Abstract
Five conjugated polyene ketones (1-5) were isolated from the methanol extract of the marine sponge Clathria (Thalysias) reinwardti (Vosmaer, 1880) living in the coastal waters of Vietnam. Their structures were determined to be 8-(2′,3′,4′-trimethylphenyl)-6-methyl-oct-3(E),5(E),7(E)-trien-2-one (1), 13-apoastaxanthinone (2), 9-apoastaxanthinone (3), 2,3-dehydro-4-oxo-β-ionone (4), and 4-(2′,3′,4′-trimethylphenyl)-but-3(E)-en-2-one (5), by extensive analysis of high-resolution electron spray ionization mass spectrum (HR-ESI-MS), one-dimensional, and two-dimensional (2D) nuclear magnetic resonance (NMR) spectra, as well as by comparison of the spectral data with those reported in the literature. Compound 1 was new, compounds 2 to 4 were isolated from nature for the first time, and the chemical structure as well as the NMR assignments, of 5 were indicated by 2D NMR for the first time. Additionally, compound 5 exhibited cytotoxic activity against the human cancer cells SK-LU-1, SK-Mel-2, MCF-7, and Hep-G2 with half-minimal inhibitory concentration (IC50) values of 15.12 ± 3.43, 17.41 ± 2.83, 33.12 ± 3.39, and 34.38 ± 3.52 µM, respectively, but displayed only a weak cytotoxic effect on the normal HEK-239A cells (IC50 64.67 ± 3.67 µM). Compound 5 also significantly increased Caspase-3 activity in SK-LU-1 cells at concentrations of 10, 15, and 20 µM.
Introduction
Marine sponges of the genus Clathria, family Microcionidae, are widely distributed in tropical shallow waters and temperate regions, especially along with the cost of the southern hemisphere.1,2 The World Porifera Database lists 381 valid species of Clathria. 3 Chemical investigation of Clathria sponges indicated the presence of novel secondary metabolites exhibiting diverse chemical structures including alkaloids,4-8 steroids9-11 terpenes.12-14 In addition, these compounds have been proposed to have numerous biological effects, such as cytotoxic,9,15,16 antiinflammatory, 17 antifungal and antibacterial.18-20 In previous papers we have reported the isolation and structural determination of several steroids from Clathria vulpina. 9 In our continuing investigation of the chemical constituents from the genus Clathria, we report herein the isolation and structure elucidation of 5 conjugated polyene ketones from the marine sponge C. reinwardti, including a new compound (1) and 3 compounds (2-4) which have not been found from nature. The cytotoxic effects of these compounds on lung carcinoma (SK-LU-1), breast carcinoma (MCF-7), hepatocellular carcinoma (HepG2), and melanoma cells (SK-Mel-2) were examined by MTT assay.
Results and Discussion
Purification of the methanol extract of the marine sponge C. reinwardti using various chromatography methods obtained 5 conjugated polyene ketone compounds (1-5). Their structures are shown in Figure 1.

Chemical structures of compounds 1 to 5.
