Abstract
Biofilm formation is considered as a crucial factor in various oral diseases, such as dental caries. The quorum sensing (QS) signaling system was proved to have a crucial role in the microbial dental plaque biofilm formation of Streptococcus mutans (S. mutans). LuxS was critical to regulating the QS system and survival of the bacterium, and, therefore, compounds which target LuxS may be a potential therapy for dental caries. The binding activities of 37,170 natural compounds to LuxS were virtually screened in this study. Baicalein and paeonol were chosen for further research of the binding mode and ΔG values with LuxS. Both baicalein and paeonol inhibited the biofilm formation without influence on the growth of S. mutans. Baicalein also distinctly reduced the production of both rhamnolipids and acids. The results provide us with a new approach to combat dental caries instead of the traditional use of antibacterial chemicals.
Dental caries is one of the most prevalent and costly bacterial biofilm (plaque) associated oral infectious diseases. 1,2 Streptococcus mutans (S. mutans) has been reported as the principal etiological agent of dental cavities and a common inhabitant of dental plaque. 3 Cariogenic biofilms are composed of a solid polysaccharide polymeric matrix synthesized by S. mutans, along with other micro-organisms. These biofilms are crucial for the survival of S. mutans, and result in dental caries. The virulence factors (eg, rhamnolipids, acids) released from the bacteria are restricted to the biofilms and consistently erode the dental enamel. 4 What is more, the biofilms also strengthen the drug resistance of the bacteria to antibiotics through several hypothetical mechanisms, such as the physical barrier preventing the antibacterial drugs from approaching the bacteria. 5 -7 It was reported that the dose of some antibacterial agents has to be raised 250 times for against the bacteria with biofilms in order to have the same inhibition ability against the same strains grown planktonically. 8 Though antibiotics are very effective in preventing dental caries in vivo and in vitro, their abuse attributes to alterations of the oral flora. 9 Therefore, inhibiting biofilm formation seems to be an effective therapy for dental caries, since it reduces the virulence and enhances the sensitivity of bacteria to antibiotics.
It has been well studied that the formation of bacterial biofilms is regulated by the quorum sensing (QS) system, through which bacteria synchronize their behavior by a series of signal molecules in a cell density dependent manner. 10,11 The QS system regulates various pathogenic events of the bacteria such as biofilm formation and virulence factor production. 12 Among the signal pathways of the bacterial QS system, S-ribosylhomocysteinase (LuxS) greatly attracts our attention as a key role in regulating the communication of S. mutans and several other genera of oral Gram-positive and Gram-negative bacteria. LuxS is responsible for the synthesis of autoinducer-2 (AI-2), which is the most broadly distributed interspecies signaling molecule known in the bacterial QS system, and which finally induces biofilm formation, as shown in Figure 1. 13 Removal of the LuxS gene from S. mutans substantially reduced biofilm formation. 14 -16 Furthermore, LuxS is also a conserved enzyme involved in the activated methyl cycle (AMC), while AMC is an important metabolic process in bacterial methionine metabolism. 17 Therefore, compounds which repress LuxS may also induce a bactericidal effect. Since LuxS is a crucial enzyme in both S. mutans metabolism and QS regulation, it is an attractive target for anti S. mutans drug development and dental caries treatment.

Roles of LuxS in S. mutans’s quorum sensing signaling circuit.
Traditional Chinese medicines (TCMs) and natural products derived from them have been proved to be effective in controlling oral bacteria and have potential for dental caries treatment. 9,18 -20 For example, 40 different TCMs have been evaluated for their antimicrobial activity against 4 common oral bacteria in 2 different studies, and 4 of them showed S. mutans inhibitory activity. 18,19 More recently, numerous TCMs, such as Houpu (Magnolia officinalis), Huangqin (Scutellaria baicalensis), Spatholobus suberectus, Zingiber officinale and Piper betle have been confirmed to have remarkable inhibitory effects on the growth of S. mutans. 21 -24 More interestingly, some TCM constituents have been proved to be effective in attenuating biofilm production through regulating the QS system. 18,21 Therefore, we anchored our hope on finding a LuxS inhibitor in TCMs that could be applied to preventing and curing dental plaques. Considering the advantage of our laboratory in the study of natural products and previous studies on QS inhibitors, 25,26 the TCM database was used for virtual screening to find potential compounds targeting the QS system of S. mutans. Moreover, the biological activities of the 2 chosen compounds, baicalein (5,6,7-trihydroxy-2-phenyl-4H-chromen-4-one) and paeonol (1-(2-hydroxy-4-methoxyphenyl)ethan-1-one), were also evaluated in vitro.
