Abstract
Objective
miRNAs as pharmaco-targets have been investigated in multifarious diseases. Our study aimed to determine whether leonurine was a potential cardioprotective agent by targeting miRNAs in hypoxia-stimulated mice and H9c2 cardiomyocytes.
Methods
Cell proliferation and apoptosis were examined by CCK-8 and TUNEL assay in hypoxia-stimulated rat H9c2 cardiomyocytes. miRNAs expression levels in cardiomyocytes in response to hypoxia stimulation were detected by RT-qPCR. Mice with myocardial injury were induced by chronic intermittent hypoxia stimulation.
Results
Leonurine alleviated hypoxia-induced cardiac hypertrophy in mice. Moreover, up-regulation of miR-31 and down-regulation of miR-210 in hypoxia-stimulated mice were reversed by leonurine administration. Leonurine exhibited cardioprotective activity in an vitro cell model of hypoxia-stimulated rat H9c2 cardiomyocytes, reflecting that the compound improved hypoxia-induced growth inhibition and apoptosis of cardiomyocytes. TUNEL assay revealed that transfection of miR-31 inhibitors or miR-210 mimics abrogated hypoxia-induced cardiomyocyte apoptosis. In contrast to that, miR-31 mimics or miR-210 inhibitors counteracted the anti-apoptotic effect of leonurine on hypoxia-treated rat H9c2 cardiomyocytes.
Conclusion
Our findings suggest that miR-31 and miR-210 as the upstream regulators of leonurine are involved in hypoxia-induced cardiomyocyte apoptosis. Leonurine can target miRNAs to protect against hypoxia-induced myocardial damage. miRNAs as potential drug targets may provide prospective therapeutic strategies for the treatment of myocardial damage.
Myocardial damage is a vicious incident in the pathophysiology of heart failure and myocardial infarction (MI). 1,2 In in vitro and in vivo experimental models of MI, myocardial damage is persistently evoked by hypoxia that can trigger mitochondrial fission, endoplasmic reticulum stress, reactive oxygen species production and apoptosis in cardiomyocytes. 3 -6 Therefore, reduction of myocardial damage by pharmacological treatment may be a potential therapeutic strategy for preventing myocardial damage.
Leonurine is a pseudoalkaloid extracted from various Leonurus species, including L. sibiricus and L. heterophyllus. 7 Leonurine possesses potent anti-inflammatory, anti-oxidative, and anti-apoptotic activity in multiple diseases. 8 -10 Leonurine performs cardioprotective effects against endotoxin, hypoxia, or doxorubicin-induced cardiomyocyte apoptosis. 11 -13 However, the leonurine-mediated molecular mechanism in hypoxia-induced rat H9c2 cardiomyocyte apoptosis has not been completely determined.
microRNAs (miRNAs) have been widely investigated as a class of non-coding RNAs with a length of about 20‐25 nucleotides. 14 Classically, miRNAs are authenticated as post-transcriptional regulators to control the translation of the target gene. 14 Accumulating evidence manifests that miRs may be the possible pharmacological targets to remedy hypoxia-induced tissue or organ injury. 15,16 Numerous miRNAs are detected in the cardiomyocytes and execute a central role in the physiopathology of cardiomyocytes. 17 For instance, overexpression of miR-23a and miR-423‐5p promote cardiomyocyte apoptosis. 18,19 miR-27a-5p and miR-532‐5p attenuate hypoxia-caused cardiomyocyte apoptosis. 20 miR-206 and miR-93‐5p alleviate ischaemia/reperfusion-induced myocardial damage in rats. 21,22 These findings suggest that miRNAs may be novel targets for blocking hypoxia-induced myocardial damage.
In our study, our results discovered that leonurine treatment is liable to activate miR-210 and block miR-31 in hypoxia-stimulated mice and H9c2 cardiomyocytes. Leonurine alleviates hypoxia-induced cardiac hypertrophy in mice. Overexpression of miR-210 or knockdown of miR-31 alleviates hypoxia-induced H9c2 cardiomyocyte apoptosis.