The molecular formula of compound 1 was deduced to be C18H22O based on the cluster of quasimolecular ion peaks in the high-resolution electron spray ionization mass spectrum (HR-ESI-MS) at m/z 255.1741 [M + H]+ (calcd. for [C18H23O]+: 255.1743, Δ = −1.3 ppm), indicating 8° of unsaturation (Supplemental Figure S1). The 1H nuclear magnetic resonance (NMR) spectrum of 1 (Supplemental Figure S2) exhibited 2 aromatic protons at δH 7.00 and 7.26 (each 1H, d, J = 8.0 Hz, H-5′ and H-6′), 5 olefinic protons at δH 6.70 and 7.10 (each 1H, d, J = 16.0 Hz, H-7 and H-8), 6.31 (1H, d, J = 11.5 Hz, H-5), 7.60 (1H, dd, J = 15.5, 11.5 Hz, H-4), and 6.21 (1H, d, J = 15.5 Hz, H-3), and 5 singlet methyls at δH 2.15, 2.21, 2.30, 2.31, and 2.31. The 13C NMR spectrum of 1 (Supplemental Figure S3) exhibited 18 carbon signals, including 1 ketone (δC 198.5), 6 aromatic (δC 123.2, 127.6, 134.1, 134.5, 135.5, and 136.7), 3 double bonds (δC 129.0, 129.7, 130.8, 133.4, 139.0, and 145.4), and 5 methyls (δC 13.4, 15.9, 15.9, 21.0, and 27.8), identified from the HSQC spectrum (Supplemental Figure S4). In the 1H-1H COSY spectrum, cross peaks were observed between H-5′ (δH 7.00) and H-6′ (δH 7.26), H-3 (δH 6.21) and H-4 (δH 7.60), H-4 and H-5 (δH 6.31), and H-7 (δH 6.70) and H-8 (δH 7.10) (Supplemental Figure S8). Three methyl groups at δH 2.31, 2.21, and 2.30 attached to C-2′, C-3′, C-4′, respectively, were confirmed by HMBC correlations from protons 2′-CH3 to C-1′ (δC 134.5)/C-2′ (δC 134.1)/C-3′ (δC 135.5), from 3′-CH3 to C-2′/C-3′/C-4′ (δC 136.7), and from 4′-CH3 to C-3′/C-4′/C-5′ (δC 127.6). 21 The HMBC correlations from H-1 (2.31) to C-2 (δC 198.5), from H-3 (δH 6.21) to C-2/C-5 (δC 129.0), from 6-CH3 protons (δH 2.15) to C-5 (δC 129.0)/C-6 (δC 145.4)/C-7 (δC 133.4), and from H-8 (δH 7.10) to C-6/C-7/C-1′ (δC 134.5)/C-2′ (δC 134.1)/C-6′ (δC 123.2) indicated the ketone group at C-2, 3 double bonds at C-3/C-4, C-5/C-6, C-7/C-8, the methyl group attached to C-6, and the conjugated polyene ketone moiety linked to C-1′ of the aromatic ring (Figures 1 and 2, Supplemental Figures S5-S7). The large coupling constants of H-7 and H-8 (J = 16.0 Hz), H-3 and H-4 (J = 15.5 Hz) suggested the trans-conjunction of these double bonds. The smaller carbon chemical shift of 6-CH3 (δC 13.4) was similar to that of 13-apoastaxanthinone, suggesting an E-geometric configuration of the C-5/C-6 double bond (Table 1). 22 From the above evidence, compound 1 was determined to be 8-(2′,3′,4′-trimethylphenyl)-6-methyl-oct-3(E),5(E),7(E)-trien-2-one, a new compound, named clathriarein A.

Key HMBC and H-H COSY correlations of compounds 1 to 5.
1H NMR and 13C NMR Data for Compounds 1 and 5 in Deuterated Chloroform.

Abbreviations: NMR, nuclear magnetic resonance; Pos., position.
Measured at 125 MHz.
Measured at 500 MHz.
Compounds 2 and 3 were identified to be 13-apoastaxanthinone (2) and 9-apoastaxanthinone (3) by comparing their NMR data with the previously reported data and found to match well (Table 2). 22 In addition, the structures of these compounds were further confirmed by HR-ESI-MS, one-dimensional (1D) and two-dimensional (2D) NMR spectra (Supplemental Figures S9-S20). The absolute configuration at C-3 of these compounds was deduced to be S by their negative specific optical rotations, as previously reported (−97.2 for 3S-hydroxy-4-oxo-β-jonon and +91.8 for 3R-hydroxy-4-oxo-β-jonon). Compounds 2 and 3 were only reported as the reaction products formed during the self-initiated auto-oxidation of astaxanthin. 22 Thus, this is the first time compounds 2 and 3 have been found from nature.
1H NMR and 13C NMR Data for Compounds 2 to 4 in Deuterated Chloroform.

Abbreviations: NMR, nuclear magnetic resonance; Pos., position.
Measured at 125 MHz.
Measured at 500 MHz.
