Abstract
The efficacy of Truong Xuan Cao Bang (TXCB), a Vietnamese herbal medicine prescription, on the quality of semen, serum testosterone (TES), and malondialdehyde (MDA) concentrations in rabbits whose reproductive ability were impaired by fluconazole was investigated. Mature male rabbits were randomly assigned into control (G1), fluconazole (G2), fluconazole and 180 mg/kg/day (G3), and 360 mg/kg/day TXCB (G4) groups. The reaction time (RT), ejaculate volume (EV), sperm concentration (SC), total sperm output (TSO), total sperm motility (TSM), progressive sperm motility (PSM), nonprogressive sperm motility (NPSM), sperm immobility (SIM), dead sperm (DS), and abnormal sperm (AS) levels were determined after TXCB administration. After 60 days, G3 showed a significant increase in serum TES and a significant decrease in the concentrations of serum MDA (SMDA) and seminal plasma MDA (SPMDA) compared with G1, in addition to increase in EV, SC, TSO, TSM, PSM, and serum TES, and decrease in RT, SIM, DS, AS, SMDA, and SPMDA compared with G2. G4 showed a steep rise in EV, SC, and serum TES and a downturn in DS, SMDA, and SPMDA compared with G1, and, compared with G2, a significant increase in EV, SC, TSO, TSM, PSM, and serum TES and a significant decrease in RT, SIM, DS, AS, SMDA, and SPMDA. Taken together, TXCB rehabilitates and simultaneously enhances spermatogenesis in rabbits whose reproductive ability was impaired by fluconazole.
Keywords
The World Health Organization (WHO) has deemed infertility a public health problem. The incidence of infertility ranges greatly between studies, but, in general, the average infertility rate of infertile couples is estimated to be 9% (or between 3.5% and 16.7%) in developed countries and 6.9%-9.3% in developing countries. 1 Female infertility accounts for about 30%-40% of infertility cases, male infertility for about 40%, and infertility of both spouses for about 20%; the cause of infertility is unknown for the remaining 10%-15% of couples. 2 There are many different causes of male infertility that lead to various changes in semen, including low sperm concentrations (oligospermia), poor sperm motility (asthenospermia), and abnormal sperm (AS) morphology (teratozoospermia). 3,4 The quantity and quality of men’s sperm are declining. 5,6 Therefore, it is essential to find measures to improve sperm quantity and quality.
The Truong Xuan Cao Bang decoction (TXCB) was formulated from a Vietnamese traditional medicine by the Vietnam Military Medical University in Ha Noi, Vietnam. The prescription contains 8 medicinal herbs: Balanophora laxiflora Hemsl. (10 g), Herba epimedii (10 g), Curculigo orchioides Gaertn. (8 g), Radix Morindae officinalis (8 g), Cornus cerviparvum (5 g), Radix Millettiae speciosae (10 g), Dendrobium officinale Kimura et Migo (10 g), and Fructus Lycii (10 g) and met national manufacturing safety standards. In this article, we investigated whether TXCB could enhance the quantity and quality of sperm in rabbits whose reproductive ability was impaired by fluconazole in vivo.
Results
Mature male rabbits were randomly assigned into 4 groups: group 1 (control group) oral administration of sodium carboxymethyl cellulose (NaCMC) 2.0%; group 2 (pathological group), group 3 (low dose group), and group 4 (high dose group) oral administration of fluconazole in NaCMC 2.0%, 50 mg/kg at 8 am. Further, rabbits in group 3 and group 4 received TXCB 180 mg/kg/day and 360 mg/kg/day, respectively, at 2 pm. After 60 days of drug administration, the body weights of the animals were measured in the range of 2.48-2.86 kg. There were no significant differences in the body weight of rabbits between the groups (Figure 1).

