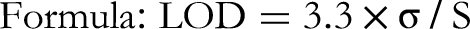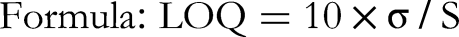Abstract
Background
Adansonia digitata (Baobab) is a nutrient-dense medicinal plant valued for its therapeutic properties, particularly its rich content of ascorbic acid. Accurate quantification of ascorbic acid is crucial for standardizing its extracts and evaluating their biological activities.
Methods
A specific and robust high-performance thin-layer chromatography (HPTLC) method was developed and validated for the quantitative determination of ascorbic acid in A. digitata fruit extract. Method validation was carried out in accordance with ICH Q2(R1) guidelines, assessing parameters such as system suitability, linearity, precision, accuracy, sensitivity (LOD and LOQ), and robustness. Additionally, its cytotoxic potential against MCF-7 breast cancer cells was evaluated.
Results
The HPTLC method demonstrated excellent linearity (R2 = 0.999) across 500–2500 µg/spot, with high precision (%RSD < 2%), accuracy (mean recovery 100.82%), and robustness. The method's sensitivity was supported by low LOD (39 µg/spot) and LOQ (500 µg/spot). Cytotoxicity analysis using MTT assay revealed dose-dependent inhibition of MCF-7 cell viability, with an IC50 of 27.90 µg/mL, suggesting moderate anticancer potential.
Conclusion
The validated HPTLC method offers a simple, precise, and reproducible approach for quantifying ascorbic acid in A. digitata fruit extract, supporting its standardization and potential application in quality control of herbal formulations and nutraceuticals.
Introduction
Adansonia digitata L., commonly referred to as the African baobab or the “Tree of Life,” is widely recognized for its exceptional nutritional and medicinal properties. The fruit is rich in bioactive constituents such as ascorbic acid (vitamin C), polyphenols, flavonoids, and antioxidants, which contribute to its immune-boosting, anti-inflammatory, and potential anticancer effects. 1 Ascorbic acid plays a vital role in human health by supporting collagen synthesis, enhancing immune response, and protecting against oxidative stress. Since humans cannot synthesize ascorbic acid endogenously, they rely on dietary sources like baobab fruit to meet their nutritional needs. 2
With the increasing interest in baobab-based nutraceuticals and pharmaceuticals, there is a growing need for reliable analytical techniques to quantify its bioactive components. High-Performance Thin-Layer Chromatography (HPTLC) offers a simple, economical, and accurate method for analysing plant-derived compounds and has been effectively used for quantifying ascorbic acid in herbal materials. 3 Thus, HPTLC serves as a valuable tool for standardizing baobab fruit powder. Adansonia digitata (baobab) was selected for breast cancer investigation based on its extensive use in traditional medicine for managing conditions associated with women's reproductive health, including breast swellings and menstrual irregularities. Its rich phytochemical composition—flavonoids, tannins, sterols, and polyphenols 4 —supports its traditional claims, as these compounds are known for their antioxidant, 5 anti-inflammatory, and anticancer potential, 6 providing a strong rationale for its scientific evaluation against breast cancer. In addition, baobab fruit extract has shown promise in anticancer research. Studies have reported that its bioactive compounds may exhibit cytotoxic effects against cancer cells. 7 To evaluate this potential, the present study employed an MTT assay using MCF-7 human breast cancer cells. The MTT assay is a standard colorimetric method that assesses cell viability by measuring mitochondrial activity, offering insight into the extract's anticancer properties.
This study aimed to develop and validate a robust HPTLC method for quantifying ascorbic acid in Adansonia digitata fruit powder, alongside evaluating its cytotoxic effects through MTT assay on MCF-7 cells. The method was optimized and validated for precision, accuracy, robustness and sensitivity, providing a reliable protocol for routine analysis. By combining nutritional profiling with cytotoxic assessment, this research not only supports the standardization of baobab fruit powder but also highlights its potential applications in cancer research and functional food development.