Materials and Methods
Animal Model
Eight week-old male C57BL/6J mice (20 ± 2 g; n = 24) were obtained from the Animal Laboratory Center of the Second Xiangya Hospital. Mice were divided into four groups (n = 6 in each group): Control group (21% O2 for 24 h/day); hypoxia group (chronic intermittent hypoxia with 7% or 21% O2 alternating 60 s for 12 h/day, 21% O2 for 12 h/day); leonurine low concentration group (chronic intermittent hypoxia combined with leonurine treatment; 5 mg/kg); leonurine high concentration group (chronic intermittent hypoxia combined with leonurine treatment; 50 mg/kg). Leonurine was obtained from Shanghai Aladdin Biochemical Technology Co., LTD (high-performance liquid chromatography ≥98%; Shanghai, China). After leonurine intragastric administration for 8 weeks, mice were sacrificed under anesthesia. The animal experiment was approved by the Ethics Committee of the Second Xiangya Hospital of Central South University. Animal research, including feeding, anesthesia, sacrifice and specimen collection, conformed to the Animal Research: Reporting in vivo Experiments guidelines 2.0. 23
miRs Microarray
miRs microarray (8 × 60K Mouse miRNA Microarray, Release 21.0; 1,881 mouse miRNAs represented miRBase database Agilent 60-mer SurePrint technology; Agilent, USA) was performed in triplicate heart specimens with or without chronic intermittent hypoxia treatment. Based on |Log2fold change| ≥ 1, P < 0.001 and false discovery rate <0.001, top 5 up-regulated (miR-31, miR-34a, miR-33, miR-155, and miR-140‐5p) and down-regulated (miR-210, miR-93‐5p, miR-214, miR-101a, and miR-202‐5p) miRs were filtered from hypoxia-stimulated mice. Finally, the differentially expressed miRs were presented by hierarchical clustering analysis, and the heatmap was drawn by MeV software (version 4.8, Institute for Genomic Research, USA).
Cell Culture
Rat H9c2 cells were purchased from ATCC (Bethesda, MD, USA) and cultured in DMEM (Gibco; Thermo Fisher Scientific, Inc.) for 2 hours at 37 ℃ with 5% CO2 and 95% air, and then cardiomyocytes were exposed to hypoxia with an atmosphere containing 5% CO2 and 1% O2 for 2 hours. Cells were pretreated with leonurine (Aladdin, Biochemical Technology Co., LTD, Shanghai, China) for 2 hours.
Cell Transfection
miR-31 (mimics: 5’-AGGCAAGAUGCUGGCAUAGCU-3’; inhibitors: 5’-AGCUAUGCCAGCAUCUUGCCU-3’) and miR-210 (mimics: 5’-CUGUGCGUGUGACAGCGGCUGA-3’; inhibitors: 5’-UCAGCCGCUGUCACACGCACAG-3’) were synthesized by Sangon Biotech (Shanghai, China). Inhibitors and mimics of miRNAs were transfected into cardiomyocytes using Lipofectamine 2000 (Invitrogen) for 48 hours at 37 ℃ according to the manufacturer’s protocol.
CCK-8 Assay
Cell proliferation was analyzed using a commercial CCK-8 kit (Beyotime Institute of Biotechnology, Haimen, China). In brief, cardiomyocytes (5 × 104) were cultured in a 96-well plate for 24 hours with different leonurine concentrations (0, 20 μM) for cytotoxicity analysis. After being in hypoxic conditions for 2 hours, cell proliferation was analyzed with a microplate reader at 450 nm.
TUNEL Assay
Cell apoptosis was analyzed using a commercial TUNEL kit (Roche). In brief, after transfection with either mimics, inhibitors or leonurine (20 µM) in hypoxic conditions for 2 hours, cardiomyocytes were plated on an 18 mm cover glass and fixed with 2% paraformaldehyde that was subsequently stained with TUNEL kit components. TUNEL positive cells were analyzed using a fluorescence microscope.
