Abstract
Thermogenesis-mediated energy expenditure is a promising strategy to combat obesity. Aframomum melegueta commonly known as grains of paradise (GP) is a popular spice with medicinal attributes in promoting health. We have demonstrated the thermogenic effects of a standardized A melegueta seed extract (AMSE) containing not <10% 6-paradol in high fat diet-fed (HFD) mice. The 6-week oral ingestion of 20 and 40 mg/kg AMSE significantly limited the weight gain, improved the brown adipose tissue (BAT) activity in HFD mice. Interestingly, AMSE markedly induced the beige adipocytes in epididymal white adipose tissue (eWAT). AMSE treatment led to the upregulation of marker proteins i.e., uncoupling protein 1 (UCP1), peroxisome proliferator-activated receptor-gamma-coactivator 1-alpha (PGC-1α), and peroxisome proliferator-activated receptor gamma (PPARγ) in eWAT and BAT. Our findings add to the current understanding of the thermogenic potentials of GP seed extract and report that the extract can stimulate the browning of WATs in addition to enhanced BAT activity. AMSE requires clinical validation to be explored as a dietary supplement/functional ingredient with thermogenic effect in food and beverages.
Introduction
Obesity and type 2 diabetes present a serious health concern globally. An imbalance in the energy intake and expenditure results in weight gain and obesity. The excess weight is stored in the form of lipids in white adipose tissue (WAT). In addition to these fat-storing cells, there also exist brown adipocytes that can transform energy into heat, a process called as thermogenesis. Activation of thermogenesis by brown adipose tissue (BAT) can substantially contribute to weight loss and glucose metabolism.1,2 These adipocytes have densely populated mitochondria containing uncoupling protein 1 (UCP1), which plays a pivotal role in thermogenesis. 3 Apart from the classical BAT and WAT, there exists a third metabolically distinct cell type called beige adipocytes resulting from the browning of WAT in response to specific stimuli.4,5 The BAT and beige cells increase the whole-body metabolism and energy expenditure via thermogenesis. 6 The activation of thermogenic adipocytes leads to body weight reduction and improved metabolism in animal models.7,8
Dietary intake of natural compounds triggers thermogenic responses in experimental models as well as humans. 9 Phytochemicals including flavonoids and nonflavonoids promote nonshivering thermogenesis thereby increasing energy expenditure.10,11 During the past decade, researchers have explored the thermogenic potentials of plant compounds. Previously, plant-based small molecules such as berberine, butein, capsaicin, 7,8-dihydroxyflavone and fucoxanthin have been reported to increase energy expenditure via stimulation of BAT activity.12-16 Resveratrol, a dietary polyphenol, is reported to exert thermogenic effect via the activation of Sirtuin 1 (SIRT1) and PR domain-containing protein 16 (PRDM16). 17 More recently, Liu et al 18 demonstrated the involvement of the mechanistic target of rapamycin protein (mTOR) pathway in resveratrol-induced WAT browning of 3T3-L1 adipocytes. Curcumin is another polyphenol having the ability to induce a thermogenic program via activation of peroxisome proliferator-activated receptor gamma (PPARγ) and AMP-activated protein kinase (AMPK). 19 There are reports suggesting the potential role of green tea catechins in increasing the energy expenditure by upregulating UCP-1 expression and thus enhancing BAT activity. 20
Aframomum melegueta (Fam. Zingiberaceae) also known as grains of paradise (GP) is a native of West Africa. GP extracts have a pungent taste due to the presence of 6-paradol, an unsaturated nonvolatile aromatic ketone. 21 Other pungent compounds include 6-shogaol and 6-gingerol. GP seeds are used to relieve pain and inflammation in ethnomedicine. Several experimental studies have reported the potential anti-inflammatory and analgesic activities in rats.22,23 Further, 6-paradol and 6-shogaol, the active principles in GP seeds have been reported to possess antihyperglycemic activity in high-fat-diet-fed mice. 24 The antihyperlipidemic activity of GP extract in hyperlipidemia model rats has been demonstrated. 25 It has been reported that the GP extracts exert thermogenic effects in rodent models. Hattori et al 26 previously studied the antiobesity effects of GP seed extract in a high-fat diet (HFD)-induced mice model. In another study, 6-gingerol, one of the active constituents in GP seeds, has been shown to have profound antiobesity activity in HFD mice. 27 As studied by Iwami et al, 28 in rats the thermogenic effect of GP extract and its active constituent 6-paradol could be a function of sympathetic nerve activity stimulation. Further an orally ingested GP seed extract was evaluated for activation of BAT activity and whole-body energy expenditure in humans. 29 However, there are no reports on the mechanism of GP extract-mediated activation of thermogenic adipocytes. Here, we have examined the effect of a standardized A melegueta seed extract (AMSE) containing not less than 10% of 6-paradol, on the regulation of transcription factors of thermogenesis in mice. In accordance with the previous literature the HFD-induced male C57Bl/6 mice were chosen as the animal model to evaluate the thermogenic effect of AMSE. 30 Our results demonstrate that GP seed extract induces browning of WAT in addition to activation of BAT.