The body weight of rabbits.
The following variables related to the quality of semen were determined at baseline (day 0) (Table 1) and at 30 days (Table 2) and 60 days (Table 3) after TXCB administration: reaction time (RT), ejaculate volume (EV), pH, sperm concentration (SC), total sperm output (TSO), total sperm motility (TSM), progressive sperm motility (PSM), nonprogressive sperm motility (NPSM), sperm immobility (SIM), dead sperm (DS), AS, serum testosterone (TES), serum malondialdehyde (SMDA), and serum plasma MDA (SPMDA).
Investigation of Indexes at Day 0.
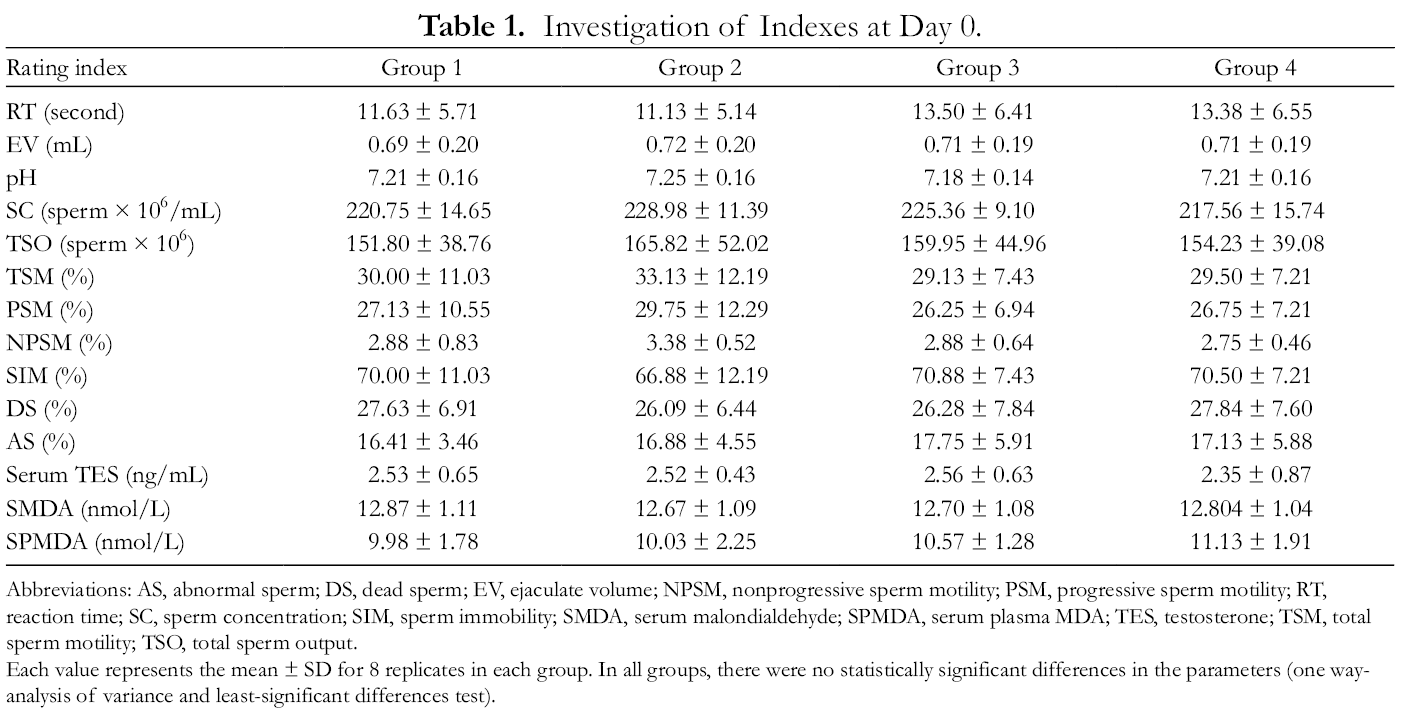
Abbreviations: AS, abnormal sperm; DS, dead sperm; EV, ejaculate volume; NPSM, nonprogressive sperm motility; PSM, progressive sperm motility; RT, reaction time; SC, sperm concentration; SIM, sperm immobility; SMDA, serum malondialdehyde; SPMDA, serum plasma MDA; TES, testosterone; TSM, total sperm motility; TSO, total sperm output.
Each value represents the mean ± SD for 8 replicates in each group. In all groups, there were no statistically significant differences in the parameters (one way-analysis of variance and least-significant differences test).
Investigation of Indexes at Day 30.

Abbreviations: AS, abnormal sperm; DS, dead sperm; EV, ejaculate volume; NPSM, nonprogressive sperm motility; PSM, progressive sperm motility; RT, reaction time; SC, sperm concentration; SIM, sperm immobility; SMDA, serum malondialdehyde; SPMDA, serum plasma MDA; TES, testosterone; TSM, total sperm motility; TSO, total sperm output.
Each value represents the mean ± SD for 8 replicates in each group. Different superscript letters (a, b, c) within row are significantly different using one way-analysis of variance with least-significant differences post hoc testing at P < 0.05.
Investigation of Indexes at Day 60.

Abbreviations: AS, abnormal sperm; DS, dead sperm; EV, ejaculate volume; NPSM, nonprogressive sperm motility; PSM, progressive sperm motility; RT, reaction time; SC, sperm concentration; SIM, sperm immobility; SMDA, serum malondialdehyde; SPMDA, serum plasma MDA; TES, testosterone; TSM, total sperm motility; TSO, total sperm output.
Each value represents the mean ± SD for 8 replicates in each group. Different superscript letters (a, b, c) within row are significantly different using one way-analysis of variance with least-significant differences post hoc testing at P < 0.05.
In group 2, 30 days after oral administration of TXCB, the mean values for RT, DS, AS, SMDA, and SPMDA were significantly different from those in the control group (P < 0.05) (Table 2). In group 3, the values of SC, TSO, and serum TES were significantly lower compared with the control group (P = 0.024, 0.013, and 0.017, respectively); the values of SMDA and SPMDA were also significantly lower than in the control group (P = 0.019 and 0.008, respectively). There was no significant difference in the other semen quality variables between groups 3 and 1. Therefore, the oral administration of TXCB at a dose of 180 mg/kg rehabilitated RT, DS, AS, EV levels but simultaneously reduced SMDA and SPMDA levels (Figure 2 and Table 2).