Materials and Method
Material and Chemical Reagents
The Baobab fruit powder was generously provided by the CSO and General Manager of Herbs and More (K) Ltd, Nairobi, Kenya. The MCF-7 cancer cell line (HTB-22) of human breast was obtained from the National Center for Cell Sciences (NCCS), Pune. Ascorbic acid standard (purity = 99.86%) was gifted by Herbs and More (K) Ltd, Nairobi, Kenya. Ethanol, methanol, ammonia, acetic acid, and HPLC-grade water used were procured from Thermo Fisher Scientific India Pvt. Ltd Additionally, MEM with high glucose (Cat No: 11965-092), FBS (Gibco, Invitrogen) (Cat No: 10270106), and Antibiotic–Antimycotic 100X solution (Thermo Fisher Scientific) were utilized in the study.
The HPTLC instrument, manufactured by Aetron, includes a Hamilton syringe with a sample applicator that operates using Spraylin software (Aetron HPTLC, Spraylin software v.2, India). Photo documentation was carried out using the Aetron IDS system, and quantification was performed with the Just TLC software. Additionally, a 96-well plate, CO2 incubator (Thermo Fisher Scientific), Biosafety cabinet (SAS Filtration Technologies Pvt. Ltd, Pune), and ELISA plate reader (Benesphera E21) was employed in the study.
Collection and Authentication of Plant
Whole plant was collected from Tirupati Hills, Andhra Pradesh, India. The plant (Adansonia digitata L.) sample (Voucher no: NPGRC/UG/AD001/2020) was authenticated by Dr P. Stayanarayana Raju, Taxonomist, Department of Botany and Microbiology, Acharya Nagarjuna University, Andhra Pradesh, India.
Preparation of the Plant Extracts
Approximately 5 g of Baobab fruit powder was weighed and placed into a 100 mL volumetric flask. The volume was adjusted to 100 mL using methanol and 1 mL of ammonia, followed by sonication for 30 min in an ultra sonicator. Ammonia was added to methanol during the extraction process to enhance the solubility of ascorbic acid, thereby improving its extraction efficiency. The resulting solution was filtered through Whatman filter paper. Subsequently, 10 mL of the filtered solution was pipetted into a 100 mL volumetric flask and diluted to the mark with methanol. Furthermore, ascorbic acid was dissolved in water instead of methanol because it is more soluble in water, which ensures better stability and consistency of the standard solution.8,9
Phytochemical Screening
Standard qualitative tests were conducted to detect the presence of alkaloids, flavonoids, tannins, phenols, glycosides, and ascorbic acid. 10
Preparation of Ascorbic Acid Standard Solution and Working Stock
Approximately 50 mg of ascorbic acid standard was accurately weighed and transferred into volumetric flask of 10 mL (5 mg/mL). The final volume was adjusted to 10 mL using distilled water. The standard concentration of 50 mg of ascorbic acid was used to ensure that the solution was concentrated enough to generate detectable and accurate results during the HPTLC analysis.
A working standard solution of ascorbic acid (100 µg/mL) was prepared by pipetting 0.2 mL of a 5 mg/mL stock solution into a 10 mL volumetric flask and diluting to volume with distilled water. The mobile phase composition and elution technique for HPTLC quantification were adapted with slight modifications from the method reported by Chakraborthy GS, which employed ethanol and acetic acid as the solvent system to achieve effective resolution of ascorbic acid. 11
The baobab fruit extract was reconstituted in distilled water to achieve concentrations of 1000, 1500, and 2000 µg/mL. These levels were selected based on the established linearity range (500-2500 µg/mL) for the HPTLC method to evaluate the method's accuracy, precision, and applicability to real sample matrices.
