Abstract
A new natural indole, vibrindole B (
Keywords
Introduction
Marine bacteria belonging to the genus Pseudovibrio (Rhodobacterales) have proven to be not only an important source of bioactive compounds, but also to play an interesting role in marine ecology.1-5 Pseudovibrio species are described by Shieh 1 as heterotrophic, marine, facultative anaerobes capable of denitrification and fermentation. Since the first type strain, P. denitrificans DN34, was isolated from seawater samples collected in Nanwan Bay, Taiwan, 1 more and more Pseudovibrio species have been found around the world in both marine organisms such as sponges, 2 corals, 6 tunicates, 7 flatworms, 8 and the environment, such as seawater.1,5,9
Pseudovibrio species not only live with their hosts, but also provide benefits to them and have diverse genes for producing multiple bioactive compounds. 4 Some of these have showed activity against other microorganisms. This may prevent their host from infection by other marine microorganisms. For example, heptylprodigiosin, produced by P. denitrificans strain Z143-1 isolated from a Philippine tunicate, displayed anti-Staphylococcus aureus activity; 7 and a Pseudovibrio species from a marine sponge produced tropodithietic acid (TDA). 10 In a previous study, TDA displayed inhibitory activity against a range of marine bacteria, such as α-Proteobacteria, γ-Proteobacteria, Actinobacteria, and Flavobacteria. 11
The metabolites from Pseudovibrio species have displayed extensive pharmacological activities. For instance, P. denitrificans UST4-50 produced compounds belonging to the di(1H-indol-3-yl)methane family, which showed antifouling activity against larval settlement of Balanus amphitrite;
12
P. denitrificans BBCC725 isolated from seawater produced a new indole derivative, tetra(indol-3-yl)ethenone, that displayed cytotoxicity against L929 mouse fibroblasts and A549 human lung epithelial carcinoma cell lines.
9
In our continuing research aimed at discovering new natural compounds from marine organisms, in an organic extract of bacterium strain P81, identified as Pseudovibrio denitrificans, which was originally isolated from a marine sponge Aaptos sp. collected from Green Island, Taiwan, five indole derivatives were discovered, including one new natural product, vibrindole B (

Structures of vibrindole B (
Results and Discussion
Vibrindole B (
1H and 13C nuclear magnetic resonance spectroscopic data for
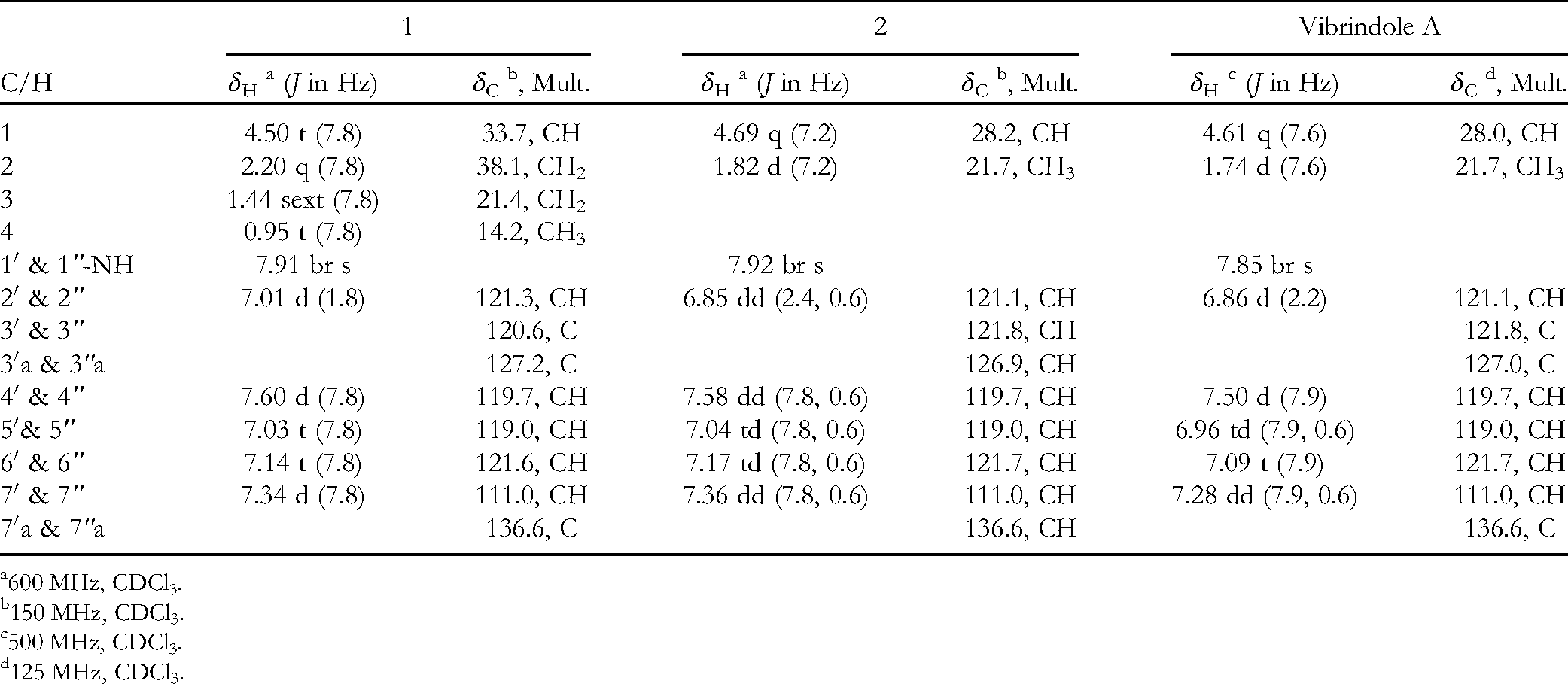
600 MHz, CDCl3.
150 MHz, CDCl3.
500 MHz, CDCl3.
125 MHz, CDCl3.
The 1H NMR spectrum of
The carbon skeleton of