Microscopic testicle images of rabbit sperm. (A) Sperm vitality. Eosin-nigrosin smear was taken at ×100 magnification. Spermatozoa with dark or dark pink heads are dead (red arrow), whereas spermatozoa with either white or light pink heads are alive. (B) Sperm morphology. Papanicolaou stain, measured at ×100 magnification, abnormal sperm morphology (red arrow).
Group 4 showed a significant increase in TSM and PSM (P = 0.026 and 0.027, respectively) and a significant decrease in SIM (P = 0.026). Like group 3, group 4 showed a significant decrease in SMDA and SPMDA compared with group 1 (P = 0.009 and P < 0.001, respectively). Thus, TXCB at a dosage of 360 mg/kg recovers RT, EV, SC, TSO, DS, AS, and serum TES values, in addition to increasing TSM, PSM values and reducing SIM, SMDA, and SPMDA (Table 2).
After 60 days of oral TXCB administration, there were significant decreases in EV, SC, TSO, TSM, and PSM compared with the control group (P = 0.024, 0.011, 0.048, 0.008 and 0.007, respectively). At the same time, RT, SIM, DS, AS, and SPMDA (P = 0.019, 0.008, 0.003, 0.027, and <0.001, respectively) were increased compared with the control. There were no significant changes in serum TES and SMDA values between baseline and 60 days (Table 3). In group 3, there were no significant changes in the study indices compared with group 1, except for an increase in serum TES (P = 0.006) and a decrease in SMDA (P = 0.001) and SPMDA (P < 0.001). In group 4, the value of serum TES was larger than in the control group (P < 0.001), whereas the values of SMDA (P = 0.001) and SPMDA (P < 0.001) were lower than in the control group. In addition, the increment of EV and SC were recorded (P = 0.046 and 0.042, respectively). The values of DS were decreased (P = 0.027) relative to the control group (Figure 2). TSO values appeared to increase relative to the control group, but the difference was not statistically significant (Table 3). Additionally, group 4 showed significant differences in RT, EV, SC, TSO, and SMDA values compared with group 3 (P = 0.035; 0.024; 0.015; 0.041 and 0.001, respectively).
In addition, the values of the various indexes are shown in Table 4 (group 2), Table 5 (group 3), and Table 6 (group 4).
Indexes of Group 2 at Days 0, 30, and 60.

Abbreviations: AS, abnormal sperm; DS, dead sperm; EV, ejaculate volume; NPSM, nonprogressive sperm motility; PSM, progressive sperm motility; RT, reaction time; SC, sperm concentration; SIM, sperm immobility; SMDA, serum malondialdehyde; SPMDA, serum plasma MDA; TES, testosterone; TSM, total sperm motility; TSO, total sperm output.
Note: *P ≤ 0.05, **P ≤ 0.001 compared with day 0.
#P ≤ 0.05, ## P ≤ 0.001 compared with day 30.
Indexes of Group 3 at Days 0, 30, and 60.

Abbreviations: AS, abnormal sperm; DS, dead sperm; EV, ejaculate volume; NPSM, nonprogressive sperm motility; PSM, progressive sperm motility; RT, reaction time; SC, sperm concentration; SIM, sperm immobility; SMDA, serum malondialdehyde; SPMDA, serum plasma MDA; TES, testosterone; TSM, total sperm motility; TSO, total sperm output.
Note: *P ≤ 0.05, **P ≤ 0.001 compared with day 0.
# P ≤ 0.05, ## P ≤ 0.001 compared with day 30.
Indexes of Group 4 at Days 0, 30, and 60.

Abbreviations: AS, abnormal sperm; DS, dead sperm; EV, ejaculate volume; NPSM, nonprogressive sperm motility; PSM, progressive sperm motility; RT, reaction time; SC, sperm concentration; SIM, sperm immobility; SMDA, serum malondialdehyde; SPMDA, serum plasma MDA; TES, testosterone; TSM, total sperm motility; TSO, total sperm output.
Note: *P ≤ 0.05, **P ≤ 0.001 compared with day 0.
# P ≤ 0.05, ## P ≤ 0.001 compared with day 30.
Microscopic images of rabbit testicular sections were taken with a 400× magnification camera (Figure 3). In group 1, sperm tubes were quite even, and the inner sperm tubes contained many sperm cells with a clear tail. The spermatogenesis wall was full of spermatozoa in the form of Sertoli cells, spermatozoa, and pre-spermatozoa, and some nuclei were scattered (Figure 3, G-1). In contrast, group 2 (Figure 3, G-2) showed signs of sperm decline (Johnson score 8). The seminal tubes were quite even, and the inner sperm tubes contained few sperm. The spermatogenesis wall contained fewer spermatozoa as Sertoli cells, spermatophores, spermatozoa, and pre-spermatozoa, and there was no split mirror. Some spermatomas had degenerated sperm cell lines (Figure 3, G-2).

