Abstract
In this study, the anti-cancer activity of linalool was investigated in MCF-7 breast cancer-bearing mice. Natural killer (NK) and B cell populations in peripheral blood were studied by flow cytometry. The expressions of proliferating cell nuclear antigen (PCNA) and Ki-67 in xenograft tumors were evaluated by immunohistochemistry. Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay was performed to investigate apoptosis induction in an in vivo model. The results indicated that linalool possesses an inhibitory effect on breast cancer growth in the xenograft model. Linalool reduced B cell counts, but increased NK cell counts in mice peripheral blood. The immunosignals of PCNA and Ki-67 were significantly lower in the linalool treatment group than those of the control group. The TUNEL assay showed that linalool significantly induced apoptosis compared to the control group. The findings of this study provide insight and evidence on the antiproliferative activity of linalool on human breast cancer.
Breast cancer is the most commonly diagnosed cancer in women and the second leading cause of cancer-associated mortality. 1 Surgery and radiation therapy are local treatments for breast cancer. In contrast, the use of drugs in chemotherapy, hormone therapy and immunotherapy are considered as systemic treatments. Even though many options are available, chemotherapy is still the backbone of systemic treatment of breast cancer at different stages. However, chemotherapeutic medications can temporarily affect the blood-forming cells in bone marrow, which leads to low white blood cell counts and a high risk of infection. 2 Therefore, developing novel therapeutic strategies has become a hot topic in cancer treatment.
Herbal remedies, used to treat cancers, may help cope with the side effects of cancer treatment. Besides, natural compounds have shown promising immune-modulating potential in a tumor-bearing animal model, such as enhancing natural killer and cytolytic T-cell immunity. 3 Linalool, a terpene found in many flowering plants, is known for its antimicrobial and anti-inflammatory properties. In addition, linalool was reported to exhibit anti-cancer potential against prostate cancer, colon cancer and leukemia. 4 -6 The anti-cancer activity of linalool may be due to its apoptotic effect, oxidative stress induction and cell cycle arrest. 7 Furthermore, linalool could induce a Th1 cellular immune response by stimulating interferon-γ, interleukin (IL)−13, IL-2, IL-21, IL-21R, IL-4, IL-6sR and tumor necrosis factor (TNF)-α secretion in T-47D cells. 8
In this study, the anti-cancer activities and immunomodulatory properties of linalool in MCF-7 breast cancer cells were investigated in an in vivo xenograft model.
Results and Discussion
Animal Efficacy Study
The antitumor effect of linalool was examined in a mouse MCF-7 xenograft model. The tumor-bearing animals were treated with 100 mg/kg linalool twice a week. As shown in Table 1, xenograft tumor growth was significant suppressed on Day 28 in the linalool treatment group (495.1 ± 156.3 mm3) compared with the solvent control group (838.7 ± 280.0 mm3) (P < 0.05).
Xenograft Tumor Volumes in Control and Linalool Treatment Groups.
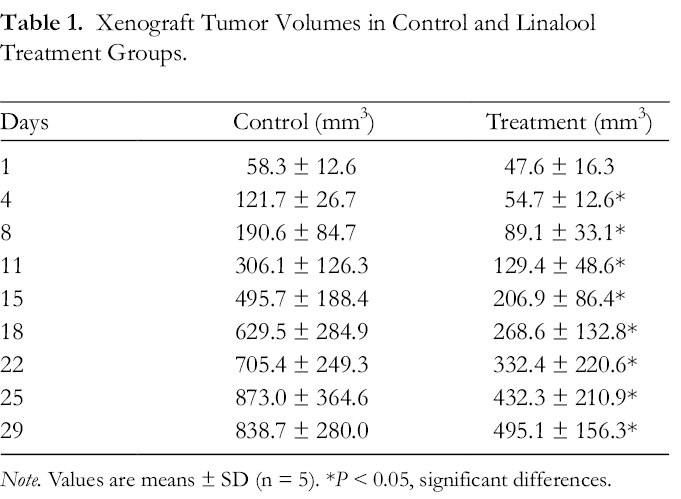
Note. Values are means ± SD (n = 5). *P < 0.05, significant differences.
NK and B Cell Population
The influence of linalool on lymphocytes was studied by the B cell and NK cell populations. On the last day of treatment, blood was collected and analyzed by flow cytometry. As indicated in Figure 1, linalool reduced B cell counts, but increased NK cell counts compared to those of the control group.

