Abstract
A new compound, 1,3,7,9-tetrahydroxy-6H-dibenzo[b,d]pyran-6-one (
Centella asiatica (L.) Urban, a stoloniferous, perennial, medicinal plant belonging to the family Umbelliferae, is well known as “Ji-Xue-Cao” and “Gotu kola” in China and the United States, respectively. This plant is widely distributed in tropical and subtropical regions such as India, Malaysia, Australia, Madagascar, Japan, and most areas of China. 1 Centella asiatica has been traditionally used to treat diverse diseases in China, India, and Madagascar. 1 In India, the species is used to treat dermatitis, diabetes, cough, cataract, and other eye troubles. 2 For Malays, C. asiatica is used in the treatments of bronchitis, asthma, gastric catarrh, dysentery, kidney trouble, urethritis, and dropsy. 3,4 Centella asiatica is widely cultivated as an edible vegetable and used as a dietary supplement in some countries, which indicates that it possesses low toxicity and has potential for an in-depth investigation. 5 Previous phytochemical studies of C. asiatica have resulted in the isolation of alkaloids, triterpenoids, tannin, flavonoids, triterpenoid, and flavonoid glycosides. 6,7 Some of these compounds exhibited significant antitumor, anti-inflammatory, antioxidant, antimicrobial, and other activities. 8,9
As part of our continuous effort for discovering bioactive compounds from C. asiatica, a new compound, 1,3,7,9-tetrahydroxy-6H-dibenzo[b,d]pyran-6-one (
Results and Discussion
Compound
Nuclear Magnetic Resonance Spectroscopic Data of Compound
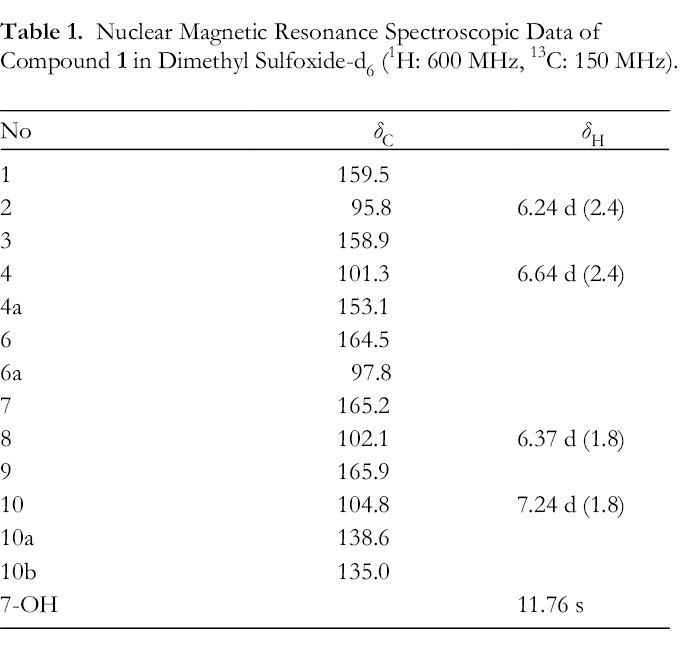

Key heteronuclear multiple bond correlation of compound 1.
Six known compounds (

The structures of compounds 1-
The in vitro cytotoxic effects of compounds
Cytotoxicities of Compounds 1-
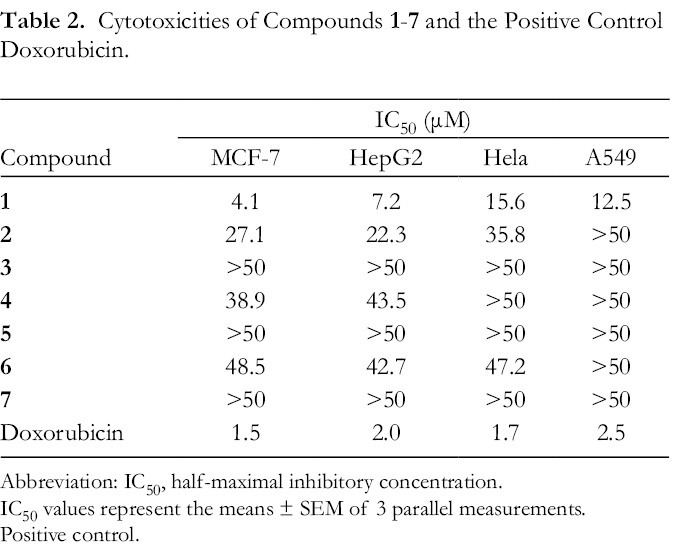
Abbreviation: IC50, half-maximal inhibitory concentration.