RT-qPCR Assay
Total RNA was extracted using miRNeasy Mini Kit (Qiagen, Inc., Valencia, CA, USA). Moloney murine leukemia virus reverse transcriptase (Invitrogen; Thermo Fisher Scientific, Inc.) and TaqMan® Universal PCR Master Mix (Thermo Fisher Scientific, Inc.) were utilized for RT-qPCR. miRs expression levels were calculated using the 2-ΔΔCq method. 24
Antioxidant Analysis
After cardiomyocyte transfection with either mimics, inhibitors or leonurine (1, 20 μM) in hypoxic conditions for 2 hours, DPPH and O2 - radicals were measured using an ultra-visible spectrophotometer at 519 nm and 325 nm, respectively, as described previously. 25 The experiments were performed three times in each group.
Statistical Analysis
Data are presented as the mean ± standard deviation. GraphPad Prism version 7.0 (GraphPad Software, Inc., La Jolla, CA, USA) was used to perform statistical analysis. Intergroup differences were analyzed by a one-way analysis of variance followed by a post hoc Tukey test. A significant difference was indicated as P < 0.05.
Results
Leonurine Alleviates Hypoxia-Induced Cardiac Hypertrophy in Mice
Histological examination by H&E staining was performed to evaluate the protective effect of leonurine on hypoxia-induced cardiac hypertrophy in mice. As shown in Figure 1(A), the size of heart was markedly expanded in hypoxia-stimulated mice compared with normal mice. However, leonurine treatment had the ability to inhibit hypoxia-induced cardiac expansion. Compared with the control group, heart weight/body weight (HW/BW) was significantly increased in the mice with hypoxia treatment. However, treatment with the high concentration of leonurine obviously reduced HW/BW in hypoxia-stimulated mice (Figure 1(B)). As shown in Figure 1(C), constrictive intercellular space and inflated cardiomyocytes were observed in hypoxia-treated mice. However, leonurine administration at high concentration observably improved the hypoxia-induced abnormal myocardial architecture in mice. In addition, hypertrophic markers, ANP, BNP, and β-MHC, were significantly up-regulated, while α-MHC was down-regulated in the heart of hypoxia-stimulated mice. Intriguingly, the mRNA expression of hypertrophic markers was reversed by leonurine treatment (Figure 1(D)).

Leonurine alleviates hypoxia-induced cardiac hypertrophy in mice. H&E staining was used to evaluate hypoxia-induced cardiac expansion (A). The ratio of HW to BW was analyzed in hypoxia-stimulated mice with or without leonurine administration (B). H&E staining was used to evaluate hypoxia-induced myocardial damage (C). The mRNA expression of hypertrophic markers, ANP, BNP, α-MHC, and β-MHC, were measured by RT-qPCR (D). n = 6 in each group. * P < 0.05 represents a significant difference in the two groups that are connected with the bracket.
Leonurine Regulates miR-31 and miR-210 Expression in Mice
miR microarray analysis was performed to determine differentially expressed miRs in response to hypoxia-induced myocardial damage. As shown in Figure 2(A), the top 5 up-regulated (miR-31, miR-34a, miR-33, miR-155, and miR-140‐5p) and down-regulated (miR-210, miR-93‐5p, miR-214, miR-101a, and miR-202‐5p) miRs were deregulated in the heart by hypoxia stimulation. More importantly, up-regulation of miR-31 (Figure 2(B)) and down-regulation of miR-210 (Figure 2(C)) were reversed by leonurine in hypoxia-stimulated mice.