Linalool induced immune responses in MCF-7 bearing mice. (
Peripheral Blood Cytokines
The levels of TNF-α and IFN-γ in peripheral blood were detected by ELISA. After the treatment, the TNF-α level in the treatment group (24.6 ± 4.7 pg/mL) was significantly higher than that of the control group (15.4 ± 5.6 pg/mL) (P = 0.04). However, there was no statistically significant difference in IFN-γ between the treatment (11.5 ± 5.0 pg/mL) and the control group (7.8 ± 2.3 pg/mL) (P = 0.089).
Tumor Immunohistochemistry
Tumor tissues were collected after all mice were euthanized. Tumor cell proliferation indices were assessed in xenograft tumors. As shown in Figure 2, the expressions of Ki67 and PCNA were significantly lower in the treatment group compared with those of the control group (P < 0.001).

Linalool suppressed breast cancer xenograft tumor cell proliferation (Ki67 and PCNA) and induced apoptosis (TUNEL). (
Tumor Cell Apoptosis
Apoptosis rates in xenograft tumors were determined by the terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay. Positive immunosignals were observed in xenograft tumor sections. A quantitative study indicated that the apoptosis rate was significantly higher compared to the control group (Figure 2).
Linalool is a naturally occurring terpene alcohol that can be found in many herbs, spices, and fruits. Linalool exhibits cytotoxic activity against different types of cancer cells by cell cycle alternation, apoptosis induction and immunomodulation. The compound holds great promise for being used as an active anti-cancer agent. 7
In this study, we have shown that linalool could inhibit MCF-7 breast cancer cell growth in a xenograft model. The expressions of both PCNA and Ki-67 were significantly lower than those of the control group. Meanwhile, the TUNEL assay showed that linalool induced apoptosis in xenograft tumors. These data are consistent with previous research that linalool could inhibit breast cancer growth and induce apoptosis. 8
In cancer patients, chemotherapy is one of the common causes of a weakened immune system that may lead to viral and bacterial infections. 9,10 NK cells play an essential role in anti-cancer immunity, and the peripheral blood NK cell count is associated with clinical outcomes in cancer patients. 11,12 In this study, we demonstrated that linalool decreased B cell counts in MCF-7 bearing mice. It is consistent with the symptoms of cancer chemotherapy. However, NK cell counts were found elevated after the linalool treatment. This is also consistent with previous studies that natural compounds can activate NK cells. 13
TNF-α and IFN-γ are the most prominent cytokines produced by NK cells. They act as promoter agents of apoptosis in tumor cells and activate other immune response cells. 14 In this study, we found that after linalool treatment, the TNF-α level was significantly higher than that of the control group. This is consistent with the NK cell counts. However, there was no statistically significant difference observed in IFN-γ levels. This may be due to nude mice having a weak immune system. The detection limit of the IFN-γ ELISA kit is 7.81 pg/mL. The IFN-γ level in the control group was very close to the detection limit.
Although additional work is needed to elucidate further the detailed mechanisms of action observed here, these data provide preliminary evidence for using linalool to treat breast cancer and other similar conditions.
Conclusions
Linalool exhibited an inhibitory effect on breast cancer growth in a xenograft model. This study indicated that the anti-tumor effect of linalool was associated with apoptosis induction and immune modulation. The obtained results provide insight and evidence on the antiproliferative activity of linalool on human breast cancer.
Materials and Methods
Materials
The MCF-7 human breast cancer cell line was obtained from the Chinese Academy of Sciences cell bank. Linalool (97% purity) was purchased from Sigma Aldrich (St. Louis, MO, USA). The in situ TUNEL cell apoptosis detection kit (KGA 702) was bought from KeyGEN BioTECH (Jiangsu, Nanjing, P.R. China), the PerCP-Cy™5.5 Hamster Anti-Mouse CD3e, PE Rat Anti-Mouse CD49b and PE Rat Anti-Mouse CD45R /B220 from BD Biosciences Pharmingen (San Diego, CA, USA), and the mouse TNF-α and IFN-γ ELISA kits (SEKM-0034 and SEKM-0031) from Solarbio Science & Technology (Beijing, China).
