Abstract
Two new pregnane glycosides were isolated from the leaves of Gymnema sylvestre (Retz.) R.Br. ex Sm. Their chemical structures were determined as (20S)−12β-tigloyloxy-5α-hydroperoxy-3β,8β,14β,17β,20-pentahydroxypregn-6-ene 3-O-β-D-glucopyranosyl-(1→4)‐6-deoxy-3-O-methyl-β-D-allopyranosyl-(1→4)-β-D-oleandropyranosyl-(1→4)-β-D-cymaropyranosyl-(1→4)-β-D-cymaropyranoside (
Keywords
Gymnema sylvestre (Retz) R. Br. ex. Schult (Apocynaceae) known as “gur-ma” in Indian folklore, means “sugar killer,” have been used as a traditional diabetes medicine all over the world. In addition, methanol and other extracts of the leaves from G. sylvestre exhibit a variety of biological activities such as hypoglycemia, antioxidant, anti-obesity and antimicrobial. 1 -4 The principle chemical component of G. sylvestre are pregnaneglycosides 5 -7 and oleanane saponins. 8 -16 In the previous paper, we reported the isolation and structure elucidation of twelve pregnaneglycosides name gymsylosides A-F and gymnepregosides R-T. 6,7 Continue researching on bioactive compounds from the leave of G. sylvestre, we reported herein the isolation and structural elucidation of two new pregnaneglycosides and their α-glucosidase and α-amylase inhibition activities.
Results and Discussion
Compound
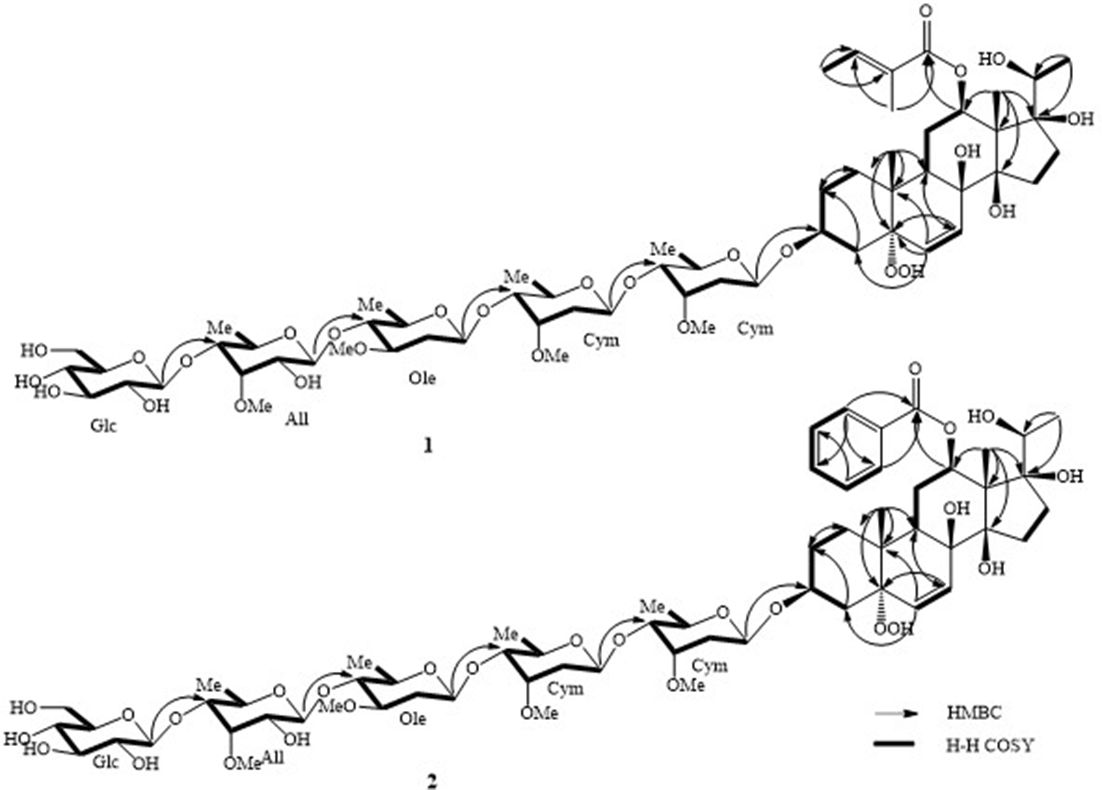
Important H-H COSY and HMBC correlations of compounds 1 and 2.