Results
High-performance Liquid Chromatography Analysis of AMSE
Figure 1 shows the high-performance liquid chromatography (HPLC) chromatograms of major active constituents in AMSE. HPLC analysis revealed the presence of 14.73% 6-paradol in AMSE. The chromatogram was confirmed using retention time values of reference standards.
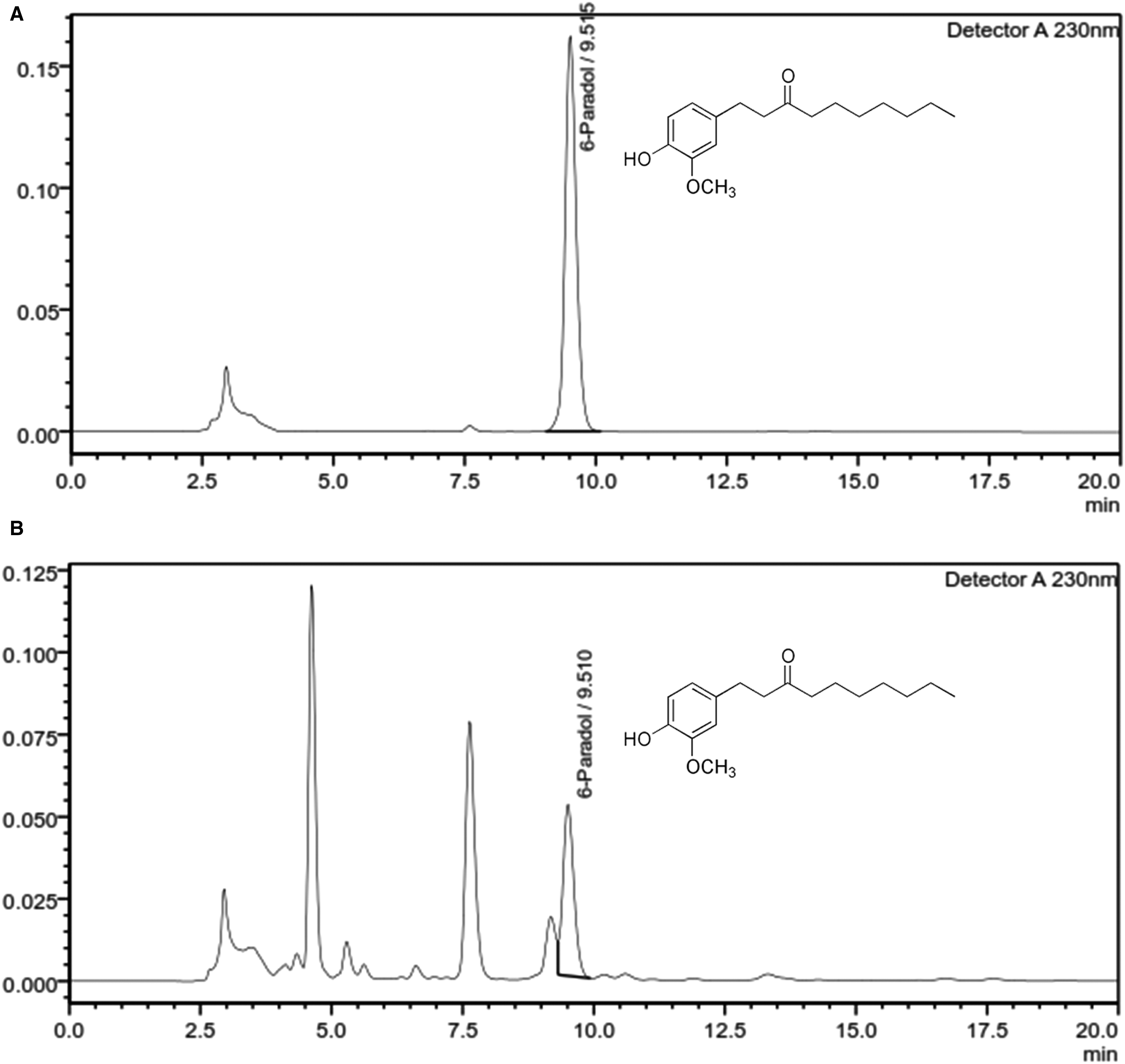
HPLC chromatogram of AMSE. 6-paradol in AMSE was quantitatively analyzed at UV detection of 230 nm using the mobile phase of a mixture of orthophosphoric acid (0.2%): acetonitrile (40:60) with isocratic flow rate of 1.2 mL/min and the injection volume of 5 µL.
AMSE Attenuates HFD-induced Weight Gain in Mice
The initial body weight of animals in all the groups was similar at the start of the experiment. Eight-week consumption of HFD resulted in marked increase in the body weight of animals as compared to the control group. The untreated HFD group mice gained significant weight up to 14 weeks (P < .001). A 6-week administration of AMSE dose-dependently reduced the body weight of HFD mice (Figure 2A). The increase in body weight with respect to the control group was 39.5%, 26.05% and 7.8% for HFD, AMSE20, and AMSE40 groups, respectively. There was a significant reduction in the mean body weight of a high-dose group (AMSE40) after 1 week of treatment and consistently decreased thereafter till the end of treatment (P < .001) compared to the untreated HFD group. The AMSE-treated groups showed significant reduction in mean body weight change from the baseline as compared to the HFD group (Figure 2B).

AMSE reduces body weight of HFD-fed C57Bl/6 mice.