Chromatographic Conditions for HPTLC
The HPTLC analysis was performed using TLC silica gel 60 F254 plates as the stationary phase. The mobile phase consisted of ethanol and acetic acid in a ratio of 9.5:0.5 (v/v), which provided effective separation of the analyte. A sample volume of 10 μL was applied as 6 mm wide bands at a dosage speed of 2 μL/sec. Prior to development, the TLC chamber was saturated for 15–20 min with the mobile phase. After the development process, the plates were air-dried using a dryer. No derivatizing agent was used in this study, as ascorbic acid exhibits native UV absorbance. The developed chromatographic plates were directly visualized and analyzed under UV light at 254 nm.
Mobile Phase Optimization
To achieve optimal resolution, the mobile phase for HPTLC development was standardized to yield an Rf value within the 0.3–0.7 range, as per analytical best practices. Several ethanol: acetic acid ratios were tested, and a composition of 9.5:0.5 (v/v) was finalized as it produced a sharp and well-defined peak for ascorbic acid at an Rf ≈ 0.774, ensuring robust quantification and separation from other sample components.
Validation of HPTLC Method
The HPTLC method for ascorbic acid quantification was validated as per the International Council for Harmonisation (ICH) guidelines (ICH-Q2-R1 2005) 12 (https://www.ich.org/page/ich-guidelines) to assess system suitability, linearity, accuracy, precision, sensitivity, and robustness. 13
System Suitability
System suitability was evaluated by applying a working standard solution of ascorbic acid (100 µg/mL) on a TLC plate. A 0.3 mL aliquot was diluted to 10 mL with distilled water. Parameters such as peak area, Rf value, resolution, and symmetry factor were assessed from replicate applications (n = 6), and the data are provided in Table 1S (Supplementary file).
Linearity
Establishing a linear relationship between analyte concentration and the HPTLC signal over a specific range. 14 A linear relationship between the concentration of ascorbic acid and the corresponding HPTLC response was established over the range of 500–2500 µg/mL. A standard stock solution of 5 mg/mL ascorbic acid was prepared by dissolving the accurately weighed amount in distilled water. From this stock, aliquots of 1.0 mL, 2.0 mL, 3.0 mL, 4.0 mL, and 5.0 mL were pipetted into separate 10 mL volumetric flasks and diluted to volume with distilled water to obtain final concentrations of 500, 1000, 1500, 2000, and 2500 µg/mL, respectively. The linearity curve was constructed by plotting the peak area against the concentration of ascorbic acid.
Accuracy
Accuracy was determined using a standard addition method by spiking pre-analyzed baobab fruit extract with known quantities of standard ascorbic acid. Recovery studies were performed at three levels: 50%, 100%, and 150% of a nominal concentration of 1500 µg/spot. Specifically, concentrations of 1000, 1500, and 2500 µg/spot were prepared and analyzed in triplicate. For the accuracy study, the concentration of 1500 µg/spot was selected based on its placement within the linearity range established for ascorbic acid, ensuring reliable and reproducible measurement. This concentration represents a mid-level concentration point, making it suitable for evaluating the method's accuracy across a realistic working range. Percent recovery was calculated to evaluate the method's accuracy, which was found within the acceptable ICH range (98-102%). 15
Precision
Precision was evaluated as repeatability (intra-day) and intermediate precision (inter-day). 16 A 100 µg/mL standard working solution was prepared, and 6 mm bands (10 μL volume) were applied (n = 6) on the same day and on three consecutive days. The % RSD of the peak areas was calculated for both intra- and inter-day precision, and values were within acceptable limits (≤2%).
Sensitivity
Determining the Limit of Detection (LOD) and Limit of Quantification (LOQ) of the method, indicating how little of the analyte can be detected and quantified. The LOD indicates the least concentration of an analyte that can be detected, though it may not be quantified with consistent precision and accuracy. It serves as a measure of the method's sensitivity.
3
The LOQ refers to the smallest concentration of an analyte that can be consistently measured with satisfactory precision and accuracy, ensuring the reliability and reproducibility of the results obtained.
