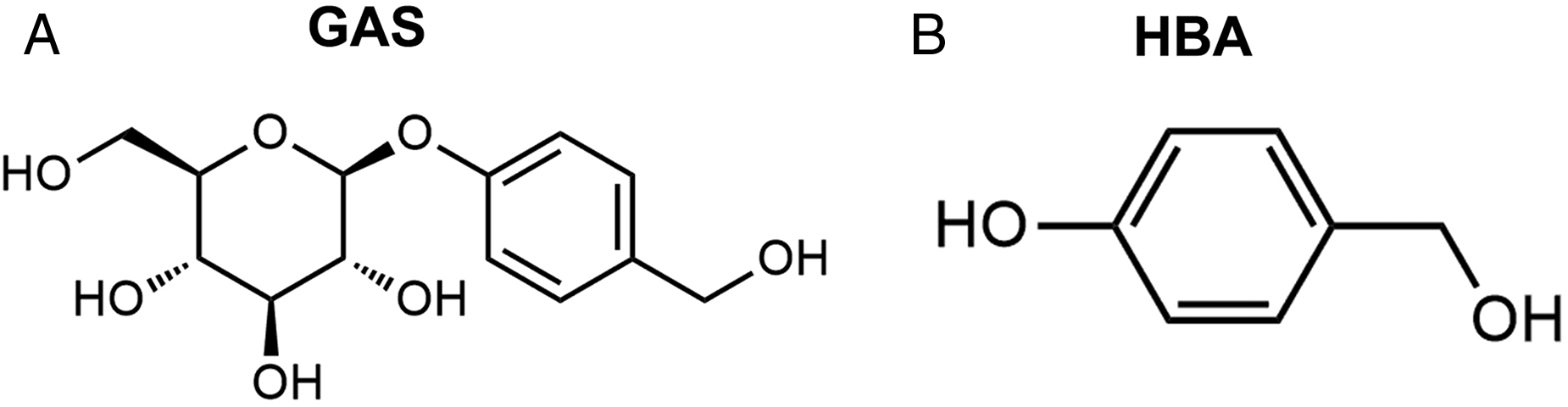
The structural formulae of GAS and HBA.
Results
Method Development
We developed an HPLC-DAD method for the accurate determination of HBA and GAS. Forty-five min after either GAS or HBA administration, typical chromatograms of blank plasma, spiked samples with standards (GAS 80 μg/mL, HBA 40 μg/mL), and plasma samples were prepared (Figure 2). The HPLC-DAD method showed no significant matrix interference on the targeted peaks. The retention times of GAS and HBA were 11.60 and 19.33 min, respectively.

Representative chromatograms of GAS and HBA. GAS standard (A), HBA standard (B), blank plasma sample (C), blank plasma samples spiked with analytes (D), plasma samples obtained from 45 min after oral administration with GAS (E) and HBA (F) at 105 mg/kg.
The calibration curves showed a high linearity (
Accuracy and Precision of the Method in Analyzing GAS and HBA.

Abbreviations: GAS, gastrodin; HBA,
The results of the stability test are shown in Table 2 and indicate that both GAS and HBA were stable, with a relative standard deviation (RSD) lower than 15%. In summary, the present analytical method is reliable and reproducible for GAS and HBA quantitation.
Stability of GAS and HBA in Rat Plasma Under Different Conditions.