AMSE Ameliorates HFD-induced Liver Weight, Histopathology and Liver Function Parameters
As seen in the representative images of histology, the untreated HFD mice showed the presence of macrovesicles and lipid droplets compared to the control group with a normal liver architecture (Figure 3A). Low- and high-dose treatment with AMSE restored the normal liver histology in HFD mice. Oil Red O staining showed considerable fat accumulation in the liver section of the HFD group. AMSE treatment markedly reduced the hepatic lipid accumulation.

AMSE lowers the HFD-induced lipid accumulation in mice. (A) Representative images of liver histological examination by H&E staining and Oil Red O staining.
Figure 3B shows the effect of AMSE administration on the liver weight, histology and serum aspartate aminotransferase (AST) and alanine transaminase (ALT) activities. The HFD mice exhibited a significant increase in the liver weight compared to the normal diet-fed animals (P < .05). A 6-week administration of AMSE at 20 and 40 mg/kg decreased the liver weight of HFD mice in a dose-dependent manner compared to the untreated HFD-fed mice.
The HFD mice showed significant increase in the serum levels of AST (42.2%) and ALT (50.4%) as compared to control (P < .01). Treatment with low and high doses of AMSE showed a dose-dependent reduction in the levels of liver function parameters. The AMSE20 group showed a reduction of 18.76% and 26.44% in serum AST and ALT levels, respectively, compared with that of the HFD group. The AMSE40 group exhibited significant decrease of 31.05% and 31.03%, respectively, in AST (P < .05) and ALT activities (P < .01) as compared to untreated HFD mice.