16
Robustness
The ability of an analytical method to remain unaffected by small, deliberate variations in method parameters, providing an indication of its reliability during normal usage. We use previously published procedure to determine the robustness.
17
We determined the following parameters to access robustness.
i. To assess the mobile phase impact, a solution of standard was prepared and applied onto TLC silica gel 60 F254. The composition of mobile phase was varied by decreasing it to ethanol: acetic acid (8.5:1.5 v/v) and increasing it to ethanol: acetic acid (9.5:0.5 v/v). The resulting effect on chromatographic separation was then evaluated. ii. To evaluate the impact of application speed, a solution of standard was spotted on TLC silica gel 60 F254 plates at varying rates. The increase in dosage speed from 2 µL/sec to 16 µL/sec and 24 µL/sec was typically performed to assess the robustness of the chromatographic method. High-speed injections (up to 24 µL/sec) are sometimes be necessary to speed up the overall process. Therefore, the dosing speed was modified to 16 µL/sec and 24 µL/sec, and the resulting chromatographic profiles were assessed to observe any changes. Same sample amount was applied when increasing the speed. iii. To evaluate the effect of band width, a standard solution was prepared and applied onto TLC silica gel 60 F254 with different band widths. The variation was set by decreasing it to 4 mm and increasing it to 8 mm. The impact of these adjustments on chromatographic resolution was then assessed.
MTT Cell Proliferation Assay
MCF-7 cells were plated in 96-well culture plates at a density of 104 cells per well. After seeding, the cells were exposed to different concentrations of the test extract (10, 40, and 100 µg/mL) to assess its effects. A standard was also included and treated at the same concentrations (10, 40, and 100 µg/mL) for comparative analysis. Control wells received 0.2% DMSO in PBS without any test compounds to serve as a baseline comparison. The plates were then incubated in triplicates under standard culture conditions (37 °C, 5% CO2) for 24 h to allow for proper cellular response. After 24 h, the medium was completely removed, and 20 μL of MTT reagent (5 mg/mL in PBS) was added to each well. The plates were further incubated for 4 h at 37 °C in CO2 incubator to allow formazan crystal formation.18–20
Formazan Formation: Metabolically active cells reduced MTT to purple-coloured formazan crystals.
Absorbance Measurement: Optical density (O.D) was recorded at 570 nm using an ELISA microplate reader (Benesphera E21). The cytotoxic effect of the test sample was determined by calculating cell viability (Eq. 1).
Statistical Analysis
All experiments were conducted in triplicate, and data were expressed as mean ± standard deviation (SD). System suitability, precision, accuracy, robustness, and other validation parameters were analyzed according to ICH guidelines (ICH Q2(R1)). Linear regression analysis was used to assess calibration curve linearity, and the correlation coefficient (R2) was calculated. For the cytotoxicity assay, percentage cell viability was calculated relative to the untreated control, and IC50 values were determined using nonlinear regression analysis. One-way analysis of variance (ANOVA), followed by Tukey's post hoc test, was applied to compare differences between groups. A p-value of less than 0.05 was considered statistically significant.
Results
Phytochemical Screening
The methanolic extract of A. digitata showed the presence of flavonoids, phenolic compounds, tannins, and a high concentration of ascorbic acid (Table 1).
Phytochemical Screening Results of Methanolic Extract of A. Digitata.

Note: ‘+’ Represent the Presence of Phytochemical.
Method Development
The HPTLC method was refined to achieve optimal separation of ascorbic acid using a mobile phase composed of ethanol and acetic acid in a 9.5:0.5 (v/v) proportion (Figure 1). This composition resulted in a well-resolved, sharp, and symmetrical peak at an Rf value of 0.774, ensuring effective separation (Figure 2). The assay was performed by applying standard and sample solutions onto TLC plates and detecting at 254 nm. Quantification was carried out by comparing the peak areas of the sample and standard (Figure 3), and the amount of ascorbic acid present in Adansonia digitata fruit powder was determined to be 282 mg/100 g.