The NMR spectra of 4 showed signals due to 2 ketone groups at δC 182.0 and 197.2, 3 double bonds at δC 126.2/δH 6.10 and δC 144.7/δH 6.31, δC 130.6 and 157.8, and δC 138.7/δH 7.32 and δC 134.6/δH 6.32, and 4 methyl groups. The NMR signals of the side chain moiety of 4 were almost the same as those of 3, but differed from C-1 to C-5. In detail, the signals of the methylene (δC 45.4) and oxygenated methine carbon (δC 69.3) in 3 were replaced by double bond signals (δC 144.7/126.2) in 4 suggesting dehydration at C-2/C-3 (Table 2). This was confirmed by the HR-ESI-MS of 4 with the appearance of a quasimolecular ion peak at m/z 205.1215 [M + H]+ [calcd. for (C13H17O2)+ 205.1223, Δ = −3.9 ppm] and further by the HSQC and HMBC spectra (Supplemental Figures S21-S25). Thus, compound 4 was determined to be 2,3-dehydro-4-oxo-β-ionone, which has not been found from nature, but is believed to be formed from β-ionone by Aspergillus niger JTS 191. 23
Compound 5 was identified as 4-(2′,3′,4′-trimethylphenyl)-but-3(E)-en-2-one, a known compound isolated from marine sponge Tedania ignis, but its NMR data were not assigned for each carbon position. 21 This is the first time that compound 5 has been found from the genus Clathria and its NMR data were assigned by 1D and 2D NMR spectra (Supplemental Figures S26-S30). C. reinwardti is characterized by its colorful appearance, being usually rich in carotenoids. Compounds 1 to 5, therefore, were proposed to be biosynthesized from carotenoids contained in the sponge, such as β-carotene and χ,χ-carotene (Supplemental Figure S31).
Compounds 1 to 5 were evaluated for their cytotoxic effects on 4 human cancer cell lines (lung carcinoma [SK-LU-1], breast carcinoma [MCF-7], hepatocellular carcinoma [Hep-G2], and melanoma [SK-Mel-2]), and a normal cell line (human embryonic kidney [HEK-293A]). At a concentration of 100 µM, compounds 1 to 5 displayed cytotoxic effects against the tested cell lines with cell death percentages in the range from 15.9% to 74.4% (Supplemental Table S1). The experiments with cell death percentages over 50% were further performed as dose-dependent studies to find the corresponding half-minimal inhibitory concentration (IC50) values. Compound 5 exhibited the most cytotoxic effects on SK-LU-1, MCF-7, Hep-G2, SK-Mel-2, and HEK-239A cell lines with IC50 values of 15.12 ± 2.20, 33.12 ± 3.39, 34.38 ± 3.52, and 17.41 ± 2.38, and 64.67 ± 3.76 µM, respectively (Table 3). The other compounds (1-4) were either weak or inactive. Ellipticine was used as the positive control in all the experiments. It exhibited high cytotoxic effects against all the tested cell lines with IC50 values of 1.71 ± 0.20 µM (SK-LU-1), 2.03 ± 0.24 µM (MCF-7), 2.07 ± 0.20 µM (Hep-G2), 1.46 ± 0.16 µM (SK-Mel-2), and 1.75 ± 0.20 µM (HEK-239A). The results suggested that compound 5 could be used for its selective cytotoxic effect on SK-LU-1 and SK-Mel-2 cancer cells in comparison with other cancer cell lines (MCF-7 and Hep-G2) and the normal cell line (HEK-239A). Compound 5 was then further investigated whether its cytotoxic effect come from the suppression of cell differentiation and/or induction of cell death. Based on the cytotoxic results (smallest IC50 value), SK-LU-1 cell line was chosen for study. As shown in Figure 3, the SK-LU-1 cell cycle did not significantly change by the presence of compound 5 (at concentrations of 5, 10, and 15 µM) suggesting that the cell differentiation is not affected by this compound. On the other hand, compound 5 exhibited activation of caspase-3, a biomarker promoting the apoptosis process (Figure 4). After 48 h incubation, compound 5 (at concentrations of 10, 15, and 20 µM) increased Caspase-3 activity in comparison with the negative control and positive control (camptothecin). The results suggest that compound 5 partly induce the apoptosis process by activation of Caspase 3.

Effects on SK-LU-1 cell cycle after 24 h incubation with negative control (a) and compound 5 at concentrations of 5, 10, and 15 µM (b-d).

Effect of compound 5 in the activation of caspase 3 SK-LU-1 cells; **P < 0.01 and *P < 0.05 compared to negative control.
Cytotoxic Effect of Compounds 1 to 5 Against various Cell Lines.