AMSE Significantly Improves the Serum Lipid Levels in HFD Mice
The untreated HFD mice showed significantly higher levels of total cholesterol and low density lipoprotein-c (LDL-c) (P < .001) compared to the control group. The HFD group also showed marked increase in triglycerides (TG) and reduction in high-density lipoprotein-c (HDL-c). However, low- and high-dose treatment groups (AMSE20 and AMSE40) significantly reduced the TC and LDL-c (P < .01) as compared to the untreated HFD group. The AMSE treated groups dose-dependently exhibited improvement in the TG and HDL-c levels (Figure 4).

AMSE improves the serum lipid profile in HFD mice. Values represent mean ± SD. ###P < .001 versus control group; **P < .01 and ***P < .001 versus HFD group.
AMSE Markedly Enhances the BAT Weight and Decreases WAT in HFD Mice
In the present study, there was a dose-dependent increase in BAT weight following treatment with AMSE. In the AMSE40 treatment group, the mean weight was significantly higher compared to the untreated HFD group (P < .01) (Figure 5A). As shown in Figure 5B, the mean weights of mesenteric and epididymal WAT in the HFD group were significantly increased compared to the control group (P < .001). AMSE20 and AMSE40 groups dose-dependently reduced the WAT weight of HFD mice. Consistent with reduced WAT mass, the HFD mice by AMSE treatment had significantly smaller adipocyte size compared to untreated HFD group (P < .001) (Figure 5C). These results correlate with the reduction in body weight of animals treated with respective doses of AMSE.

AMSE influences fat depots in HFD mice. (a) Brown adipose tissue weight,
AMSE Triggers the Expression of Markers of BAT Activity