Densitometric Scanning Image at 254 nm Highlighting the Matched Band Intensity and Position Between Standard and Extract.

HPTLC Densitogram of Ascorbic Acid Standard (Red) and Baobab Sample (Purple).

Comparative HPTLC Densitogram of Ascorbic Acid Standard (Blue and Red Peak) and A. digitata (Baobab) Fruit Extract (Green and Brown Peak), Recorded at 254 nm. The Overlapping Peaks Indicate the Presence of Ascorbic Acid in the Plant Extract, with Matching Rf Values Confirming Compound Identity.
HPTLC Method Validation
The ICH-Q2-R1 criteria were used to test the proposed ascorbic acid content determination method in a number of validation settings. By plotting ascorbic acid concentrations against its recorded TLC response, the linearity of the substance was examined. For the developed approach, the linearity of ascorbic acid was found in the 500–2500 μg/spot range. The retardation factor (Rf) and peak area were used to assess the system suitability criteria for the developed approach. The validation parameters included system suitability, linearity, accuracy, precision, sensitivity, and robustness.
System Suitability
The system suitability test confirmed that the HPTLC system was operating within acceptable limits. Six replicate applications of standard ascorbic acid (1500 µg/spot) produced consistent peak areas with an Rf range of 0.761–0.779. The average peak area was 591.83 ± 3.53 and a % RSD of 0.65%, indicating excellent system performance and reproducibility (Table 2, supplementary file, Table 1S).
HPTLC Method Validation Parameters.

Linearity
The linearity of the method was evaluated across five concentrations ranging from 500 to 2500 µg/spot (500, 1000, 1500, 2000, and 2500 µg/spot). A strong linear correlation was observed between concentration and peak area, as shown in the calibration plot (Figure 4). The regression equation was determined to be y = 0.4099x–1.8571 with a correlation coefficient (R2) of 0.999, confirming excellent linearity over the tested range (Table 2S).

The Linearity Graph of Ascorbic Acid Standard.
Accuracy
The accuracy of the method was confirmed through recovery studies conducted at 50%, 100%, and 150% levels (Table 3S). The mean recovery ranged from 99.83% to 101.35%, with an overall recovery of 100.82%, demonstrating the method's trueness and absence of interference from the matrix (Table 2).
Precision
The precision of the method was evaluated through both system (Intra-day) and method precision (Inter-day) studies. Intra-day precision (system precision) was assessed by analysing (1500 µg/spot) six replicates of the standard solution within the same day, yielding a %RSD of 0.80% (average area of 591.67). Inter-day precision (method precision) was evaluated over three consecutive days, resulting in a %RSD of 0.72% (average area of 591.33) (Tables 4S and 5S). Both values fall within the acceptable ICH threshold (≤2%), confirming the method's repeatability and intermediate precision.
Sensitivity (LOD and LOQ)
The method exhibited good sensitivity with a LOD of 39 µg/spot and a Limit of LOQ of 500 µg/spot, indicating its ability to detect and quantify low concentrations of ascorbic acid (Table 2).
Robustness
Robustness was evaluated by deliberately altering method parameters including mobile phase ratio, band width, and dosage speed. Under all tested variations, the % RSD remained below 2%, ranging from 0.24% to 1.04%, confirming the robustness and reliability of the developed method (Table 6S).
Cytotoxic Evaluation by MTT Assay
The cytotoxic potential of A. digitata extract was evaluated against MCF-7 human breast cancer cells using the MTT assay (Table 3). The extract exhibited a concentration-dependent reduction in cell viability. At 100 µg/mL, the extract reduced viability to 35.79%, compared to 58.54% at 10 µg/mL. The calculated IC50 value for the extract was 27.90 µg/mL. In contrast, the standard drug 5-Fluorouracil (5-FU) demonstrated stronger cytotoxicity, with an IC50 of 6.00 µg/mL. Tukey's post hoc test confirmed that both 5-FU and A. digitata extract significantly reduced cell viability compared to the control (p < 0.001), and 5-FU was significantly more potent than the extract (p < 0.01). This confirms that 5-FU is significantly more cytotoxic than the extract against MCF-7 cells.