Main COSY (–) and selective key HMBC () correlations of
Indoles
A proposed biosynthetic pathway for

The proposed biosynthetic pathway for
The cytotoxicity of indole derivatives
Cytotoxic data of indoles

Percentage of cell viability at 10 μM concentration in 72 h. Results are presented as mean ± S. E. M. (n = 3). **p < 0.01, ***p < 0.001 compared with the control (dimethylsulfoxide, DMSO).
Experimental
General
IR spectra were measured on a Thermo Scientific Nicolet iS5 FT-IR spectrophotometer (Thermo Fisher Scientific). 1H and 13 C NMR spectra were recorded on Jeol ECZ NMR spectrometers (Jeol) at either 600 or 400 MHz for 1H NMR, and either 150 or 100 MHz for 13 C NMR, respectively, with residual solvent as the internal standard. Positive-ion ESIMS and HRESIMS were acquired on a Bruker 7 Tesla solariX Fourier transform mass spectroscopy (FTMS) (Bruker). Silica gel column chromatography was carried out with C18 (17%) reverse-phase silica gel (230–400 mesh, SiliCycle Inc. ). Precoated silica gel 60 F254 plates (E. Merck; 0.25 mm in thickness) were used for thin-layer chromatographic analyses; compounds were visualized by spraying with a 10% solution of H2SO4 and heating on a hotplate. Isolation and purification of isolates by normal-phase high performance liquid chromatography (NP-HPLC) and reverse-phase high performance liquid chromatography (RP-HPLC) were performed using (a) a Hitachi L-7110 instrument equipped with a normal-phase column (Supelco Ascentis Si, 250 × 21.2 mm, 5 µm; Sigma-Aldrich); and (b) a Hitachi L-2130 pump, a Hitachi L-2455 photodiode array detector, and a reverse-phase column (Luna 5 µm, 250 × 21.2 mm; Phenomenex).
Culture Conditions, Marine Bacterial Isolation, and Extraction
Marine bacterium number P81 was isolated from a marine sponge, Aaptos species, collected off the coast of Green Island at a depth of − 5 m. P81 was 100% identical to Pseudovibrio denitrificans DN34 (Genebank accession no. NR_029112) on the basis of the 16S rRNA gene sequence. The marine bacterium was cultured in 2.5 L flasks containing 1.0 L M1 broth (1% starch, 0.4% yeast extract, and 0.2% peptone) with 80% seawater and incubated at 25 °C on an orbital shaker at a rotation speed of 120 rpm. After five days of incubation, extraction of the culture broth (45.0 L) with EtOAc (2 × 45.0 L) yielded 4.62 g of crude extract in total. The extract obtained was then stored at − 20°C until further use.
Isolation
The extract was fractionated on a C-18 silica gel column and eluted using a mixture of MeOH and H2O (stepwise, 3:7 − MeOH) into 8 fractions (1to 8). Fraction 6 was purified separately by NP-HPLC, using n-hexane − acetone (7:3), to afford 18 fractions, 6A to 6R. 6D and 6G were purified using RP-HPLC, using MeCN − H2O (11:9), to yield
Vibrindole B (
Vibrindole A (
Trisindoline (
Norharmane (
3-(Hydroxyacetyl)indole (
Cell Culture and Cell Viability Assay
T24 human bladder carcinoma cells were cultured at 37°C in Roswell Park Memorial Institute 1640 Medium (RPMI 1640, Gibco) supplemented with 1% antibiotic/antimycotic solution (100 U/mL penicillin, 100
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211033735 - Supplemental material for Natural Indoles From the Bacterium Pseudovibrio denitrificans P81 Isolated From a Marine Sponge, Aaptos Species
Supplemental material, sj-docx-1-npx-10.1177_1934578X211033735 for Natural Indoles From the Bacterium Pseudovibrio denitrificans P81 Isolated From a Marine Sponge, Aaptos Species by Shu-Yen Fang, Sheng-Yuan Chen, You-Ying Chen, Tsu-Jen Kuo, Zhi-Hong Wen, Yu-Hsin Chen, Tsong-Long Hwang and Ping-Jyun Sung in Natural Product Communications
Footnotes
Acknowledgments
The authors are thankful to Ms. Hsiao-Ching Yu and Chao-Lien Ho, the High Valued Instrument Center, National Sun Yat-sen University, for the mass (MS000600) and NMR (NMR001100) spectra (MOST 110-2731-M-110-001).
Author Contributions
S-YF carried out the investigations; S-YC took part in the investigations; Y-YC participated in the investigation; T-JK participated in the investigations; Z-HW took part in the investigations; Y-HC was part of conceptualization, investigation, and draft preparation; T-LH was involved in conceptualization, investigation, and draft preparation; P-JS took part in conceptualization and draft preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zuoying Branch of Kaohsiung Armed Forces General Hospital (grant number KAFGH-ZY-A-ZBH107-05, MOST107-2320-B-291-001-MY3, MOST109-2320-B-291-001-MY3).
Funding
This research was funded by grants from the National Museum of Marine Biology and Aquarium; Zuoying Branch of Kaohsiung Armed Forces General Hospital (KAFGH-ZY-A-ZBH107-05); and the Ministry of Science and Technology, Taiwan (Grant Nos.: MOST 107-2320-B-291-001-MY3 and 109-2320-B-291-001-MY3), awarded to Zhi-Hong Wen and Ping-Jyun Sung.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