Western blot analysis of BAT revealed that AMSE treatment induced the expression of key proteins activating a thermogenic program (Figure 6). The expression of UCP-1, a classical marker of BAT thermogenesis, was significantly increased in AMSE20 and AMSE40 groups compared to the HFD group (P < .01). The other transcription factors such as peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) (P < .001) and PPARγ (P < .01) were strongly activated in low- and high-dose AMSE treatment groups. These results suggest that oral administration of AMSE could activate BAT activity in HFD mice.
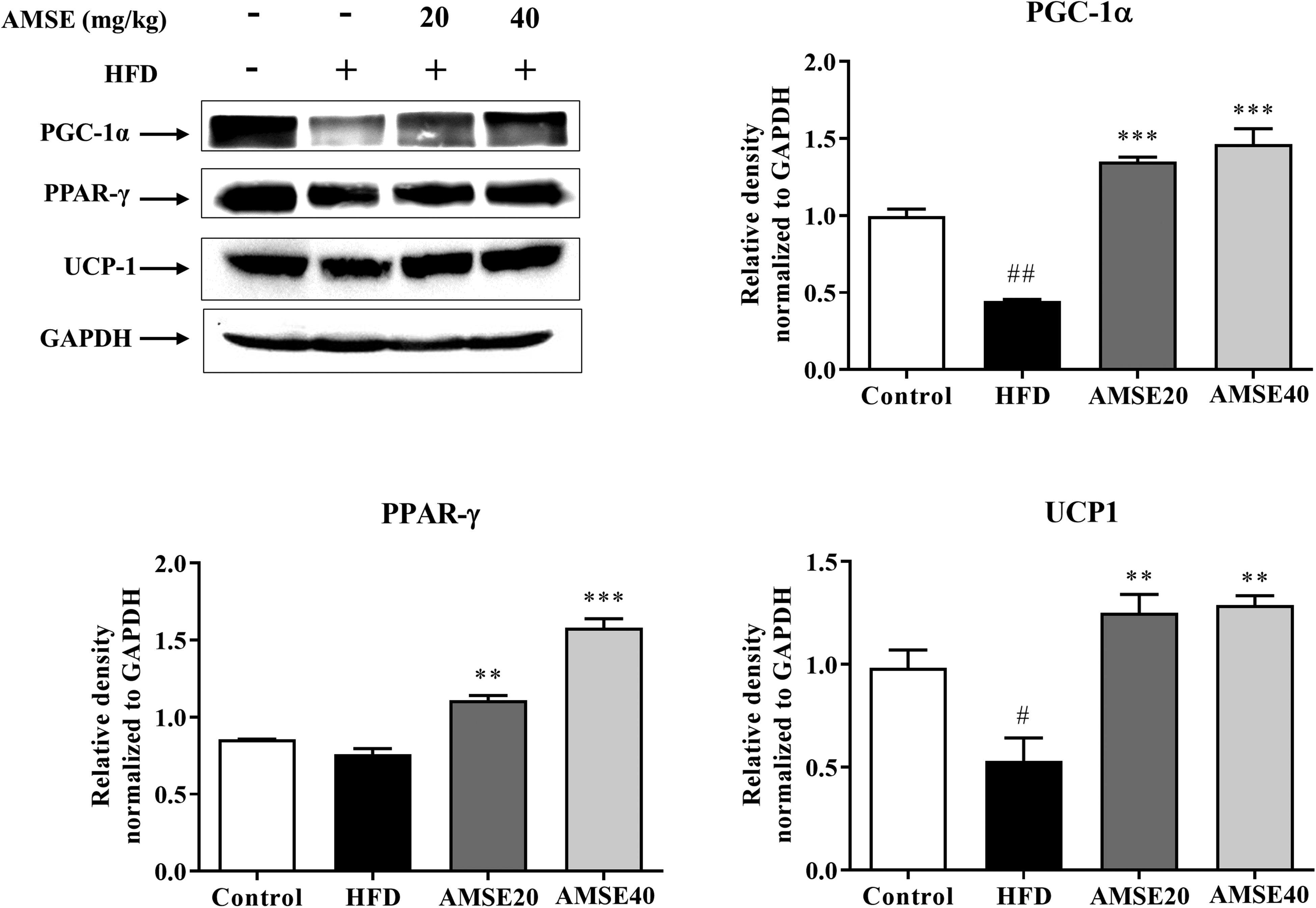
AMSE stimulates thermogenic program in brown adipose tissue (BAT). Representative western blot images showing changes in the markers of BAT activity. Densitometry analysis of blots was performed using ImageJ software (version 1.46, National Institutes of Health). Relative expression of proteins was normalized to GAPDH. #P < .05 and ##P < .01 versus control group; **P < .01 and ***P < .001 versus HFD group. Displayed are the cropped blot images.
Interestingly, AMSE induced the markers of brown adipocytes in eWAT. We observed that 40 mg/kg of AMSE (AMSE40) strongly induced the expression of PGC-1α and PPARγ (P < .01) as compared to the untreated HFD group (Figure 7). The expression of UCP-1 was increased in the AMSE-treated groups. However, the data were not significant compared to the untreated HFD group. These data suggest that AMSE may lead to browning of eWAT, which can substantially contribute to its antiobesity effects.
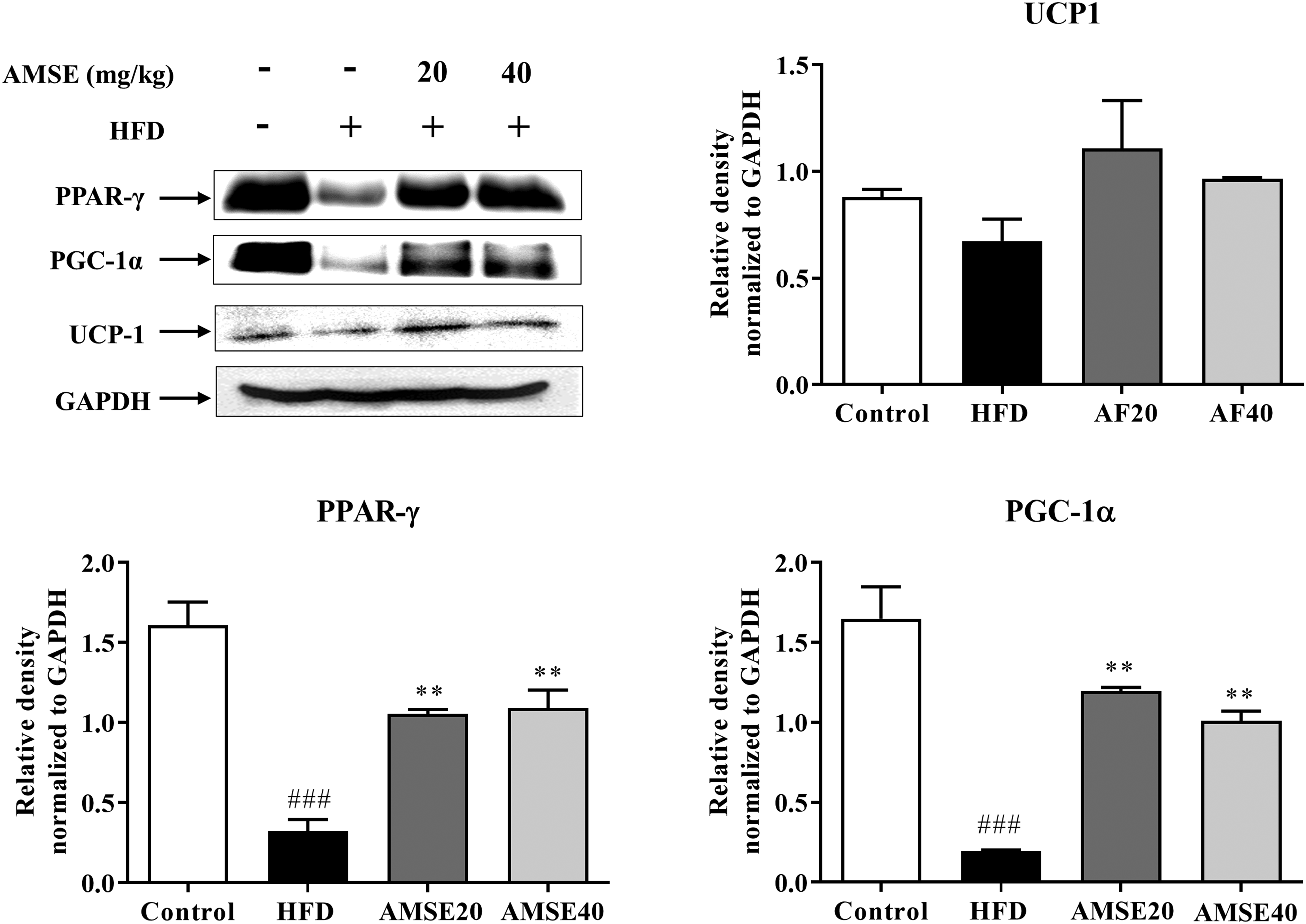