Effects Against MCF7 (Cell Line of Breast Cancer) by MTT Assay.

Note: Control versus 5-FU: p < 0.0001 (***), Control versus Extract: p < 0.001 (**), 5-FU versus Extract: p < 0.01 (**).
Morphological Analysis of MCF-7 Breast Cancer Cells
Microscopic evaluation of MCF-7 cells treated with 5-FU and A. digitata extract revealed distinct differences in cellular morphology, supporting the quantitative cytotoxic findings. Cells treated with 5-FU exhibited significant morphological alterations indicative of apoptosis, including reduced cell density, irregular shapes, cytoplasmic shrinkage, and nuclear condensation. These changes are consistent with the drug's known cytotoxic efficacy and its low IC50 value (Figures 5 & 6). In contrast, cells treated with the A. digitata extract appeared more intact and aggregated, displaying relatively round morphology and well-defined nuclei with minimal signs of nuclear fragmentation. Although some apoptotic characteristics were observed, the extent was markedly less than in the 5-FU group, indicating a moderate level of cytotoxicity (Figure 6). This visual evidence complements the MTT assay data, suggesting that while the extract does exhibit anticancer potential, its cytotoxic impact is significantly milder than that of the standard chemotherapeutic agent.

Microscopic Visualization of Cytotoxic Effects on MCF-7 Cells.

Presents Comparative Morphological Analysis of MCF-7 Breast Cancer Cells Treated with 5-FU and A. digitata Extract, Likely Stained with a Nuclear dye Under Fluorescence Microscopy.
Discussion
The validated HPTLC method developed in this study for quantifying ascorbic acid in Adansonia digitata demonstrated excellent performance across multiple analytical parameters. The method exhibited strong linearity over a concentration range of 500–2500 µg/spot, with a correlation coefficient (R2) of 0.999, indicating a robust linear relationship between concentration and peak area. The precision of the method was confirmed by low % RSD values for both system precision (0.80%) and method precision (0.72%), underscoring its repeatability and reliability.
Accuracy assessments through recovery studies at 50%, 100%, and 150% levels yielded recovery rates ranging from 99.83% to 101.35%, with an overall mean recovery of 100.82%. These results align with the ICH guidelines, which stipulate that recovery rates should fall within 98%–102% for analytical methods to be considered accurate. The method's sensitivity was demonstrated by its low LOD and LOQ, determined to be 39 µg/spot and 118 µg/spot, respectively. Robustness testing, involving deliberate variations in method parameters such as mobile phase composition and band width, resulted in % RSD values below 2%, indicating the method's resilience to minor procedural changes. The developed HPTLC method for quantifying ascorbic acid in Adansonia digitata demonstrated excellent linearity across a wide range (500-2500 µg/spot), high accuracy, and good precision (%RSD <1%), comparable to the Stability-Indicating Green RP-HPTLC (SI-G-RP-HPTLC) method reported for Flibanserin, which showed slightly lower linearity (R2 = 0.9953) and accuracy (98.18%–99.58%). 21 While the SI-G-RP-HPTLC offered higher sensitivity in the ng range and used eco-friendly solvents (acetone: water), the ascorbic acid method demonstrated greater robustness and broader applicability in bioactivity assessment. Incorporating green solvent systems and optimizing the lower end of the linearity range could further enhance the analytical performance. In comparison to the HPTLC method for gallic acid, our method exhibited higher accuracy and comparable precision. The gallic acid method reported accuracy between 97.67% and 99.21%, 22 while our method slightly exceeded this range. The intra-day and inter-day precision values were slightly better in the gallic acid method, with %RSD as low as 0.26%, but the robustness and broader working range of the present method offset this minor variation. These findings confirm the suitability of present method for routine quality control and analytical quantification of ascorbic acid in complex herbal matrices.