NMR Spectral Data for the Aglycone Moieties of 1 and 2 in Deuterated Methanol.

Abbreviation: NMR, Nuclear magnetic resonance.
Asterisk indicates overlapped signals.
aMeasured at 100 MHz.
bMeasured at 400 MHz.
NMR Spectral Data for Sugar Moieties of 1 and 2 in Deuterated Methanol.
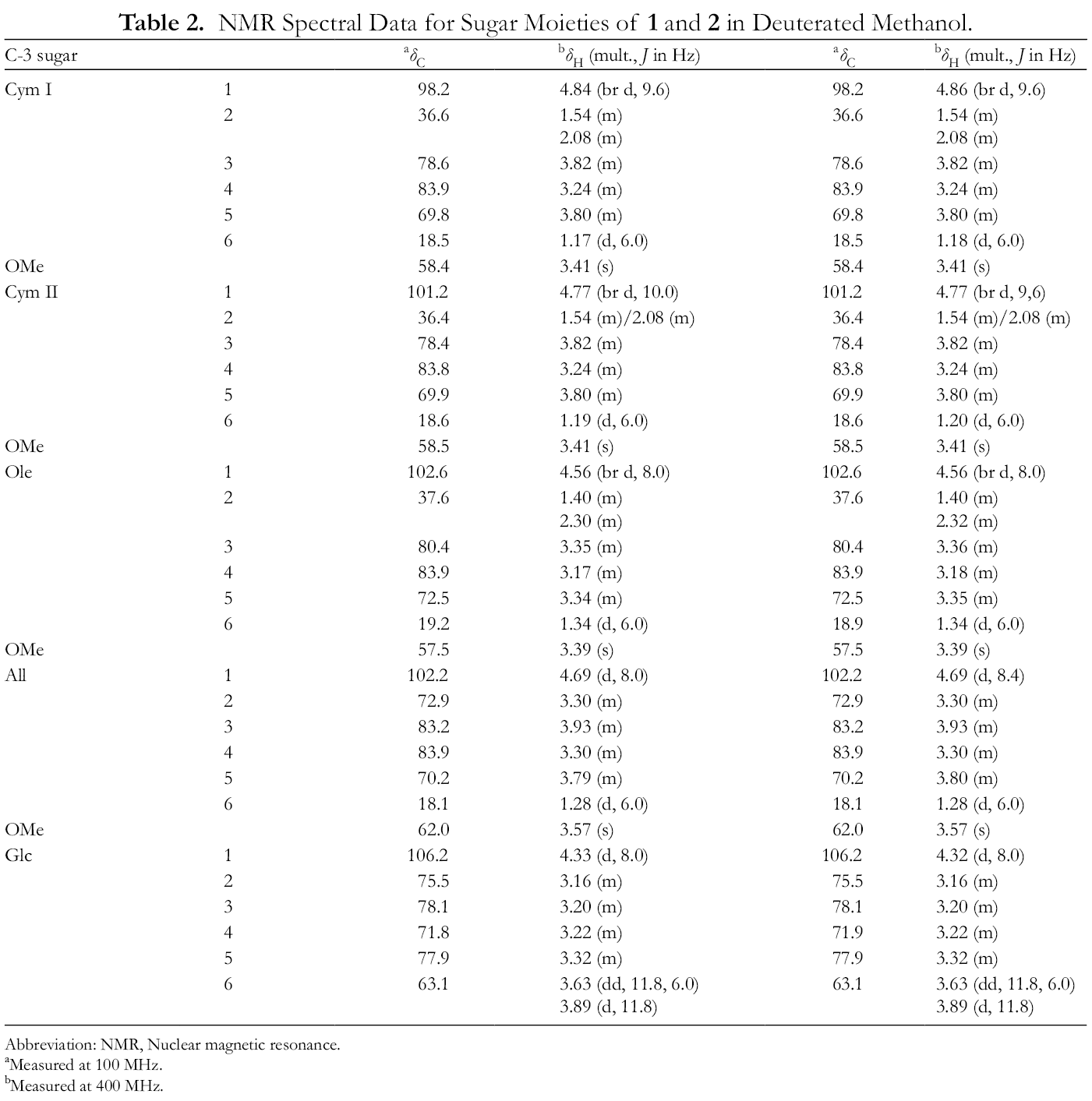
Abbreviation: NMR, Nuclear magnetic resonance.