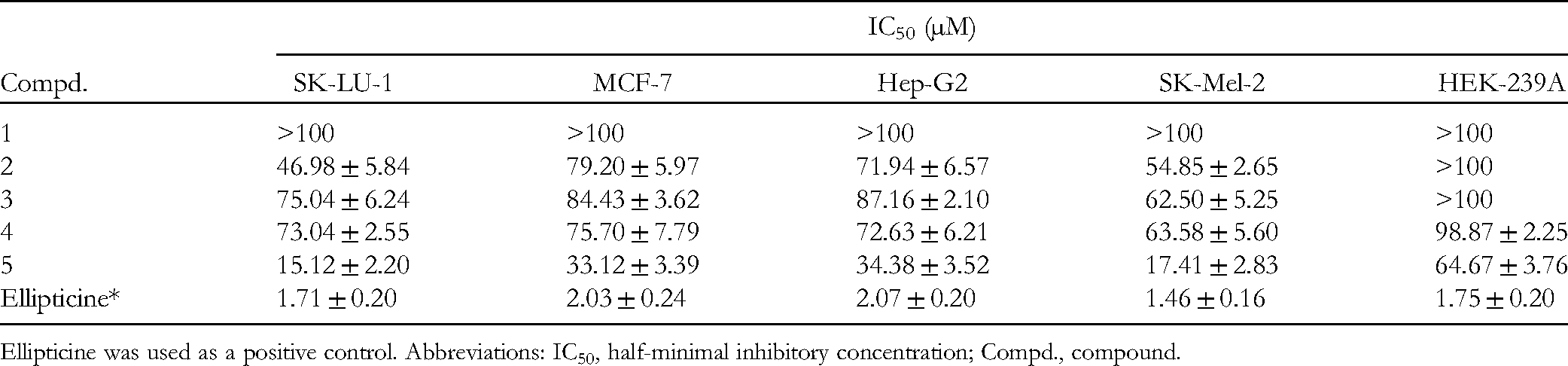
Ellipticine was used as a positive control. Abbreviations: IC50, half-minimal inhibitory concentration; Compd., compound.
Material and Methods
General Experimental Procedures
Optical rotation was measured on a Jasco P-2000 polarimeter, NMR spectra on a Bruker 500 MHz spectrometer, and HR-ESI-MS on an Agilent 6530 Accurate Mass Q-TOF LC/MS. Flash column chromatography was performed using either silica gel or reversed-phase C-18 resin (RP-18) resins as adsorbent. Thin-layer chromatography (TLC) was carried out on precoated silica gel 60 F254 and/or RP-18 F254S plates. TLC plates were visualized either under ultraviolet (UV) irradiation (254 and 365 nm) or by spraying with H2SO4 solution (5%) followed by heating with a heat gun. HPLC was carried out using an AGILENT 1100 HPLC system.
Animal Material
The sponge samples were collected in Vanphong Bay, Nha Trang, Vietnam in May 2020 and identified as Clathria (Thalysias) reinwardti Vosmaer, 1880 by one of our authors, Prof. Do Cong Thung, the Institute of Marine Environment and Resources, Vietnam Academy of Science and Technology (VAST). A voucher specimen (NCCT-B130) was deposited at the Institute of Marine Biochemistry, VAST.
Extraction and Isolation
The frozen sponge sample Clathria reinwardti (15.0 kg) was defrosted, washed with water, and cut into small pieces, and then extracted 3 times with methanol in an ultrasonic bath (each 30 L MeOH, room temperature, 60 min). After filtration and removal of the solvent under reduced pressure, the methanol extract (115 g) was suspended in distilled water (2 L) and extracted with dichloromethane (2 L) to give a dichloromethane soluble fraction (CR1, 50 g). This was chromatographed on a silica gel column, eluting with a gradient solvent system of hexane/acetone (100/1-0/1, v/v) to give 5 fractions CR1A (5.7 g), CR1B (4.2 g), CR1C (4.3 g), CR1D (12.1 g), and CR1E (6.0 g). Fraction CR1E (6.0 g) was chromatographed on an RP-18 column, eluting with acetone/water (5/1, v/v) to give 3 fractions CR1E1-CR1E3. CR1E1 was chromatographed on a silica gel column, eluting with hexane/ethyl acetate (12/1, v/v) to obtain 2 fractions, CR1E1A and CR1E1B. CR1E1A was purified on an HPLC column (J'sphere ODS H-80, 250 mm × 20 mm column), eluting with 80% acetonitrile in water to yield compound 1 (3.8 mg). CR1E1B was purified on an HPLC column (J'sphere ODS H-80, 250 mm × 20 mm column), eluting with 75% acetonitrile in water to yield compound 2 (5.1 mg). CR1E2 was chromatographed on an HPLC column using the same conditions to yield compound 5 (4.8 mg). Compounds 3 (6.0 mg) and 4 (7.0 mg) were isolated from CR1E2 by HPLC (J'sphere ODS H-80, 250 mm × 20 mm column), eluting with 65% acetonitrile in water.