Materials and Methods
Virtual Screening and Molecular Docking
Virtual screening based on molecular docking and pharmacophore modeling has been widely used as an important tool in finding new small molecules for targeting proteins. In this research, TCM@Taiwan in Zinc database, which contains 37,170 (32,364 non-duplicate) TCM natural products from 352 TCM ingredients, was introduced to the screening. 27 Surflex-Dock in SYBYL 8.1 was employed to study the molecular docking. 28 The crystal structure of LuxS was retrieved from the RCSB Protein Data Bank (PDB entry: 1JVI). All ligands and water molecules were removed. The polar hydrogen atoms and Gasteiger charges were added. Protomol, a computational representation of the receptor’s binding cavity to which putative ligands are aligned, was automatically generated with a threshold parameter of 0.5 and a bloat parameter of 1 Å, and composed of a collection of fragments or probe molecules such as CH4, N-H, and C = O that characterize steric effects, hydrogen bond donors and acceptor groups in the binding pocket. Using the previously optimized parameters, the database was docked into the binding pocket. Prior to the screening procedure, a decoy set of 1000 compounds built with DUD tools was introduced to validate the docking method, aiming to verify the ability to distinguish the active and inactive molecules in the database. The enrichment factor and goodness of hit (GH score) were calculated to examine our docking protocol. As shown in Table 1, the GH score was 0.689, which indicated that the screening method is reliable. In addition, the percentage yield of actives, percent ratio of actives, enrichment factor (EF), false negatives, false positives, and goodness of hit score (GH) were also calculated. In the first step of screening, 3823 compounds whose total scores were higher than 6 were saved, and a Gemo mode docking was then applied to refine the results. The top 500 compounds were retained and used for cluster analysis to reduce the analogs in order to keep the diversity of the hits. The top 100 compound clusters (listed in Supplemental Table S1) in the first-round docking were used for high precision docking in GemoX mode. The total scores are expressed in -log10 (K d) to represent binding affinities. The polar score, which reflects the effects of hydrogen bonds and salt bridges, is the overall sum of all pairs of complementary polar atoms. Crash is the degree of inappropriate penetration into the protein, as well as the degree of internal self-clashing of the ligand. Additionally, CScore integrates the G score, D score, PMF score and ChemScore functions for ranking the affinity of ligands bound to the active site of the receptor. 29
Statistical Parameters of the Docking Protocol.

aEF = (Ha/Ht)/(A/D)
bGH = (Ha(3A+Ht)/4HtA)(1-(Ht-Ha)/(D-A))
Binding Free Energy Calculation
The binding conformations with the highest score were subjected to molecular docking simulations in AMBER 16.0 molecular dynamics package. 27,30 In an explicit solvent, molecular docking is 10 ns. Two parameters determining the extent of protomol, and threshold and bloat parameters were 0.5 and 1 Å, respectively. The force field ff14SB was also loaded to the receptor. The truncated octahedral water box by the TIP3P water model was chosen to encompass the complexes before minimization and molecular docking. After the procedure run, analysis was performed with the cpptraj analysis tool based on the last 2ns trajectories of the simulation. The binding free energies of complexes were calculated using MM-PBSA. 31
Bacterial Strain and Culture
S. mutans, isolated and kept by the Third Affiliated Hospital, Jinan University, was stored at −80 °C with 50% of glycerol. LB (Luria-Bertani) liquid medium was prepared according to the manufacturer’s instructions. The bacterium was recovered and inoculated into 5 ml of sterilized trypticase soy broth (TSB), and cultured at 37 °C for 16‐18 h.