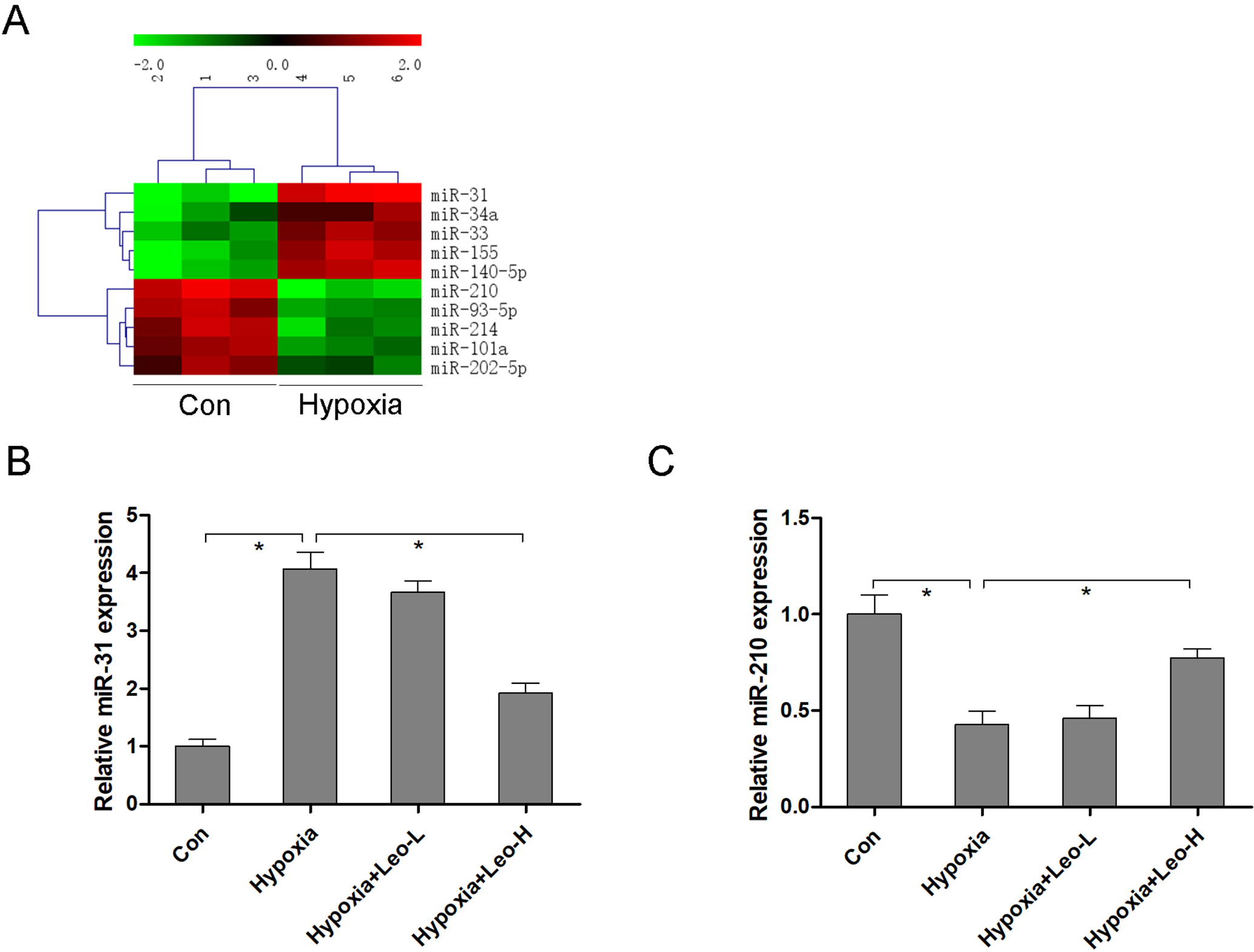
Leonurine regulates miR-31 and miR-210 expression in mice. miR microarray analysis was performed to determine differentially expressed miRs in response to hypoxia-induced myocardial damage (A). miR-31 (B) and miR-210 (C) expression were measured by RT-qPCR in hypoxia-stimulated mice with or without leonurine administration. n = 6 in each group. * P < 0.05 represents a significant difference in the two groups that are connected with the bracket.
Leonurine Eliminates Oxidative Stress in Hypoxia-Stimulated Rat H9c2 Cardiomyocytes
After cardiomyocyte exposure to leonurine at different concentrations, the cytotoxicity of the compound on cardiomyocytes was evaluated using CCK-8 assay. Cardiomyocyte growth had no significant change when the concentration of leonurine was less than 20 µM, suggesting that it alone in normoxic conditions had no obvious cytotoxicity on H9c2 cardiomyocytes (Figure 3(A)). The antioxidant activity of leonurine was assessed using DPPH and O2 - assays. As shown in Figure 3(B) and (C), leonurine exhibited an ability to scavenge DPPH and O2 - radicals in a dose-dependent manner in hypoxia-stimulated rat H9c2 cardiomyocytes. The 50% effective concentrations (EC50) of leonurine for DPPH and O2 - was 10.62 µM and 22.12 µM, respectively.

