Abstract
Asana (the heartwood of
Asana (Indian kino tree, heartwood of
An improvement in blood fluidity affects blood circulation in the microsystem, and this can lighten dull facial skin. 4,5 Thus, asana may be utilized as a skin care agent and it has been used medically for serious skin diseases. 6,7 From this point of view, we focused on the skin-whitening effect of asana as a promising candidate skin-whitening agent as it possesses skin-whitening and anti-oxidative activity and improves blood fluidity. Among the various targets for skin-whitening, tyrosinase is a major enzyme in melanin biosynthesis which catalyzes the oxidative reaction from L-tyrosine to 3,4-dihydroxyphenyl-L-alanine (L-DOPA) and from L-DOPA to dopaquinone. Dopaquinone is polymerized by oxidation to produce melanin. Thus, an agent possessing tyrosinase and displaying oxidation inhibition would be a suitable target for a skin-whitening treatment. Furthermore, inhibitory activity against melanin production in a cell line would be a suitable indicator of skin-whitening potential. Melanin is synthesized by advanced glycation end-products (AGEs) through AGEs receptors. 8 A suppressive activity against AGEs production may also be suitable to achieve a skin-whitening effect.
Based on the above, we tested the inhibitory activities of asana extract against tyrosinase, melanin production in a cell line, oxidation, and production of AGEs. In this report, the results of inhibitory assays are presented and some of the active components are discussed.
A crude extract obtained from
Inhibitory Activity Against Tyrosinase by PM-Ext.

Each value represents the mean ± SD of triplicates. Significantly different from the control group, **
The high potency of PM-ext prompted us to look for the active component, and activity-guided purification using various liquid chromatography techniques allowed us to identify a stilbene, oxyresveratrol (

Chemical structure of oxyresveratrol (1), isoliquiritigenin (2), pterostilbene (3), resveratrol (4), and piceatannol (5).
Inhibitory Activity Against Tyrosinase by Compound 1.

Each value represents the mean ± SD of triplicates. Significantly different from the control group, **
Inhibitory Activity Against Tyrosinase by Compound 2.

Each value represents the mean ± SD of triplicates. Significantly different from the control group, **
Compound
Inhibitory Activity Against Tyrosinase by Compounds 1, 3, 4, and 5.

Each value represents the mean ± SD of triplicates. Significantly different from the control group, **
Next, suppression of melanin production using a B16 melanoma cell line and PM-ext and compounds
Effects of PM-Ext on Melanin Content in Cultured B16 Murine Melanoma Cells.