Detection of MIC
Minimum inhibitory concentration (MIC) was determined by the double broth dilution method, according to the guidelines of the Clinical and Laboratory Standards Institute (CLSI). Briefly, a stock solution of compounds (baicalein, paeonol and (5Z)−4-bromo-5-(bromomethylene)−2(5 hours)-furanone (C-30)) was prepared in dimethylsulfoxide (DMSO) and serial of solutions at different concentrations (512, 256, 128, 64, 32, 16, 8, and 4 µg/mL) were obtained by diluting with LB medium in a 96-well polystyrene plate. Equal volumes (50 µL/well) of S. mutans culture (2 × 106 CFU/mL), which had been sub-cultured to the early logarithmic phase (OD600 nm=1.0), were then seeded. The solutions were mixed on a microplate shaker (Eppendorf, Hamburg, Germany) at 900 rpm for 1 minute and then cultured at 37 °C for 24 hours. Compound C-30 was used as the positive control and vehicle (medium without drugs) as the negative control. The MIC value was determined as the lowest concentration where inhibition of bacterial growth was observed by the microplate reader.
Evaluation of Inhibitory Effects on S. Mutans Biofilm Formation
S. mutans was cultured in LB medium overnight at 37 °C and the effects on biofilm formation were determined in 96-well plates. A standardized inoculum (5 µL of a 1‐5 × 105 CFU/mL suspension) was inoculated with 100 µL of fresh LB medium in either the presence or absence (non-treated control) of compounds at different concentrations (16 and 8 µg/mL). Following 24 hours of incubation, non-adherent bacteria were removed by washing with sterile phosphate buffer saline (PBS, pH 7.2). Biofilms were stained with 1% crystal violet solution and the absorbance at 570 nm was measured to evaluate the biofilm formation. The inhibition of biofilm formation was calculated from the formula: Inhibition% = [(ODnegative control)-ODx) ⁄ ODnegative control] ×100%, where x refers to the tested compounds. The average inhibition of 3 trials was calculated for statistical analysis.
Biofilm Formation Detection
Scanning electron microscopy (SEM) experiments were carried out using an ultra-high 165 resolution Hitachi S-4800 SEM (Tokyo, Japan). All chemicals were purchased from commercial sources (Aladdin Chemicals, Shanghai, China) and used without further purification. Briefly, the S. mutans was diluted with LB broth to 0.5 MCF (McFarland standard). Test compounds were added to the culture media to a final concentration of 64 µg/mL. After the polyvinyl chloride catheter (0.5 cm × 0.5 cm) had been set on the bottom of each tube, bacteria were cultured at 37 °C for 7 days. The medium solution was removed and the catheter washed twice with PBS buffer, and then the bacteria on the catheter were fixed with 2.5% glutaraldehyde/cacodylate (v/v) solution for 5 hours. Afterwards, the samples were dehydrated with ethanol using the follow procedure: 50%, 75%, 80%, 90%, and anhydrous ethanol for 10 minutes, respectively, and isoamyl acetate for another 10 minutes. Samples were frozen at −65 °C and freeze dried at −53 °C, and then covered with gold prior to SEM measurement.
Rhamnolipids Detection
Rhamnolipids production was quantified using a previously published procedure, but with some modification. 32 An overnight culture of S. mutans was diluted into fresh LB medium (OD600 = 0.1). Baicalein and paeonol were added to the culture medium with a final concentration of 2 µg/mL and cultured for 48 hours at 37 °C. DMSO at the same concentration was used as the blank and chlorhexidine as the control. After that, the bacterial culture media were centrifuged at 3000 rpm for 10 minutes. Two milliliter of supernatant was removed using a pipette and extracted with 2 ml of diethyl ether, twice. The organic phase was collected and evaporated to afford a white solid. The solid was dissolved in 500 µL of deionized water with vigorous shaking in an 80 °C water bath. Twenty microliter of solution was removed by pipette and added into 180 µL of orcinol solution (prepared with 0.19% orcinol and 50% sulfuric acid), and incubated at 80 °C for 30 minutes. After cooling to room temperature, the OD421 was measured. The concentrations of rhamnose derived from the hydrolysis of rhamnolipids were calculated by comparing with rhamnose standards.