Leonurine eliminates oxidative stress in hypoxia-stimulated rat H9c2 cardiomyocytes. After cardiomyocyte exposure to leonurine in normoxic conditions for 24 hours, the cytotoxicity of leonurine was evaluated using the CCK-8 assay (A). DPPH (B) and O2 - (C) radicals were measured to evaluate the antioxidant activity of leonurine in hypoxia-stimulated rat H9c2 cardiomyocytes. n = 6 in each group.
Leonurine Alleviates Hypoxia-Induced Cardiomyocyte Apoptosis
As shown in Figure 4(A), hypoxia stimulation led to a significant reduction of cell viability of rat H9c2 cardiomyocytes, while leonurine supplementation alleviated this effect. We hypothesized that inhibition of cell growth might be associated with the acceleration of apoptosis in hypoxia-stimulated rat H9c2 cardiomyocytes. TUNEL assay was used to determine the effect of leonurine on hypoxia-induced cardiomyocyte apoptosis. Our results indicated that the apoptotic cell proportion was significantly increased following hypoxia stimulation. However, leonurine supplementation protected against this (Figure 4(B)).
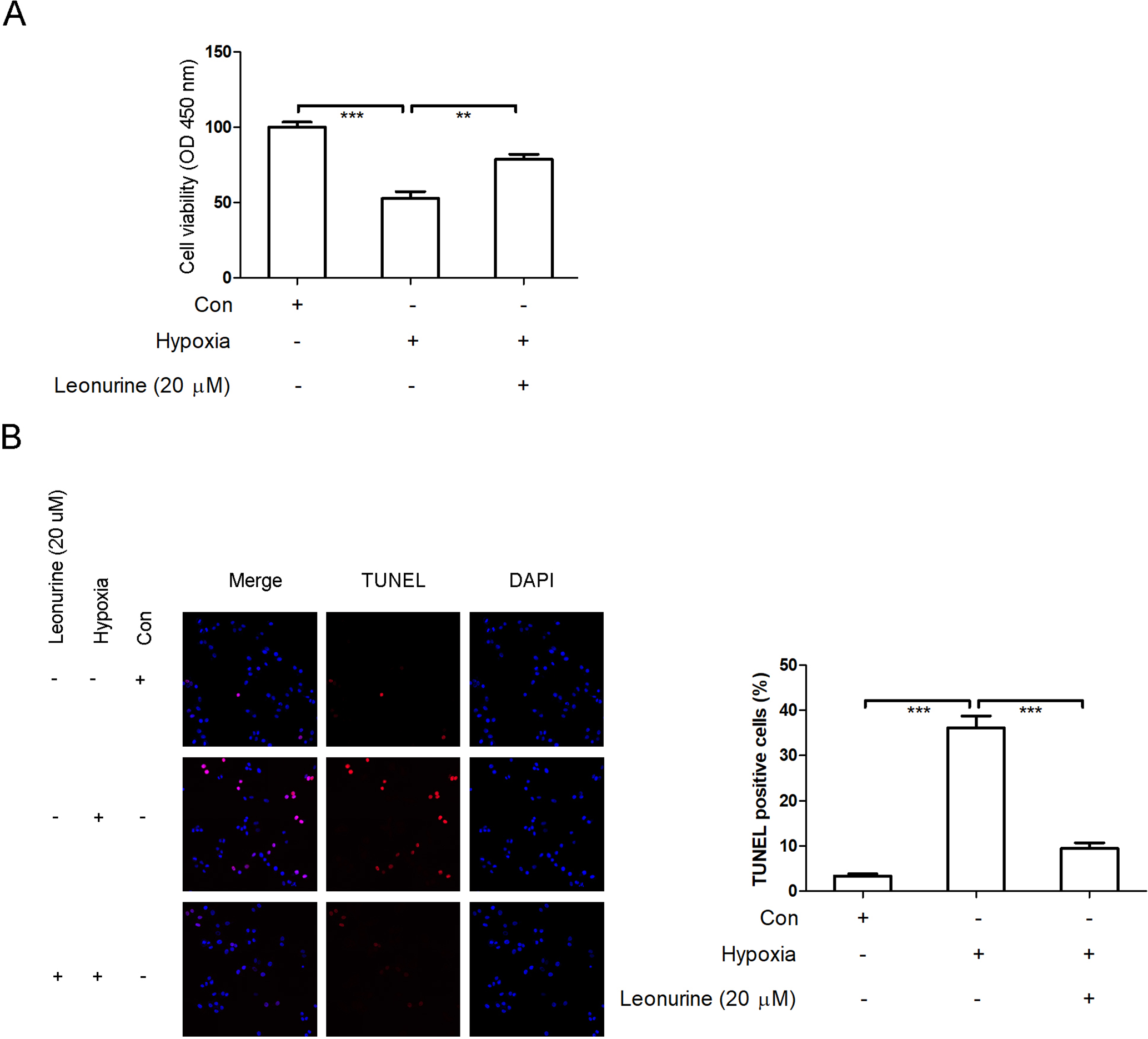
Leonurine alleviates hypoxia-induced cardiomyocyte apoptosis. After cardiomyocyte exposure to hypoxia for 2 hours with or without leonurine supplementation, cell viability was evaluated using the CCK-8 assay (A); cell apoptosis was measured using TUNEL assay (B). n = 6 in each group. ** P < 0.01 and *** P < 0.001 represent significant difference in the two groups that are connected with bracket.
Leonurine Targets miR-31 and miR-210 in Hypoxia-Stimulated Rat H9c2 Cardiomyocytes
miRNAs as pharmacological targets have emerged in cardiovascular disorder therapeutics. 26 Herein, we aimed to determine whether leonurine could regulate miRNAs in hypoxia-stimulated rat H9c2 cardiomyocytes. As shown in Figure 5(A) and (B), hypoxia-induced up-regulation of miR-31 and down-regulation of miR-210 were significantly reversed by leonurine treatment in cardiomyocytes, suggesting that miR-31 and miR-210 may be the potential targets of leonurine to prevent hypoxia-induced cardiomyocyte apoptosis.