Each value in melanin content represents the mean ± SD of triplicates. Significantly different from the control group, ##
Compound
Effects of Compounds 1, 3, 4, and 5 on Melanin Content in Cultured B16 Murine Melanoma Cells.
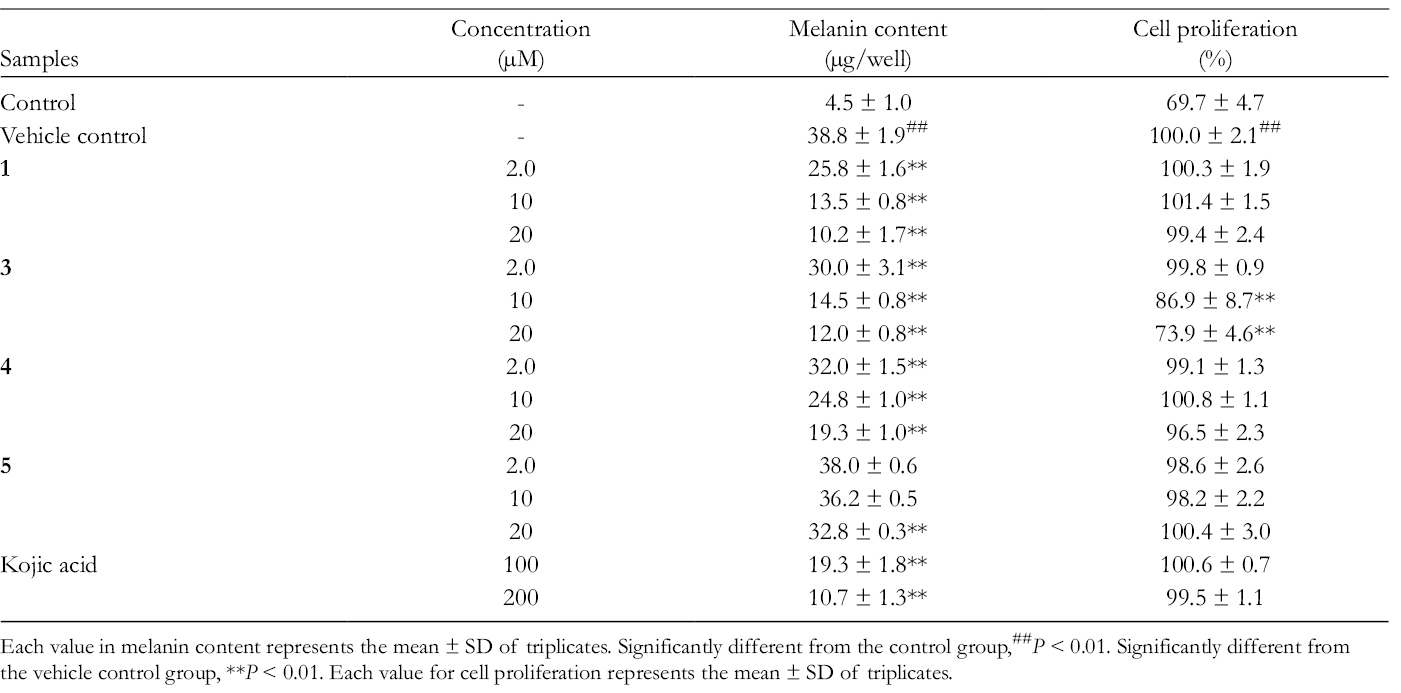
Each value in melanin content represents the mean ± SD of triplicates. Significantly different from the control group,##
Auto-oxidation is one of the major processes in melanin biosynthesis 14 and anti-oxidative activity would be a desirable trait for an anti-melanogenesis agent. We investigated the 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical-scavenging activity and suppression of AGEs production by PM-ext and 4 stilbenes.
First, DPPH radical-scavenging activity was investigated. PM-ext showed 16%, 33%, and 73% DPPH radical-scavenging activity at 10, 20, and 50 µg/mL (Table 7).
Radical-Scavenging Activity of PM-Ext.
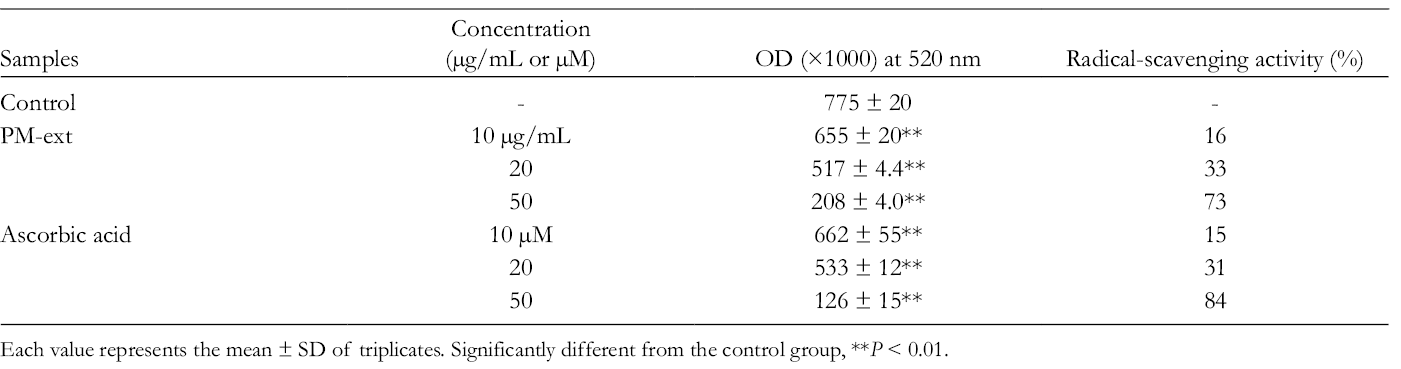
Each value represents the mean ± SD of triplicates. Significantly different from the control group, **
The DPPH radical-scavenging activity of 4 stilbenes was investigated. Compounds
Radical-Scavenging Activity of Compounds 1, 3, 4, and 5.