Acidogenicity Evaluation
The effect of compounds on bacterial acidogenicity was evaluated by changes of pH. The pH drop was measured using a published method, but with minor modification. 33 An overnight culture of S. mutans was diluted into fresh LB medium (OD600 = 0.1). Baicalein and paeonol at different concentrations (0.5, 1 and 2 µg/mL) were added to the medium. S. mutans was co-cultured with glucose at a final concentration of 1% (w/v) for 24 hours at 37 °C. DMSO was used as the blank control and chlorhexidine as the positive control. The pH of the supernatant was recorded by an electronic pH detector before and after the incubation. Changes in pH were calculated using the formula: ΔpH = pHt-pH0, where pHt is the pH after incubation and pH0 is that at the initiation of incubation.
Statistical Analysis
Data were analyzed using Graphpad 8.0 and expressed as the means ± SEM. One-way ANOVA followed by Tukey-Kramer post hoc tests and two-way ANOVA followed by Dunnett’s multiple comparisons test were used to evaluate statistical differences. The value of statistical significance was set at P < 0.05.
Results and Discussion
Virtual Screening and Molecular Docking
The top 100 compounds of the virtual screening are listed in Supplemental Table S1. As the main effective components of Radix scutellariae and Paeonia suffruticosa, baicalein and paeonol, respectively, were more easily to extract, purify and procure than the other 8 of the top 10 compounds (listed in Table 2), they were chosen for further molecular docking considering both the affinity and drug likeness. As shown in Figure 2, baicalein formed firm interaction with LuxS receptors by H-bonds with ASP73, THR75, and THR115, while paeonol formed H-bonds with TRP60 and SER129 (Figure 3). The ΔG values calculated from the molecular docking simulation results are shown in Table 3, and the RMSD of the 2 compounds in Supplemental Figure S1. Since the ΔG of baicalein is lower than that of paeonol, baicalein may bind more tightly to LuxS than paeonol, which means baicalein is more effective than paeonol.

Binding mode of baicalein with LuxS receptor. (A) Residue interaction of the binding pocket; (B) Electrostatic surface of binding pocket.

Binding mode of paeonol with LuxS receptor. (A) Residue interaction of the binding pocket; (B) Electrostatic surface ofbinding pocket.
Top 10 Compounds in the Virtual Screening.
Estimated ΔG in MM-PBSA Calculations.

The QS system is an intercellular cell-cell communication mechanism that controls the expression of genes involved in a variety of cellular processes that play critical roles in the adaption and survival of bacteria. 34 Through the activation of LuxS, the bacterium synthesizes AI-2 to initiate the QS system and modulates the various cellular processes involved mainly in the regulation of virulence factors, motility and biofilm formation. 35 -37 The LuxS/AI-2 system also decreases the sensibility of the bacterium to drugs through multi-mechanisms, including promotion of the expression of drug efflux pumps, 38,39 changes in mobile genetic elements, 40 inhibition of folate synthesis 41 and facilitation of biofilm formation. 42,43 In silico study, including docking and MD simulation, showed excellent scores in binding and inhibition of LuxS by baicalein and paeonol. Both have been reported to have anti-bacterial activity. Baicalein synergizes the effects of antibiotics against oral bacteria and protects periodontal tissues from them. 44,45 Paeonol inhibits both Gram-positive and Gram-negative bacteria, 46 but the mechanisms are not clear. The virtual screening results revealed that baicalein inhibited the bacteria maybe related with binding to LuxS and regulating the QS system.