aMeasured at 100 MHz.
bMeasured at 400 MHz.

Observed ROESY couplings of compounds 1 and 2.
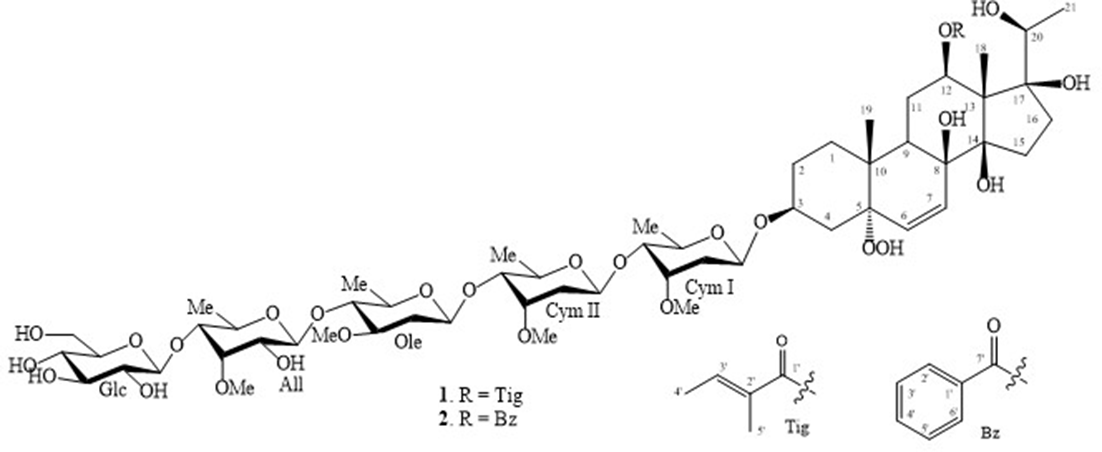
Chemical structures of compounds 1 and 2.
Compound
Compounds
Α-Glucosidase and Α-Amylase Inhibitory Effects of Compounds 1 and

aAcarbose was used as a positive control.
b500 µg/mL.
c100 µg/mL.
Material and Methods
General Experimental Procedures
All NMR spectra were recorded on an Agilent 400-MR-NMR spectrometer operated at 400 and 100 MHz for 1H and 13C, respectively. Data processing was carried out with the MestReNova ver.6.0.2 program. HRESIMS spectra were obtained using an AGILENT 6550 iFunnel Q-TOF LC/MS system. Optical rotations were determined on a Jasco DIP-370 automatic polarimeter. Preparative HPLC was carried out using an AGILENT 1200 HPLC system. Column chromatography was performed on silica-gel (Kieselgel 60, 70‐230 mesh and 230‐400 mesh, Merck) or YMC RP-18 resins (30, 50 μm, Fujisilisa Chemical Ltd.). For TLC, a pre-coated silica-gel 60 F254 (0.25 mm, Merck) and RP-18 F254S plates (0.25 mm, Merck) were used.