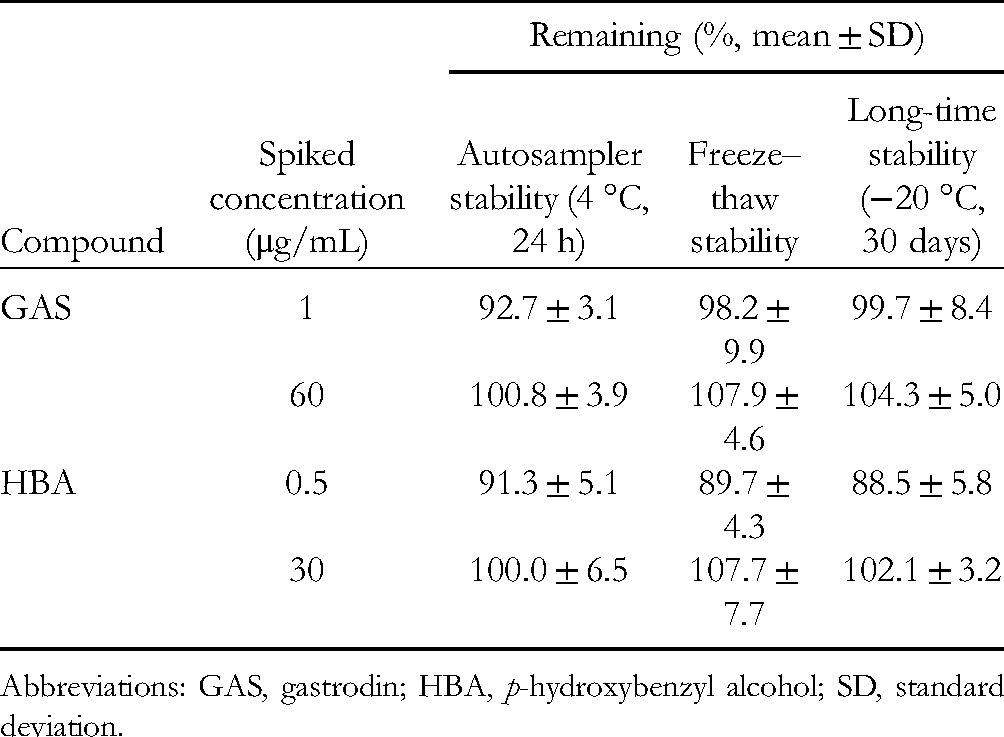
Abbreviations: GAS, gastrodin; HBA,
Biotransformation of HBA to GAS In Vivo
The GAS and HBA plasma concentration–time curves, following administration of either GAS or HBA in rats, are shown in Figure 3. As previously reported,8,15 GAS quickly enters the bloodstream where it is transformed into small amounts of HBA, which is rapidly absorbed and cleared, thereby producing a smaller area under curve (AUC) compared with GAS. Interestingly, GAS was seen to have formed and reached its peak about 45 min after administration. Furthermore, GAS was detected in the plasma samples from HBA-treated rats by liquid chromatography–tandem mass spectroscopy (LC–MS), which confirms the transformation of HBA to GAS.

Plasma concentration–time curves and metabolite identification. Plasma concentrations of GAS and HBA following single oral administration of GAS (A) or HBA (B) in rats. Each point represents mean plasma concentration ± SD for 7 animals. And representative extracted ion chromatograms of HBA (C), GAS standard (D), and plasma sample (E).
Effect of GAS and HBA on Locomotor Activity
The open-field test (OFT) is commonly used to assess the locomotor activities and sedative effects of various compounds. 16 The effect of GAS and HBA on zebrafish larvae motion was expressed as the change in mean velocity within each 10 min interval versus the baseline velocity. As shown in Figure 4, GAS and HBA treatment resulted in a decrease in the velocity of zebrafish larvae when compared with the control treatment. Additionally, the onset of the sedative effects caused by GAS occurred earlier than those of the HBA treatment. However, the maximal and overall sedative effects were similar in both.

Comparisons of the sedative effects of GAS and HBA in zebrafish larvae. (A) Representative heatmap showing the trajectory and amount of motion of zebrafish larvae in OFT at different time periods. (B) Total distance moved by the zebrafish larvae in 60 min. (C) Change in mean velocity within each 10-min interval versus baseline velocity. Data were expressed as mean ± SD with 3 zebrafish larvae per group.
Discussion
In the modern Chinese pharmacopoeia, GAS and HBA are both used as quality control markers for GEB. 17 They exert similar pharmacological effects and are regarded as major active ingredients in GEB and other formulae. 18 The majority of previous studies have explored the mechanisms and new indications for GAS and HBA, but have ignored the timing of their effects. In this study then, we compared their sedative effects in zebrafish larvae after administration. Given that GEB possesses sedative effects, both GAS and HBA also caused time-dependent sedative effects (Figure 4B). Notably, however, the onsets of the sedative effects were different between the GAS- and HBA-treated groups, demonstrating that the 2 compounds have different pharmacokinetic properties.