MIC of Compounds
After the screening and validation in silico, we firstly measured the minimum inhibitory concentrations (MIC) of baicalein and paeonol against S. mutans (Table 4). C-30 was used as the positive control, since it displays an enhanced antagonistic activity in the QS systems and does not affect growth of the planktonic cultures. 47 The MIC of C-30 against S. mutans was 64 µg/mL, and that of paeonol and baicalein was 256 µg/mL. These data indicate that neither paeonol nor baicalein significantly affected the growth of S. mutans when the concentration was below 256 µg/mL.
The MIC of Compounds.

Biofilm Inhibitory Effect of Compounds on S. Mutans
The activity of the compounds on biofilm formation against S. mutans was assayed in vitro. As illustrated in Table 5, all 3 compounds showed moderate inhibitory effects (25%) at a concentration of 8 µg/mL. At 16 µg/mL, baicalein showed 47.5% inhibition, while that of C-30 was 42.0%, which means that the biofilm inhibition of S. mutans was superior to that of C-30.
Inhibition Effects of Compounds on S. Mutans Biofilm Formation.

The effect of baicalein on biofilm formation was further confirmed by SEM. Compared with the blank (Figure 4(A), there was a thick layer of exopolysaccharide surrounding each cell), incubation with baicalein at sub-MIC (64 µg/mL) resulted in significant reduction of the biofilm, but without significant bacterial death (Figure 4(B)). Baicalein dramatically decreased the thickness of the biofilm. A similar effect was also observed for paeonol (data not shown).

SEM images of the biofilm formation of S. Mutans. (A) blank; (B) baicalein.
As baicalein and paeonol were effective in inhibiting biofilm formation, we wondered whether the biofilm inhibition derived from bacterial growth suppression. Thus, the effects of compounds on bacterial growth were measured. As illustrated in Supplemental Figure S2, both baicalein and paeonol did not significantly affect the growth of S. mutans at a concentration of 16 µg/mL. This result suggested that the biofilm inhibitory activity offered by paeonol and baicalein was not based on the growth inhibition effect of S. mutans. We further evaluated their effects on LuxS expression (Supplemental Figure S3). Compared with the control, both baicalein and paeonol significantly reduced LuxS in a concentration dependent manner, which means that the compounds which targeted LuxS also inhibited the QS of S. mutans. Considered the MIC of both paeonol and baicalein were high (256 µg/mL), the remarkable inhibitory effect on biofilm formation at low concentrations (1/32 and 1/16 of MIC) illustrated that both of the 2 compounds significantly inhibit biofilm formation without suppressing the regular life cycle of S. mutans, thus reducing the risk of drug resistance.
Effects on Rhamnolipids and Acid Production of S. Mutans
The impressive biofilm inhibitory activity of paeonol and baicalein encouraged us to evaluate further dental application of these compounds. Notably, some virulence factors such as rhamnolipids and acids were more actively catabolized by the attached bacteria compared to planktonic cells. Additionally, we chose rhamnolipids and acid production as targets to test the repressing effect of compounds on virulence factors, which were also important factors in dental caries. As shown in Figure 5, rhamnose, the hydrolyzed compound of rhamnolipids, was distinctly reduced in the baicalein treated group, from 450 μg/mL to 320 μg/mL, while paeonol showed no effect. The effect of the compounds on acidogenicity was similar to that of rhamnolipids (Figure 6). Baicalein, and the positive control, chlorhexidine, repressed the pH drop of S. mutans culture media in a concentration dependent manner. These data indicated that baicalein was effective in reducing the virulence factors of S. mutans.

Effects of baicalein and paeonol on rhamolipids production of S. Mutans. Baicalein reduced the rhamnolipids production of S. mutans. *** P < 0.001 vs blank group.