Plant Material
The leaves of Gymnema sylvestre (Retz.) R.Br. ex Sm. were collected in Hai Loc, Hai Hau, Nam
Dinh in November 2015, and identified by Dr. Nguyen The Cuong, Institute of Ecology and Biological Resources. A voucher specimen (NCCT-P20) was deposited at the Herbarium Institute of Marine Biochemistry, VAST.
Extraction and isolation
The dried powders of G. sylvestre leaves (4.0 kg) were sonicated with hot methanol (3 times × 10 L, each 3 hours) to give MeOH extract (450 g) after evaporation of the solvent. The MeOH extract was suspended in water and successively partitioned with hexane, CH2Cl2 and EtOAc to obtain the hexane (GS1, 47.0 g), CH2Cl2 (GS2, 60.0 g), EtOAc extracts (GS3, 27.0 g) and H2O layer (GS4). GS2 was chromatographed on a silica gel column eluting with gradient solvent of hexane: acetone (40:1, 20:1, 10:1, 5:1, 1:1, and 0:1, v/v) to give seven fractions, GS2A-GS2G. Fraction GS2G was chromatographed on an RP-18 column eluting with MeOH:H2O (4:1, v/v) to give 4 smaller fractions, GS2G1-GS2G4. GS2G4 was chromatographed on a J’sphere H-80 column (150 × 20 mm), solvent condition of 35% CH3CN in H2O to give to yield
Gymsyloside F (1)
White amorphous powder;
Gymsyloside G (2)
White amorphous powder;
Acid hydrolysis of compounds 1 and 2
Each compound (
α-Glucosidase inhibitory assay
The α-glucosidase (G0660-750UN, Sigma-Aldrich, St. Louis, MO) enzyme inhibition assay was performed according to the previously described method. 25 The sample solution (2 ml dissolved in dimethyl sulfoxide (DMSO) and 0.5 U/mL α-glucosidase (40 ml) were mixed in 120 ml of 0.1 M phosphate buffer (pH 7.0). After 5 minutes pre-incubation, 5 mM p-nitrophenyl-α-D-glucopyranoside solution (40 ml) was added, and the solution was incubated at 37 °C for 30 minutes. The absorbance of released 4-nitrophenol was measured at 405 nm by using a microplate reader (Molecular Devices, Sunnyvale, CA).
α-Amylase inhibitory assay
The α-amylase (A8220, Sigma-Aldrich, St. Louis, MO) enzyme inhibitory activity was measured using the reported method. 25 Substrate was prepared by boiling 100 mg potato starch in 5 ml phosphate buffer (pH 7.0) for 5 minutes, then cooling to room temperature. The samples (2 ml dissolved in DMSO) and substrate (50 ml) were mixed in 30 ml of 0.1 M phosphate buffer (pH 7.0). After 5 minutes pre-incubation, 5 mg/mL α-amylase solution (20 ml) was added, and the solution was incubated at 37 °C for 15 minutes. The reaction was stopped by adding 50 ml 1 M HCl and then 50 ml iodine solution was added. The absorbances were measured at 650 nm by a microplate reader.
Conclusions
Two new pregnane glycosides, gymsyloside F (
Supplemental Material
Online supplementary file 1 - Supplemental material for Gymsyloside F and Gymsyloside G, Two New Pregnane Glycosides From the Leaves of Gymnema sylvestre and Their α-Glucosidase and α-Amylase Inhibitory Activities
Supplemental material, Online supplementary file 1, for Gymsyloside F and Gymsyloside G, Two New Pregnane Glycosides From the Leaves of Gymnema sylvestre and Their α-Glucosidase and α-Amylase Inhibitory Activities by Pham Hai Yen, Duong Thi Hai Yen, Nguyen Thi Viet Thanh, Nguyen Anh Hung, Ngo Anh Bang, Bui Huu Tai, Nguyen Xuan Nhiem and Phan Van Kiem in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Vietnam Academy of Science and Technology under grant number: UDSXTN.04/20-21.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