Administration of AMSE induces browning of eWAT in HFD mice. Representative western blots showing expression of BAT proteins in eWAT. Densitometry analysis of blots using ImageJ software (version 1.46, National Institutes of Health). Relative expression of proteins was normalized to GAPDH. ###P < .001 versus control group; **P < .01 versus HFD group. Displayed are the cropped blot images.
Discussion
It is well known that consumption of high fat diet is one of the prime causes of metabolic diseases such as obesity, insulin resistance, and cardiovascular diseases alongside satiety lifestyle. 31 Dong et al 32 previously reported that the fat diet exposure leads to maternal obesity which in turn is associated with cardiovascular complications in the offspring. There are several strategies reported to ameliorate metabolic health. Energy expenditure via thermogenesis is a promising therapeutic approach to combat obesity. 33 Promoting a thermogenic program in BAT or inducing beige adipocytes in WAT is an attractive strategy against obesity and associated complications.34,35 Several phytochemicals have shown potential as BAT activators and thermogenic agents. 11
We have investigated the thermogenic effects of a standardized GP seed extract containing 6-paradol as a key bioactive ingredient in HFD mice. A 6-week treatment with the extract significantly reduced body weight and improved liver function and serum lipid profile. Also, the HFD-induced changes in weights of fat depots were restored dose dependently by the AMSE treatment.
It is further demonstrated that AMSE could initiate thermogenesis by upregulating the key regulatory factors of BAT activity. UCP1, the mitochondrial membrane protein, is known to be the central component of BAT activity and is the classical marker of thermogenesis. 36 HFD-mice treated with 20 mg and 40 mg/kg doses of AMSE could significantly enhance the expression of UCP-1 in BAT.