We used a straightforward mobile phase of ethanol and acetic acid (9.5:0.5 v/v), which is easier to prepare and safer than complex mixtures like ethyl acetate–methanol–water–formic acid or n-butanol–acetic acid–water. 23 Unlike methods that require derivatization with reagents such as DCPIP for spot detection, our method detects ascorbic acid directly at 254 nm, saving both time and chemicals. The method was fully validated according to ICH Q2(R2) guidelines, showing excellent precision (% RSD below 1%), accuracy (mean recovery of 100.82%), and linearity (R2 = 0.999), whereas some other studies did not perform complete validation or reported lower recovery ranges.24,25 Overall, our method stands out as cost-effective, robust, and biologically relevant, making it ideal for both routine quality control and pharmacological screening.
Comparatively, previous studies have explored the phytochemical properties and antioxidant activities of A. digitata. 26 A. Wasihun et al 27 conducted an GC-MS analysis of aqueous extracts from various parts of A. digitata, identifying multiple phenolic compounds contributing to its antioxidant potential. However, their study did not focus on the quantification or validation of ascorbic acid specifically. In contrast, a study by A. Foudah et al 25 developed a green HPTLC method for estimating ascorbic acid in extracts of Phyllanthus emblica, Capsicum annuum, and Psidium guajava. Their method demonstrated a linearity range of 25–1200 ng/band with an R2 of 0.9956, and LOD and LOQ values of 8.63 ng/band and 25.89 ng/band, respectively. While their method exhibited higher sensitivity, it was applied to different plant matrices and utilized a different solvent system.
The HPTLC method developed for quantifying ascorbic acid in Adansonia digitata extract offers a chromatographic approach that is simple, rapid, and suitable for herbal matrices, while the electrochemical method utilizing a defects-regulated graphene ink-coated glass electrode (G-30) presents a highly sensitive and selective alternative designed for complex biological and pharmaceutical samples. 28 In comparison, the electrochemical method using a G-30 graphene electrode demonstrated higher sensitivity with a lower LOD (17.8 µM) and a good linear range (50-1000 µM; r2 = 0.9987), but exhibited higher variability with %RSD values of 2.4%–3.1% and 7.6% under environmental fluctuations. 29 Thus, the HPTLC method is more robust and suitable for routine herbal analysis, while the electrochemical approach is better suited for sensitive, low-concentration determinations in pharmaceutical matrices.
The current study focuses on A. digitata and provides novel insights into the quantification of ascorbic acid in this species, filling a gap in the existing literature. In addition to establishing a validated HPTLC method for ascorbic acid quantification, this study evaluated the anticancer potential of A. digitata extract on MCF-7 breast cancer cells. The extract demonstrated moderate cytotoxic activity, with an IC50 of 27.90 µg/mL. While this is higher (ie, less potent) than the reference chemotherapeutic agent 5-FU (IC50 = 6.00 µg/mL), the extract still showed significant dose-dependent inhibition of cell viability. These findings are consistent with the known phytochemical profile of A. digitata, which includes antioxidant and bioactive compounds such as ascorbic acid, flavonoids, and phenolics. 8 Previous narrative review of M. Silva et al, 30 have reported anticancer and apoptosis-inducing properties of baobab extracts, attributing them to phenolic and flavonoid-rich fractions. The cytotoxic activity observed here supports these findings and suggests potential therapeutic value of A. digitata in integrative cancer care or as a complementary agent.