Leonurine targets miR-31 and miR-210 in hypoxia-stimulated rat H9c2 cardiomyocytes. After cardiomyocyte exposure to hypoxia for 2 hours with or without leonurine supplementation, miR-31 and miR-210 were measured using RT-qPCR. n = 6 in each group. ** P < 0.01 and *** P < 0.001 represent significant difference in the two groups that are connected with bracket.
miR-31 Inhibitors or miR-210 Mimics Inhibit Hypoxia-Induced Cardiomyocyte Apoptosis
Based on the above findings, miR-31 was up-regulated and miR-210 was down-regulated in hypoxia-stimulated cardiomyocytes, indicating that overexpression of miR-31 or reduction of miR-210 may be correlated with hypoxia-induced cardiomyocyte apoptosis. To investigate the function of miR-31 or miR-210 in the process of hypoxia-induced cardiomyocyte apoptosis, miR-31 inhibitors or miR-210 mimics were transfected into cardiomyocytes under hypoxic conditions. Our results demonstrated that knockdown of miR-31 (Figure 6(A)) or overexpression of miR-210 (Figure 6(B)) could repress hypoxia-induced cardiomyocyte apoptosis.

miR-31 inhibitors or miR-210 mimics inhibit hypoxia-induced cardiomyocyte apoptosis. miR-31 inhibitors (A) or miR-210 mimic (B) transfected cardiomyocytes exposed to hypoxia for 2 hours; cell apoptosis was measured using TUNEL assay. n = 6 in each group. *** P < 0.001 represents a significant difference in the two groups that are connected with the bracket.
Overexpression of miR-31 or miR-210 Inhibitors Counteract the Protective Effect of Leonurine on Hypoxia-Induced Cardiomyocyte Apoptosis
To discuss further the association between leonurine and miR-31/miR-210 hypoxia-induced cardiomyocyte apoptosis, miR-31 mimics or miR-210 inhibitors were transfected into leonurine-treated cardiomyocytes in hypoxic conditions. Inhibition of hypoxia-induced cardiomyocyte apoptosis by leonurine was revoked by miR-31 mimics (Figure 7(A)) and miR-210 inhibitors (Figure 7(B)). These findings revealed that leonurine could prevent hypoxia-induced cardiomyocyte apoptosis via inhibiting miR-31 and elevating miR-210 expression.