PGC-1α and PPARγ are reported to play vital roles in BAT activity. PGC-1α regulates the expression of UCP1 including a few other BAT genes.37,38 PGC-1α-mediated stimulation of β-adrenergic pathway leads to the induction of UCP-1. 37 AMSE-treated mice showed increased expression of PGC-1α to a significant extent. PPARγ partly mediates brown adipocyte functionality. 39 Foellmi-Adams et al 40 have shown that Pioglitazone, a PPARγ agonist, enhanced UCP1 expression in mouse BAT. The activation of PPARγ could lead to the increased expression of glycerol kinase (GYK) protein, which is highly correlated with BAT activity in humans. 41 Our results demonstrated marked increase in PPARγ expression in BAT following the administration of AMSE to HFD mice. Overall, these results clearly indicate increased BAT activity upon AMSE ingestion in mice. Previously, Iwami et al 28 reported that GP seed extract could enhance BAT activity driven by the sympathetic nerve system.
To gain insights into the effects of AMSE on beige adipocyte induction in WAT in HFD mice, we have examined the expression of classical brown adipocyte markers in epididymal WAT. Here we have chosen to evaluate the browning of epididymal WAT since it is a comparatively more pathogenic depot than subcutaneous WAT, associated with high risk of cardiometabolic disease. 42 Furthermore, in humans, visceral fat depot has a higher expression of browning genes. 43 Like the trend in BAT, expression of PGC-1α and PPARγ, the transcriptional regulators of UCP1 were increased significantly in AMSE-treated HFD mice. These results indicated that the extract could induce browning of epididymal WAT.
AMSE-mediated induction of thermogenesis could be attributed to the presence of pungent vanilloid compounds such as 6-paradol, 6-gingerol, and 6-shogaol. These characteristic compounds of the ginger family have previously been shown to possess antihyperglycemic activity in experimental in vitro and in vivo models. 24 Haratake et al 44 reported the antiobesity effect of 6-paradol via the activation of BAT activity, compared to 8- and 12-paradols due to reduced alkyl chain length. In another study, injection of 6-paradol significantly triggered the BAT thermogenesis in rats through activation of sympathetic nerve activity. 28 The findings from our study are in line with the previous reports highlighting the potential of vanilloid compounds in the GP extract contributing to weight loss by thermogenesis.
The present study has some limitations as we have not used the AMSE alone group alongside the other treatment groups. Also, the metabolic rate of the animals was not recorded during the study, which could have substantiated the outcome of the study. However, this study demonstrated that the standardized GP extract could ameliorate the diet-induced obesity in mice via induction of beige adipocytes in WAT. Further studies are required in a clinical scenario to further elucidate the efficacy and tolerability of the extract in human subjects.
Materials and Methods
Plant Extract
AfperFitTM is a standardized AMSE containing 10% 6-paradol, procured from Department of Phytochemistry, Vidya Herbs Pvt. Ltd., Bangalore, India.
Reagents and Antibodies
The reference compound 6-paradol (HPLC grade 98%, WBKLS0100) was procured from Chemfaces. All reagents and solvents were of analytical grade. High-fat diet (D12451, 45% kcal fat, Research Diet) was procured from Hylasco Biotechnology (India) Pvt. Ltd. Antibodies for UCP-1 (sc-293418), PPAR-γ (sc-7273), PGC-1α (sc-13067), glyceraldehyde 3-phosphate dehydrogenase (GAPDH, sc-47724) and horseradish peroxidase (HRP)-conjugated antimouse immunoglobulin G (WBKLS0100) were obtained from Santa Cruz Biotechnology.
Quantitative Analysis of AMSE by HPLC
HPLC was performed on a Shimadzu LC2030 C Prominence-i system equipped with a quaternary low-pressure gradient solvent delivery LC2030 pump with high-pressure switching valves, online LC2030 degasser unit, a high sensitivity LC2030 ultraviolet (UV) detector, high-speed drive LC2030 autosampler and a large capacity column oven. The system was controlled and data analyzed by LabSolutions software. A separation was carried out in Kinetex C-18 column (100 Ǻ, 250 mm × 4.6 mm, 5 μm pore size).