Each value represents the mean ± SD of triplicates. Significantly different from the control group, **
Second, suppressive activity on AGEs production was investigated. AGEs are involved in the production of reactive oxygen species and enhance melanin biosynthesis through binding to AGEs receptors. 16 For this reason, suppression of AGEs production is important to protect against skin aging.
PM-ext showed 29%, 48%, and 80% inhibition at 3.1, 13, and 50 µg/mL, respectively (Table 9).
Suppressive Activities of PM-Ext on AGEs Production.
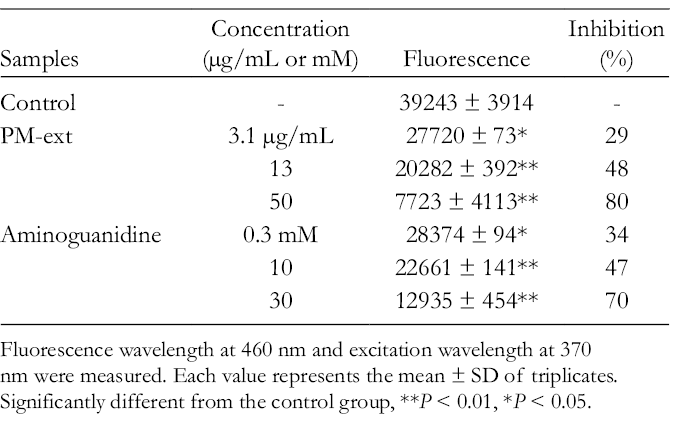
Fluorescence wavelength at 460 nm and excitation wavelength at 370 nm were measured. Each value represents the mean ± SD of triplicates. Significantly different from the control group, **
Compounds
Suppressive Activities of Compounds 1, 3, 4, and 5 on AGEs Production.

Fluorescence wavelength at 460 nm and excitation wavelength at 370 nm were measured. Each value represents the mean ± SD of triplicates. Significantly different from the control group, **
From these results, PM-ext is a promising candidate as a multifunctional skin-whitening agent; it showed anti-melanogenesis activity in a cell line as well as anti-oxidative activities. Moreover, PM-ext has both suppressive activity on AGEs production and DPPH radical-scavenging activity. These activities can lead to younger looking skin. PM-ext was recognized as an anti-aging compound from this study and is expected to be developed as a functional ingredient. Further study to determine its active components is now underway in our laboratory.
Experimental
Reagents
Reagents used in this study were purchased from Fujifilm Wako Pure Chemicals (Osaka, Japan), Nacalai Tesque (Kyoto, Japan), or Sigma-Aldrich (St Louis, MO), unless otherwise stated.
Preparation of Extracts From Heartwood of P. marsupium
Heartwood of
Tyrosinase Inhibitory Activity
Tyrosinase activity was measured according to the method described in previous reports. 17 -20 Test samples were dissolved with dimethyl sulfoxide (DMSO) and diluted with 15 mM phosphate buffer (KH2PO4 15 mM, K2HPO4 15 mM, pH 6.8) to a final DMSO concentration of 5% v/v (no effect to the assay). After incubation of 50 µL of the test solution at 25℃ for 10 minutes, 50 µL of mushroom tyrosinase (135 U/mL, Sigma-Aldrich Japan, Tokyo, Japan) and 50 µL of 0.03% DOPA solution were added to a 96-well plate. The mixture was incubated at 25℃ for 5 minutes. The amount of dopachrome in the mixture was determined based on the optical density (OD) at 475 nm using a microplate reader (TECAN, Fujifilm Wako Pure Chemicals, Osaka, Japan). Kojic acid was used as a standard agent. The inhibitory percentage of tyrosinase was calculated as follows:

where
Fractionation of PM-Ext
PM-ext (40 g) was suspended in water (600 mL) and extracted with ethyl acetate (AcOEt, 600 mL × 3) (Figure 2). The water was then extracted with
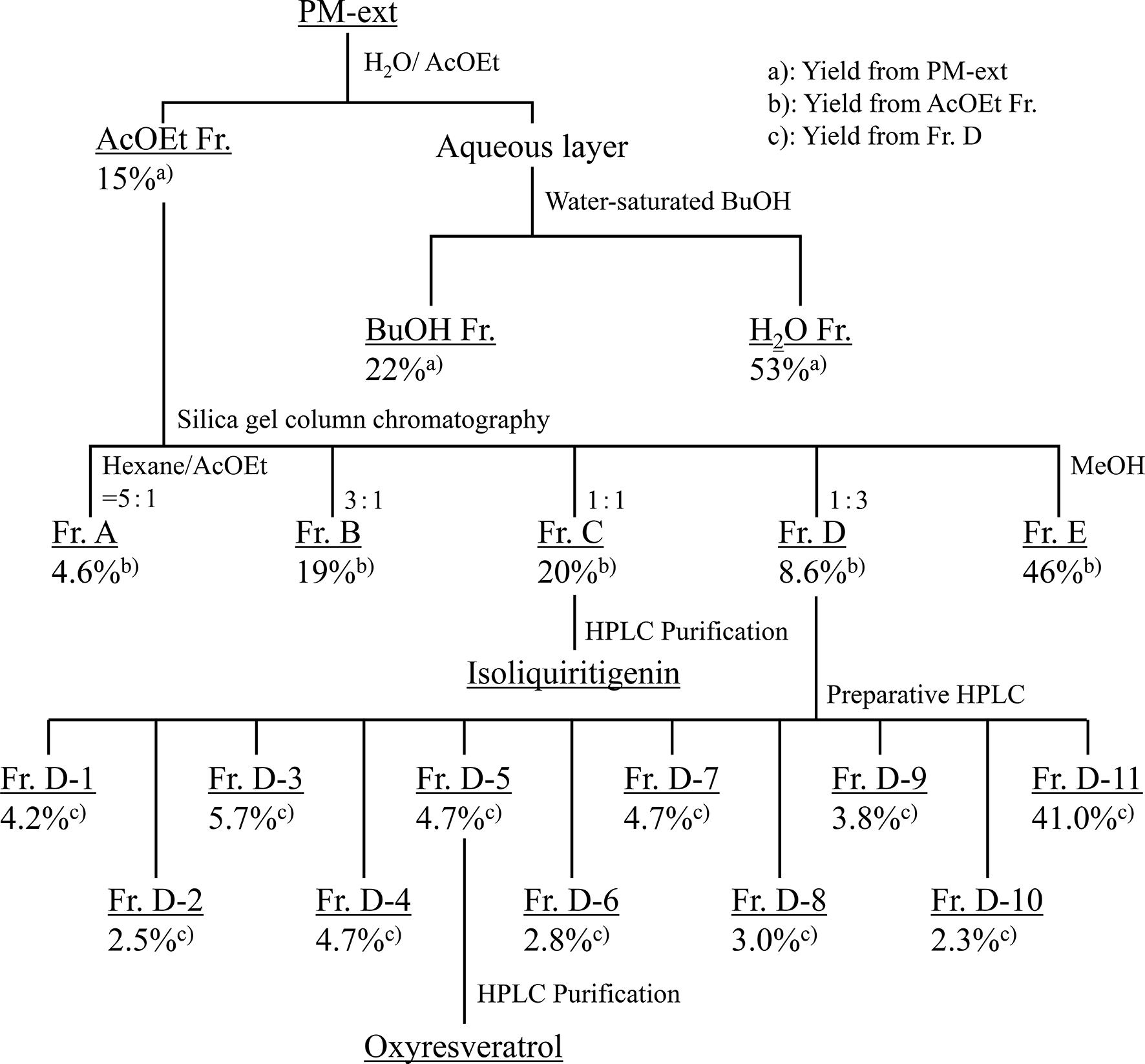
Fractionation of PM-ext.
Fr. C-3 (155.5 mg) was then subjected to preparative HPLC under the following conditions: column, Waters SunFire C18 column (19 i.d. × 250 mm); mobile phase, 35% MeCN containing 0.1% formic acid; flow rate, 10 mL/min; detection, UV 254 nm to obtain isoliquiritigenin (
Cell Culture
The B16 cell line (B16F1) was purchased from Dainippon Sumitomo Pharmaceutical Co., Ltd (Osaka, Japan) in February 2017. B16 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% (v/v) fetal bovine serum (Nichirei Biosciences Inc., Tokyo, Japan) and 1% antibiotic-antimycotic solution (a mixture of 10 000 U/mL penicillin, 10 000 µg/mL streptomycin sulfate, and 25 µg/mL amphotericin B; Invitrogen Corp., Carlsbad, CA) at 37°C in a humidified, CO2-controlled (5%) incubator.
Anti-melanogenic Assays
The amount of intracellular melanin in B16 cells was measured at the indicated times after treatment of samples according to the method of Ohguchi et al. 22 Briefly, cells (2 × 104 cells) were seeded on 24-well plates with 800 µL of DMEM with supplements and treated with test samples (100 µL) and α-melanocyte stimulating hormone (α-MSH, 100 µL) at 24 hours after seeding. Test samples were dissolved in DMSO and then diluted with DMEM to an appropriate concentration. The final concentration of DMSO was 0.1% v/v. In the control and vehicle control groups, DMSO solution was used instead of the sample solution. α-MSH was added to the medium in the vehicle control and test groups. α-MSH was dissolved in aqueous acetic acid solution (5%, v/v) and then diluted with DMEM to give a final concentration of 1 µM. Melanogenesis was initiated by the addition of α-MSH followed by incubation for 72 hours. The B16 cells were washed twice with phosphate-buffered saline (PBS: 137 mM NaCl, 2.7 mM KCl, 8.0 mM Na2HPO4, 1.5 mM KH2PO4, pH 7.4) and dissolved in 2 N NaOH for 1 hour at 65°C to extract the generated melanin. The OD at 490 nm of each sample was measured by using a microplate reader, and the melanin amount was determined by using the authentic standard of synthetic melanin. The cell proliferation of B16 cells was assessed using the WST-8 as described previously. 