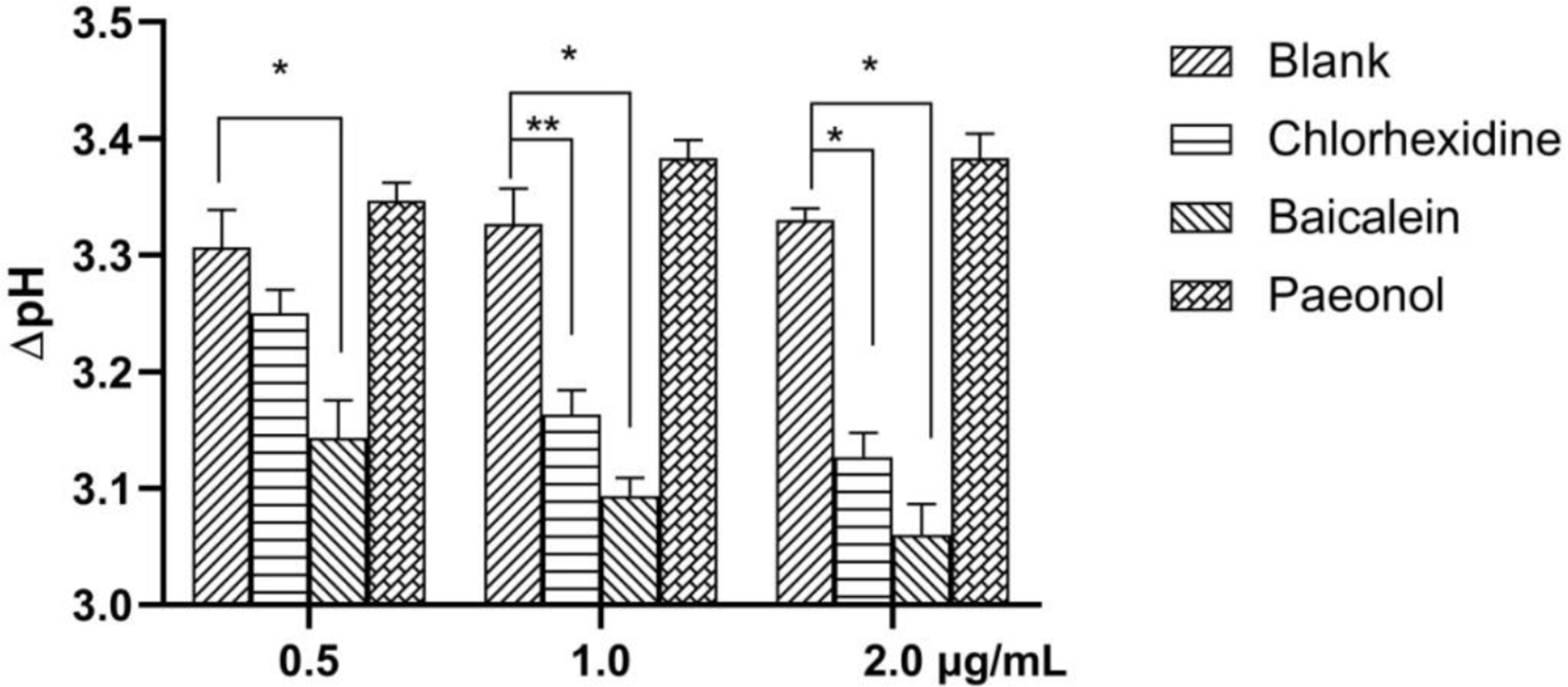
Effects of baicalein and paeonol on acidogenicity of S. Mutans. Baicalein inhibited the pH decrease of S. mutans culture medium. * P < 0.05 and ** P < 0.01 vs blank group.
Virulence factors play a fundamental role in bacterial invasion, pathopoiesia and resistance. Compared to killing the bacterium directly, attenuation of its virulence and capacity to induce disease is an effective therapeutic stratagem since it will not trigger bacterial evolution and resistance to antibiotics. 48 Rhamnolipids play many roles in bacterial growth. Firstly, they promote bacteria to assimilate and metabolize hydrophobic substrates and make it easier to colonize in comparison with other organisms. What is more, they further regulate inter-bacterial communication and the flow of nutrients by making channels and pores in the structure of mature biofilms. 49 Rhamnolipids also facilitate the detachment and dispersion of bacteria from the mature biofilm, which promotes bacterial migration. 50 Baicalein reduced rhamnolipids production, while paeonol had almost no effect. S. mutans ferments carbohydrates to increase acidification and demineralization of the tooth surface. 33 According to the crucial role of LuxS in virulence production, the difference in the effects between baicalein and paeonol in rhamnolipids and acid production is related to their different affinity to LuxS.