The content of 6-paradol in AMSE was quantitatively analyzed at UV detection of 230 nm using a mobile phase of a mixture of orthophosphoric acid (0.2%): acetonitrile (40:60) with isocratic flow rate of 1.2 mL/min and the injection volume of 5 μL. The compound was quantified at UV detection of 280 nm. All solutions were degassed and filtered through a 0.45 μm pore size filter. The column was maintained at 26 °C throughout the analysis. Methanol was used as a diluent and the total liquid chromatography (LC) run time was 20 min. The peaks of compounds in AMSE were quantified by comparing with the retention time (RT) of respective reference standards separately.
HFD-Induced Obesity Model
Animals
Male C57Bl/6 mice (12 weeks old) were procured from Hylasco Biotechnology Pvt. Ltd. India (Reg No. 1808/PO/RcBt/S/15/CPCSEA). Mice were housed in a room under controlled temperature (22 ± 3 °C), humidity (30%–70%) with a 12 h light/dark cycle. All animals had access to food and water ad libitum. The animal experiments were conducted after approval from the Institutional Animal Ethics Committee (VHPL/PCL/IAEC/15/18 dated 21/09/2018) of Vidya Herbs Pvt. Ltd.
After a 1-week acclimatization, the animals were randomized into 4 groups (n = 10 per group): control group (Control); HFD-fed mice; HFD plus 20 mg/kg body weight AMSE (AMSE20); HFD plus 40 mg/kg body weight AMSE (AMSE40). The untreated- and AMSE-treated HFD mice were given HFD for 14 weeks. The mice in AMSE20 and AMSE40 were administered p.o with respective doses of AMSE from week 9 to the end of week 14 (6 weeks in total). The animals were monitored for daily feed consumption. The body weight changes were recorded on a weekly basis. After 14 weeks, all the mice were sacrificed using an overdose of gaseous anesthesia (5% isoflurane). Blood samples were collected by heart puncture and centrifuged at 3500 rpm for 5 min to obtain serum. Liver and fat depots were harvested and weighed. WAT and BAT tissues were homogenized in tissue lysis buffer (50 mM Tris pH 7.5, 150 mM NaCl, 1% Triton X-100) for further analysis.
Serum Biochemical Measurements
The serum samples were analyzed for AST, ALT, cholesterol, TG, LDL-c and HDL-c using commercial kits from Randox Laboratory in a biochemical analyser (Randox RX Imola, Co.).
Histopathology
The histological changes in the liver were examined using hematoxylin and eosin (H&E) staining. Briefly, the tissues were fixed in 4% formalin and embedded in paraffin. The paraffin sections were then cut into 4 µm thickness and stained with H&E and observed for liver histomorphology. Oil Red O staining was performed to examine the lipid droplets in the liver. The images were captured using a microscope (Leica).
Similarly, paraffin-embedded sections of epididymal WAT were prepared and observed for histological changes; images were photographed. Adipocyte size was determined using a microscope at 100 × magnification in 10 random fields per section.
Western Blotting
The eWAT homogenates were measured for total protein using the Bradford assay. A total of 50 to 75 μg of homogenates were resolved on 8% to 12% sodium dodecyl sulf-polyacrylamide gel electrophoresis gels and transferred to a polyvinylidene fluoride membrane. The membranes were blocked with 5% bovine serum albumin for 1 h and incubated overnight at 4 °C with appropriately diluted primary antibodies. The blots were washed thrice with 0.1% Tween 20 in Tris-buffered saline (TBS), later incubated for 1 h with HRP-conjugated secondary antibody (1:5000 dilution) at room temperature. The protein bands were detected using ImageQuantTM LAS 500 (GE Healthcare Life Sciences). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as loading control.
Statistical Analysis
GraphPad Prism 9.0 was used for statistical analysis. The data are expressed as the mean ± standard deviation (SD). Statistical comparisons were performed using one-way analysis of variance followed by Tukey’s multiple comparison test. The data were considered statistically significant at P < .05.
Footnotes
Acknowledgments
The authors thank Dr Muralidhar Talakad for the technical support in histological examination of tissue samples.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Animal Ethics Committee (IAEC) of Vidya Herbs Pvt. Ltd.