Efficacy Study in Nude Mice
Female Balb/c nude mice (SPF, 6 weeks) were purchased from the Department of Experimental Animals, Kunming Medical University (permit: SYXK (Dian) K2015-0002). The animals were fed on a standard commercial diet purchased from Keaoxieli Feed Co, Ltd (permit: Jing Si Zheng (2018) 06073). They were housed in the SPF animal experiment center that is specific and pathogen-free with a 12 hours light-dark cycle.
A MCF-7 cell suspension (1ⅹ107 cells in 0.1 ml PBS) was inoculated subcutaneously into the rear flanks of the mice. Once xenograft tumors were palpable (~50 mm3), the animals were randomly divided into 2 groups (5 mice/group) and treated with either a solvent control (15% polyethylene glycol 400) or linalool at a dose of 100 mg/kg body weight twice a week for 4 weeks via subcutaneous injection. 7 Tumor growths were measured twice a week with calipers, and the tumor volume was calculated by the following formula: Length ⅹ width ⅹ height ⅹ 0.5236. Animals were euthanized by cervical dislocation once the xenograft tumor in control groups reached the limit of tumor size (1,500 mm3).
NK and B Cell Population
Each mouse was anesthetized before the procedure. After eyeball removal, blood was collected in an EDTA-coated microcentrifuge tube. For natural killer (NK) cell population analysis, 0.5 µg CD3e-PerCP-CY5.5 and 0.5 µg CD49b-PE were added to 50 µl anticoagulated whole blood. For B cell analysis, 0.5 µg CD3e-PerCP-CY5.5 and CD45R-PE were added to a 50 µl blood sample. After 15 minutes incubation at room temperature, 1.2 ml red blood cell lysis buffer was added. Subsequently, NK cells (CD3-CD49b+) and B cells (CD3-CD45R+) were quantified by a flow cytometer (CyFlow Space; Sysmex Partec GmbH) and analyzed by FloMax cell cycle analysis software, version 2.82 (Quantum Analysis GmbH).
Peripheral Blood Cytokines
The whole blood was clotted at room temperature for 30 minutes, and then centrifuged at 1,000 rpm for 10 minutes. The levels of TNF-α and IFN-γ in the serum were tested by ELISA kits. Samples were prepared according to the manufacturer’s instructions. Briefly, the plates were washed 3 times. In each well, 100 µL of either standard solution or serum sample was added, and the plates were incubated for 90 minutes at 37 ℃. After the plates had been washed 4 times, 100 µL biotinylated antibody was added to each well, and the plates were incubated for 60 minutes at 37 ℃. After the plates had been washed 4 times, 100 µL enzyme reagent was added to each sample, and the plates were incubated for 30 minutes at 37 ℃. The plates were then washed for another 5 times before 100 µL of TMB substrate solution was added to each well, and the plates incubated for 15 minutes at 37 ℃. Finally, 50 µL stop solution was added to each well, and the plates were read at 450 nm.
Tumor Immunohistochemistry
After the last treatment (on day 28), all mice were euthanized, and tumor tissues were collected. The tumor was washed with PBS, fixed in 10% neutral formalin and embedded in paraffin. After antigen retrieval, the slides were incubated with primary antibodies, Ki-67 (1:250; clone H-300; cat. no. sc-15402; Santa Cruz Biotechnology, Inc.) and proliferating cell nuclear antigen (PCNA; 1:250; clone PC10; cat. no. sc-56; Santa Cruz Biotechnology, Inc.), for 1 hours at room temperature. Then, the slides were washed and incubated with a secondary antibody for 30 minutes. The sections were visualized using a DAKO LSAB detection system. The immunosignal quantification was performed with the following formula: (positive cell number/total cell number) ⅹ 100%.
Apoptosis in Xenograft Tumors
The TUNEL assay was used to detect apoptotic cell death in xenograft tumors. Slides were prepared according to the manufacturer’s instructions. The positive rate was calculated as follows: (apoptotic cell number/total cell number) ⅹ 100%.
Statistical Analysis
GraphPad Prism 5 software (GraphPad Software, Inc.) was used for statistical analysis. t-Test was used for comparisons between two means. P ≤ 0.05 was considered to indicate a statistically significant difference.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 32060226); and the Seventh Batch of Yunnan Specialty Plant Polysaccharide Engineering Research Center Construction Plan [No. (2019)-57].