TCMs have increasing value since more and more of them and their individual herbs have been proved effective by modern pharmacological methods. However, to identify and predict the pharmacological basis of the activity is still a challenge needed to be resolved, in view of the complex and varied chemical constituents. 51 The results of our study and others 51 indicated that virtual screening is a potential way to find new activities of molecules and identify the pharmacological basis of TCMs.
Huangqin (Scutellaria baicalensis) is a widely used Chinese medicine with a long history of economic and medicinal value. 52 Present pharmacology research has revealed that huangqin is effective in treating diseases such as hypertension, acute respiratory infection and acute gastroenteritis. Its broad anti-microbial and anti-inflammatory effects have been well learned. 22,53 For its strong antimicrobial properties and few side effects, huangqin might be a new option to solve the increasingly serious problem of bacterial resistance. 54 However, the mechanism of huangqin on drug-resistant bacteria is not clear. Our findings give a novel insight into the possible explanation of its good activity in combating refractory infection. Furthermore, it may also explain some antimicrobial effects of the classical Huangqin Chinese toothpaste (mainly containing Scutellaria baicalensis extracts). Especially, baicalein demonstrated better activity than chlorhexidine (an antibacterial component commonly used in toothpaste) in rhamnolipids and acid reduction.
Conclusions
Through virtual screening, 2 compounds, baicalein and paeonol, out of a total of 37,170 compounds, were chosen for in vitro screening. Both showed moderate effects in inhibiting bacterial growth, with MICs of 256 µg/mL. Furthermore, compared to paeonol, baicalein was more effective in inhibiting biofilm formation, and virulence production. Thus, it can be seen that this integrated method can be an effective strategy to discover more biofilm inhibitors from TCMs. Considering its effects on biofilm inhibition, baicalein has the potential to serve in dental caries prevention and therapy against S. mutans. The results also revealed that the screening method is effective in discovering novel activities of molecules and identifying their pharmacological basis in complex TCM.
Supplemental Material
Online supplementary file 1 - Supplemental material for Virtual Screening and Biological Evaluation of Anti-Biofilm Agents Targeting LuxS in the Quorum Sensing System
Supplemental material, Online supplementary file 1, for Virtual Screening and Biological Evaluation of Anti-Biofilm Agents Targeting LuxS in the Quorum Sensing System by Zheng Liu, Lihua Zhang, Jincai Wang, Yanping Li, Yiqun Chang, Xiaoling Huang, Jun Duan, Yilong Ai, Xuxin Zeng and Jialiang Guo in Natural Product Communications
Supplemental Material
Online supplementary file 2 - Supplemental material for Virtual Screening and Biological Evaluation of Anti-Biofilm Agents Targeting LuxS in the Quorum Sensing System
Supplemental material, Online supplementary file 2, for Virtual Screening and Biological Evaluation of Anti-Biofilm Agents Targeting LuxS in the Quorum Sensing System by Zheng Liu, Lihua Zhang, Jincai Wang, Yanping Li, Yiqun Chang, Xiaoling Huang, Jun Duan, Yilong Ai, Xuxin Zeng and Jialiang Guo in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by National Natural Science Foundation of China (Grant: 81872832), the Guangdong Basic and Applied Basic Research Foundation (Grant: 2019A1515010806), Foshan Science and Technology Innovation Project (Grant: 2017AG100161 and 2015AG10015), Key Field Projects of Guangdong Universities (Intelligent Manufacturing) (2020ZDZX2057), Developmental and Applied Basic Research of Advanced Technology in Stomatology (CGQ020), the Scientific Research Projects (Characteristic Innovation) of General Universities in Guangdong Province (Grant: 2019KTSCX195) and the Initial Scientific Research Fund of Doctor in Foshan University (Grant: GG040952 and CGG07246).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