23 Cell proliferation is shown in percentages. Each percentage in the treated cells was calculated with reference to that of the vehicle control cells.
Radical-Scavenging Activity
Radical-scavenging activity was measured according to the method of Blois 24 with minor modifications. The test sample was dissolved with DMSO and diluted with 0.5 M acetate buffer (pH 5.5) to a final DMSO concentration of 5% v/v. A mixture of test sample solution (100 µL), EtOH (ethanol, 64 µL), 0.5 M acetate buffer (pH 5.5; 16 µL), and 1.0 mM DPPH in EtOH solution (20 µL) was allowed to stand for 30 minutes at room temperature. The OD of the resulting mixture at 520 nm was determined with a microplate reader. L-Ascorbic acid was used as a reference agent. The scavenging activity of each sample was expressed as a percentage of the decrease in OD compared with that of a control DPPH solution.
Suppression Activity of AGEs Production
Suppression activity of AGEs production was measured according to the method of Shimoda et al
25
and Itoh et al
26
with minor modifications. The test sample was dissolved with DMSO and diluted with sodium phosphate buffer (0.2 M KH2PO4, 0.2 M NaOH, pH 7.2) to a final DMSO concentration of 1% v/v. The reaction mixture of glucose (10% w/v) and bovine serum albumin (BSA, 1% w/v) dissolved in phosphate buffer (900 µL) was incubated for 48 hours at 60°C in microtube (2 mL) with or without a test solution. After incubation, the fluorescent intensity (
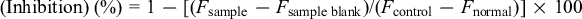
where
Calculation of IC50 Value
Data were plotted (
where
First approximate equation was calculated and
Statistical Analysis
The experimental data were statistically analyzed with Statcel 3 (Publisher: OMS, Tokorozawa, Japan), add-in software for Excel, using one-way analysis of variance. Statistical significance was analyzed with Bonferroni/Dunn’s multiple range tests.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