IC50 values represent the means ± SEM of 3 parallel measurements.
Positive control.
Experimental
General
UV spectra were recorded in methanol (MeOH) using a Shimadzu UV-2401PC spectrophotometer (Shimadzu, Tokyo, Japan), IR spectra with a JASCO FT-IR 620 spectrophotometer (JASCO, Tokyo, Japan), and NMR spectra with a Bruker Avance III 600 MHz spectrometer with tetramethylsilane as an internal standard (Bruker, Karlsruhe, Germany). HREIMS were obtained using a Bruker micro-TOF-Q mass spectrometer. Column chromatography was conducted using silica gel 60 (200 μm particle size, Yantai Xinde Chemical Co., Ltd, Yantai, China) and RP-18 (150, 63 μm particle size, Merck, Darmstadt, Germany). Thin-layer chromatography was performed with precoated silica gel GF254 glass plates (Qingdao Marine Chemical Co., Ltd). A Waters 2535 high-performance liquid chromatography (HPLC) fitted with a 2998 Photodiode Array Detector and 2707 Autosampler (Waters) was used for the semipreparative separations.
Plant Material
Centella asiatica was collected in Dali, Yunnan Province, People’s Republic of China, and authenticated by Professor Jing Chen (College of Pharmacy, Dalian Medical University). A voucher specimen of the plant (No. 20181108) was deposited at the College of Pharmacy, Dalian Medical University, Liaoning, People’s Republic of China.
Extraction and Isolation
Dried whole plants of C. asiatica (10.0 kg) were extracted with MeOH at room temperature for 24 hours, and the solution was concentrated to obtain a crude extract (1.7 kg). This was suspended in water (H2O) and partitioned successively with light petroleum, dichloromethane (CH2Cl2), EtOAc, and n-butanol. The EtOAc fraction displayed moderate in vitro cytotoxicity to 4 of the cancer cell lines and was, therefore, investigated.The EtOAc fraction (152.3 g) was chromatographed over a silica gel column using a gradient of CH2Cl2-MeOH (from 80:1, 50:1, 40:1, 30:1, 20:1, 10:1, 5:1, 2:1 to 0:1) and was separated into 10 fractions (Fr.1-Fr.10). Fr.5 (14.9 g) was separated into 12 fractions (Fr.5.1-Fr.5.12) by column chromatography on silica gel using a gradient of CH2Cl2-MeOH (from 100:1 to 1:1). Fr.5.4 (1.7 g) was subjected to RP-18 column chromatography using MeOH-H2O as the eluting solvent to afford 9 fractions (Fr.5.4.1-Fr.5.4.9). Fr.5.4.6 (95.3 mg) was applied to a C18 reversed-phase HPLC column and eluted with a gradient of MeOH-H2O (45:55‐55:45) at a flow rate of 3.0 mL/min over 70 minutes. This resulted in the isolation of compound
1,3,7,9-Tetrahydroxy-6H-dibenzo[b,d]pyran-6-one (1 )
Yellow amorphous powder; UV (MeOH) λmax (log ε): 210 (4.02), 231 (4.35), 236 (3.78) nm; IR (potassium bromide disc) ν max 3425, 1670, 1631, 1598, 1571 cm−1; 1H (600 MHz) and 13C NMR (150 MHz) spectral data in DMSO-d 6, see Table 1; HREIMS: m/z 260.0306 [M]+ (calcd for C13H8O6, 260.0321).
Cytotoxic Activity
The MTT assay was used to determine the cytotoxicities of compounds
Supplemental Material
Supplementary Material 1 - Supplemental material for New Antiproliferative Dibenzo-α-Pyrone From Whole Plants of Centella asiatica
Supplemental material, Supplementary Material 1, for New Antiproliferative Dibenzo-α-Pyrone From Whole Plants of Centella asiatica by Yi Liu, Wei Wang and Jian Miao in Natural Product Communications
Footnotes
Acknowledgments
We are grateful to the Department of Instrumental Analysis of Dalian Medical University for the measurement of the UV, IR, HREIMS, and NMR.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of the Second Affiliated Hospital of Dalian Medical University, China, and approved by the Administration Committee of Experimental Animal, Liaoning Province, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