Few studies have compared the pharmacokinetic properties of GAS and HBA following oral administration. GAS, which is composed of HBA and glucose, is considered to be the more hydrophilic of the 2, with poorer oral absorption properties when compared with HBA. However, the presence of the glucose transporters (GLTs) in the intestines have been shown to facilitate the oral absorption of GAS. 19 In our study, GAS absorption was just as fast as HBA absorption, while, when administered at the same dose, plasma concentrations of HBA were generally lower than those of GAS (Figure 3). This could be attributed to HBA's larger plasma protein-binding rate. 20 Metabolism, also referred to as biotransformation, is a vital and complex process in which drugs are broken into various metabolites for easier excretion or optimal pharmacological activity. HBA, formed from the hydrolysis of GAS, is further either conjugated with glucuronic acid, sulfuric acid and glycine, or oxidized to aldehydes and carboxylic acids. 12 In this study, the transformation of HBA to GAS was discovered in rats for the first time. Glucosidation is uncommon during drug disposition, and it is a critical step for glycoside biosynthesis following the catalysis mediated by uridine diphosphate-glycosyltransferases (UGTs). 21 Several UGT subtypes which catalyze the glucuronidation reactions of endogenous or exogenous substances are expressed in both animals and humans. 22 The mechanisms through which HBA is transformed to GAS in vivo still require further investigation, however.
The present results indicate that differences between the effects of GAS and HBA might be due to the formation of GAS from HBA. However, whether the low transformation rate is sufficient to influence the HBA treatment outcomes has not been established yet. Further comparative pharmacodynamic experiments based on short-term administration of GAS and HBA in rodents are, therefore, needed to verify the universality of the phenomenon observed in our study.
GAS and HBA exert similar effects, but their different pharmacokinetic characteristics and in vivo transformation may lead to different onsets of their effects. Therefore, the total concentrations and proportions of GAS and HBA used over the course of GEB treatment, can affect its curative effect and therapeutic efficacy, especially when only employing a single-dose administration. The contents of GAS and HBA in GEB vary depending on the processing method applied. 23 Therefore, in order to enhance GEB's therapeutic effect, further studies will need to determine the optimal ratios and most effective processing methods.
Intestinal flora plays an important role in many metabolic processes, such as in sugar and lipid metabolism.24,25 The absorption of glycosides, such as baicalin and eniposide, require gut microbes.26,27 GAS is a glycoside, which is metabolized in the body by gut bacteria to produce other metabolites, including
Conclusion
Using the simple HPLC-DAD method, we determined the plasma concentration levels of HBA and GAS in rats. GAS was only detected in the rat's plasma following oral administration of HBA, which indicates that HBA can be bio-transformed into GAS in vivo. This glycosylation process is rare in drug disposition and, therefore, the location, mechanism and significance of this biotransformation deserve further investigation. The present results also reveal that HBA and GAS exert sedative effects, but at different onset times, in zebrafish larvae. This implies that they possess distinct pharmacokinetic characteristics, with the transformation of HBA to GAS perhaps being a reason for this.
Methods and Materials
Chemicals and Regents
GAS and HBA were purchased from the National Institutes for Food and Drug Control and Chengdu Push Bio-technology Co., Ltd, respectively. The purity of the 2 compounds was above 99%. HPLC grade methanol was obtained from Tedia company, and the water for the HPLC was prepared by a Millipore Milli-Q purification system (Millipore). MS grade acetonitrile was obtained from Merck, phosphoric acid from Sinopharm Chemical Reagent Co., Ltd, and formic acid from Agilent.
Animals, Treatment, and Sample Collection
Adult male Sprague–Dawley rats (200 ± 20 g) were purchased from the Shanghai Laboratory Animal Co. (SLAC), Ltd. Prior to drug administration, the rats were made to fast for a night, but with free access to water. The rats were divided into 2 groups, with one receiving oral administration of GAS (105 mg/kg), and the other receiving oral administration of HBA (105 mg/kg). Blood samples were collected in heparinized tubes from inner canthus vein plexuses at 0, 0.083, 0.25, 0.5, 0.75, 1, 1.5, 2, 4, 8, and 12 h after drug administration. Blood samples were centrifuged at 3000 rpm for 10 min and the supernatant was separated. The plasma samples were stored at −80 °C until analyzed.
The zebrafish embryos were generated through the natural spawning of parent wild-type AB-line adult zebrafish. On the seventh day post-fertilization, the zebrafish larvae without any maldevelopments were selected and used for the experiments. The grouping and treatment scheme will be described later. All the animal experiments were approved by the Science and Technology Department of Jiangsu Province and followed Jiangsu Province's standard ethical guidelines for the use of experimental animals.