Clathriarein A (1)
A dark yellow solid. UV (CHCl3) λmax (logε) 270 (3.65), 351 (4.28). Infrared (IR) (KBr) νmax 2956, 2917, 2849, 1730, 1633 cm−1. HR-ESI-MS m/z 255.1741 [M + H]+ (calcd. for [C18H23O]+, 255.1743, Δ = −1.3 ppm). 1H-NMR (CDCl3, 500 MHz) and 13C-NMR (CDCl3, 125 MHz): see Table 1.
13-Apoastaxanthinone (2)
A dark yellow solid.
9-Apoastaxanthinone (3)
A dark yellow solid.
2,3-Dehydro-4-oxo-β-Ionone (4)
A dark yellow solid. UV (CHCl3) λmax (logε) 225 (3.40), 271 (3.62). IR (KBr) νmax 2919, 1656, 1638 cm−1. HR-ESI-MS m/z 205.1215 [M + H]+ (calcd. for [C13H17O2]+, 205.1223, Δ = −3.9 ppm). 1H-NMR (CDCl3, 500 MHz) and 13C-NMR (CDCl3, 125 MHz): see Table 2.
4-(2′,3′,4′-Trimethylphenyl)-but-3(E)-en-2-one (5)
A dark yellow solid. UV (CHCl3) λmax (logε) 214 (3.81), 236 (3.15), 303 (4.02). IR (KBr) νmax 2955, 2918, 2850, 1734, 1642 cm−1. HR-ESI-MS m/z 189.1270 [M + H]+ (calcd. for [C13H17O]+, 189.1274, Δ = −2.1 ppm). 1H-NMR (CDCl3, 500 MHz) and 13C-NMR (CDCl3, 125 MHz): see Table 1.
Cytotoxic assay
Refer to Supplemental Material.
Conclusions
One new (1) and 4 known (2-5) conjugated polyene ketones were isolated and structurally determined from the marine sponge Clathria (Thalysias) reinwardti (Vosmaer, 1880) living in the coast waters of Vietnam. Although chemical structures of compounds 2 to 4 were previously described, this is the first report on the identification of these compounds from a natural source. Assignments of NMR spectral data of compound 5 are also firstly indicated in this report. Cytotoxic assay suggested that compound 5 could have selective cytotoxic activity on SK-LU-1 (IC50 15.12 ± 3.43 µM) and SK-Mel-2 (IC50 17.41 ± 2.83 µM) cancer cell lines, compared to other cancer cell lines [MCF-7 (IC50 33.12 ± 3.39 µM) and Hep-G2 (IC50 34.38 ± 3.52 µM)], and normal cells HEK-239A (IC50 64.67 ± 3.67 µM). Compound 5 did not affect the SK-LU-1 cell cycle, but significantly increased Caspase-3 activity, which promotes cell death through apoptosis.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211043732 - Supplemental material for Conjugated Polyene Ketones From the Marine Sponge Clathria (Thalysias) Reinwardti (Vosmaer, 1880) and Their Cytotoxic Activity
Supplemental material, sj-docx-1-npx-10.1177_1934578X211043732 for Conjugated Polyene Ketones From the Marine Sponge Clathria (Thalysias) Reinwardti (Vosmaer, 1880) and Their Cytotoxic Activity by Bui H. Tai, Dan T. Hang, Do T. Trang, Pham H. Yen, Phan T. T. Huong, Nguyen X. Nhiem, Do C. Thung, Do T. Thao, Nguyen T. Hoai and Phan V. Kiem in Natural Product Communications
Footnotes
Acknowledgments
This research is funded by Vietnam Academy of Science and Technology under grant number TĐDLB0.01/20 to 22.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vietnam Academy of Science and Technology (grant number TĐDLB0.01/20-22).
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