Limitations and Future Prospects
Despite the promising results, this study has certain limitations. The HPTLC quantification focused solely on ascorbic acid, though A. digitata contains a wide array of bioactive compounds—such as flavonoids, tannins, and polyphenols—that may also contribute to its pharmacological effects. A broader phytochemical analysis using hyphenated techniques like HPTLC-MS or HPLC-DAD could provide a more comprehensive phytochemical fingerprint of the extract. Furthermore, the cytotoxic evaluation was limited to a single breast cancer cell line (MCF-7). To better understand the anticancer potential of A. digitata, further studies should include additional cancer cell lines and normal human cell lines to assess selectivity and safety. Additionally, the in vitro cytotoxicity does not directly translate to clinical efficacy; in vivo studies and mechanistic investigations are needed to confirm the therapeutic potential and elucidate the molecular pathways involved. Future studies should also focus on bioassay-guided fractionation to isolate and identify specific cytotoxic constituents. Investigating synergistic effects between ascorbic acid and other phytochemicals within the extract could enhance its efficacy and support its use as a complementary therapeutic agent.
Conclusion
The validated HPTLC method is precise, accurate, robust, and suitable for routine quantification of ascorbic acid in Adansonia digitata fruit extract. The method showed a strong linearity over the concentration range of 500–2500 µg/mL with an R2 value of 0.991, and acceptable sensitivity with LOD and LOQ values of 39 µg/spot and 118 µg/spot, respectively. Accuracy was confirmed through recovery studies at 50%, 100%, and 150% levels, yielding recoveries within the acceptable range (98.5%–101.3%). Precision studies indicated %RSD values below 2%, confirming reproducibility. The method was also robust under minor variations in chromatographic conditions. Furthermore, the cytotoxic evaluation using the MTT assay revealed a dose-dependent inhibition of MCF-7 human breast cancer cells, with the extract showing considerable activity at higher concentrations and an IC50 value of 27.90 µg/mL. Hence, this method is suitable for routine quality control of ascorbic acid in baobab-derived formulations. These findings warrant further phytochemical characterization and in vivo studies to explore therapeutic applications.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251383761 - Supplemental material for Phytochemical Fingerprinting and Cytotoxic Insight: HPTLC Quantification and Validation of Ascorbic Acid in Adansonia digitata with MCF-7 Cell Evaluation
Supplemental material, sj-docx-1-npx-10.1177_1934578X251383761 for Phytochemical Fingerprinting and Cytotoxic Insight: HPTLC Quantification and Validation of Ascorbic Acid in Adansonia digitata with MCF-7 Cell Evaluation by K. N Rajini Kanth, Rama Rao Nadendla, M. Hemanth Kumar, P. Venkata Suresh, M. Neha Chowdary, M. Honey Sharapova, P. Ravali, Sk. Shabana Parvin, Y. Lavanya, Somashekhar M. Metri and Santosh Prasad Chaudhary Kurmi in Natural Product Communications
Footnotes
Acknowledgements
The authors acknowledge the facilities rendered by Chalapathi Drug Testing Laboratory (CDTL), Andhra Pradesh, India for analytical studies and Biocyte Institute of Research and Development, Sangli, Maharashtra, India for assistance provided in Cancer cell line studies. Authors profoundly acknowledge the facilities and support rendered by management, principal, teaching and non-teaching staff of Chalapathi Institute of Pharmaceutical Sciences, Guntur, Andhra Pradesh.
Author Contribution
K. N Rajini Kanth & Rama Rao Nadendla M.: Concept, Methodology, Analysis, Original draft Writing; Hemanth Kumar, P. Venkata Suresh, M. Neha Chowdary, M. Honey Sharapova, P. Ravali, Sk. Shabana Parvin, and Y. Lavanya: Methodology, Experiment, Data Curation, Visualization; Somashekhar M. Metri and Santosh Prasad Chaudhary Kurmi: Validation, Review and Editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All the data provided within the manuscript and supplementary file.
Patient Consent Statement
This study does not involve experiments on animals or human subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.