Overexpression of either miR-31 or miR-210 inhibitors counteracts the protective effect of leonurine on hypoxia-induced cardiomyocyte apoptosis. miR-31 mimics (A) or miR-210 inhibitor (B) transfected cardiomyocytes exposed to hypoxia for 2 hours with or without leonurine supplementation; cell apoptosis was measured using TUNEL assay. n = 6 in each group. *** P < 0.001 represents a significant difference in the two groups that are connected with the bracket.
Discussion
Multiple lines of evidence corroborate that miRNAs are effective targets for natural bioactive components to ameliorate cellular damage and restrain carcinogenesis. 27 For instance, curcumin exhibits anti-cancer properties via mediating a variety of miRNAs. 15,27 -29 In addition, miRNAs are the determinants of ischemia-reperfusion injury and function as potential pharmaco-targets to relieve hypoxia/re-oxygenation related cell apoptosis. 15 Curcumin activates miR-7a/b to protect against hypoxia-induced cardiac myocyte apoptosis. 28 Bioactive polysaccharides mitigate hypoxia-induced rat H9c2 cardiomyocyte apoptosis via repressing miR-22. 29 Based on these findings, we deduced that miRNAs as pharmacological targets are implicated in the cardioprotection of hypoxia-induced cardiomyocyte damage.
In our study, leonurine showed cardioprotective activity in an in vitro cell model of hypoxia-stimulated H9c2 cardiomyocytes and in vivo mice model, reflecting that leonurine improves hypoxia-induced growth inhibition and apoptosis of cardiomyocytes and alleviates hypoxia-induced cardiac hypertrophy in mice. Furthermore, we plan to explore whether cardiac-specific miRNAs as pharmacological targets of leonurine are involved in the cardioprotection. Two representative cardiac-specific miRNAs, miR-31, and miR-210, were screened out from previous studies. 30,31 Overexpression of miR-31 is proven to exacerbate cardiac hypertrophy and hypoxia-induced cardiomyocyte apoptosis. 30,32 miR-210 exhibits an opposite function of miR-31 in the cardioprotection. 31 miR-210 modulates proliferation, survival and anti-apoptosis of cardiomyocytes in response to hypoxia stimulation. 31,33 Consistent with previous results, 30,31,33 our findings suggested that both up-regulation of miR-31 and reduction of miR-210 were observed in hypoxia-treated H9c2 cardiomyocytes. Interestingly, leonurine could reverse the hypoxia-induced up-regulation of miR-31 and the reduction of miR-210 in cardiomyocytes.
Functionally, TUNEL assay reveals that transfection of miR-31 inhibitors or miR-210 mimics abrogates hypoxia-induced cardiomyocyte apoptosis. Previous studies also indicate that miR-210 mimics protect cardiomyocyte apoptosis from oxygen-glucose deprivation/reperfusion or hydrogen peroxide stimulation. 34,35 In contrast, miR-31 mimics or miR-210 inhibitors counteract the anti-apoptotic effect of leonurine on hypoxia-treated rat H9c2 cardiomyocytes. Our findings suggest that miR-31 and miR-210 as the upstream regulators of leonurine are involved in hypoxia-induced cardiomyocyte apoptosis.
In conclusion, leonurine can target miRNAs to protect against hypoxia-induced cardiomyocyte apoptosis and cardiac hypertrophy. The utilization of miRNAs agomir or antagonist or miRNAs specific chemical drugs may provide a therapeutic strategy for the treatment of myocardial damage.
Footnotes
Statement of Animal Rights
All procedures in this study were conducted in accordance with the Institutional Animal Care and Use Committee of the Second Xiangya Hospital of Central South University, Hunan Province, China according to the Animal Research: Reporting in Vivo Experiments guidelines 2.0.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the science and technology planning of Changsha City, Hunan Province, China (Grant number: kq1907058).