Preparation of Analytical Sample
Precooled methanol (800 μL) was added to 200 μL plasma samples in an EP tube. After vortexing for 1 min, the mixture was centrifuged at 14 000 rpm for 15 min. The supernatant (800 μL) was then transferred into a new EP tube and left to evaporate by vacuum at 45 °C until dry. The residue was reconstituted in 20 μL of initial mobile phase and centrifuged again. The supernatant was then taken for HPLC analysis.
Analytical Conditions
An Agilent 1260 system equipped with a Zorbax SB – C18 column (4.6 mm × 250 mm, 5 μm, Agilent) was used for the analysis of GAS and HBA. The mobile phase consisted of 0.1% phosphoric acid in water (A) and methanol (B). The elution gradient was set as follows: mobile phase B was started at 7%, increased to 8% within 10 min, increased to 12% after the next 5 min and then kept for 6 min, then it was again increased to 40% within 4 min (then returned to the beginning). The flow rate was maintained at 0.8 mL/min. The column and autosampler temperatures were respectively maintained at 25 °C and 4 °C during analysis. The injection volume was 5 μL. The wavelength of the detector was set at 220 nm.
Analytical Validation
The validation of the analytical method was conducted in line with FDA guidelines. 28 Accurately weighed GAS and HBA were dissolved in methanol–water (50:50, v/v) until achieving the final concentrations of 1680 and 840 μg/mL, respectively. GAS and HBA working solutions were prepared through successive dilutions with the same solvent. About 10 μL of the working solution was added to 200 μL blank rat plasma in order to prepare the calibration standard samples of GAS (0.1-80 μg/mL) and HBA (0.05-40 μg/mL). The quality control (QC) samples, which comprised 4 different GAS concentrations (0.1, 1, 40, and 60 μg/mL) and 4 different HBA concentrations (0.05, 0.5, 20, and 30 μg/mL), were prepared similarly to the above. Before injection, the samples were de-proteinized, as described above. Linearity was assessed by a linear regression analysis of the peak area versus the analyte concentration of the samples. Accuracy and precision (intra and interbatch) were calculated using 4 QCs over 3 consecutive days, which were respectively expressed as the RE and RSD. Stability studies of QCs were based on the following 3 operations: (a) stored at 4 °C for 24 h; (b) stored at −20 °C for 30 days; and (c) underwent 3 freeze–thaw cycles (−20 °C to 25 °C).
Liquid Chromatography–Tandem Mass Spectroscopy
A further identification of HBA metabolites was performed using the HPLC system coupled with a 6530 quadrupole time-of-flight (Q-TOF) MS (Agilent). Separation was achieved on an XSelect HSS T3 column (2.1 × 100 mm, 2.5 μm, Waters) at 40 °C with a pump flow rate of 0.4 mL/min. The mobile phase system consisted of 0.1% formic acid aqueous solution (A) and acetonitrile (B). The gradient elution program was as follows: 0 to 2 min, 2% mobile phase B; 2 to 7 min, 2% to 60%; 7 to 20 min, 60% to 100%; and 20 to 23 min, 100%.
The electrospray ionization (ESI) source was set to its positive mode. The ion data were collected between 50 to 1000
Behavior Test
To compare the GAS and HBA effects on motion, an OFT was performed on the zebrafish larvae. The apparatus used for the OFT was a white plastic dish with a diameter of 3.6 cm. The zebrafish larvae were divided into 3 groups, and gently transferred to their respective dishes with 4 mL water containing either GAS (10 mM) or HBA (10 mM) or being drug-free. One zebrafish was added per dish, and each of their tracks was respectively recorded and analyzed through Ethovision XT 8.5 software. The duration of each recording lasted for 1 h, and all the behavioral tests were made in a quiet environment within the time period of 9:00 to 14:00.
Footnotes
Acknowledgments
We sincerely thank Dr Ma (China Pharmaceutical University) and Dr Yang (Zhaotong University) who provided kind support in this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific research fund of Yunnan Education Department (grant number 2020J0743), and the University Science and Technology Innovation Team Project of Yunnan Province (7th batch).
Ethical Approval
The study was approved by the Ethical Committee of China Pharmaceutical University, Nanjing University, and Laboratory Animal Management Committee of Jiangsu Province (SYXK SU 2018-0019, 14 May 2018).