Photomicrographs (×400) of animal testicular sections. Haematoxylin and eosin were used for staining. Seminiferous tubules containing normal germ cells, including spermatogonia and Sertoli cells, and normal maturation stages with the presence of spermatozoa within their lumen (blue arrow). The connective tissues outside the seminiferous tubules have congested blood capillaries (black arrow). The seminiferous tubule walls contained small numbers of spermatogonia cells with a small number of rows of cells (red arrow). Some seminiferous tubules contained degenerated spermatogonia cells (yellow arrow).
Both treatment groups (groups 3 and 4) showed signs of sperm proliferation. The spermatogenesis tubes were quite even, and the inner sperm tubes contained many sperm with a clear tail. The spermatogenesis wall was particularly full of spermatozoa in group 4, in the form of Sertoli cells, spermatozoa, and pre-spermatozoa. Some nuclei were scattered in both groups 3 and 4 (Figure 3, G-3 and G-4).
Discussion
In order to study the effect of drugs on reproductive processes, animal models of reproductive impairment are required. Some fertility impairment methods include thermal depletion, stress, and the use of either drugs or chemicals; older animals can also be used. 7,8 In our experiment, we used fluconazole (FLZ) for fertility impairment. FLZ is a synthetic azo (triazol) antifungal drug that inhibits cytochrome P450 14-alpha-demethylase and inhibits the synthesis of ergosterol, a major sterol in fungal cell membranes. FLZ has a strong affinity for the fungal P450 enzyme and a weaker affinity for the mammalian P450 enzyme, but this is still sufficient to induce interactions. 9 It has been reported that FLZ significantly increases prolactin and follicle-stimulating hormone (FSH) levels in rats when administered for 30 days, 10 indicating that FLZ also affects TES concentrations as well as fertility. FLZ has also been shown to impair male fertility via antiandrogenic effects 11 resulting in impaired semen volume and sperm quality, sperm count, and motility. 12,13
Rabbits are the third most frequently used experimental mammal species for in vivo tests (2.8%), after mice (59.3%) and rats (17.7%). Furthermore, rabbits are the smallest and cheapest experimental animals for which it is possible to obtain semen samples from the vagina of a recent mate, enabling semen analysis and evaluation at many time points. The normal male reproductive process of rabbits, that is, the cycle of spermatogenesis and the maturation of the testicles, is well known. Females can be artificially inseminated with semen taken from males to assess fertility, number of offspring in a litter, and female survival. Moreover, the reproductive cycle of rabbits is predictable (female rabbits are typically pregnant for about 30 days). 14 -18
In our study, FLZ at 50 mg/kg/day and TXCB at doses of 180 and 360 mg/kg/day had no effect on rabbit body weight, and no signs of toxicity were observed. In the pathology group (group 2, which received FLZ only), serum TES concentration significantly decreased from baseline after 30 and 60 days (P = 0.002 and 0.020, respectively). This is consistent with previous literature. 19 It has been demonstrated that FLZ does not induce oxidation in Leydig cells in vitro 20 so the decrease in TES we observed with FLZ could be explained by inhibition of TES biosynthesis in Leydig cells. 21 In the groups that received both FLZ and TXCB (groups 3 and 4), TES concentration was significantly higher than in the FLZ group at the 30 and 60 day timepoints (P < 0.05). In the 180 mg/kg TXCB group (group 3), TES concentration decreased relative to baseline after 30 days (P = 0.015) and was significantly lower than the control group (P = 0.017) at the same timepoint; after 60 days, the TES concentration increased and was significantly higher than baseline (P = 0.006) (Tables 2 and 3). In the 360 mg/kg TXCB group (group 4), TES concentration at 30 days did not change significantly from baseline and was not statistically different from the control group at the same timepoint; however, after 60 days, TES concentration increased to a level significantly higher than baseline and the control group. Thus, TXCB increased serum TES concentration in fertility-impaired rabbits in a dose-dependent manner.
A therapy like TXCB that increases TES levels in the blood may be effective. This is based on 2 main mechanisms: (1) increased synthesis of TES in the testicles and (2) the drug itself acts as exogenous TES.
RT is considered a sign of libido. It is typically calculated as the time elapsed between the female’s arrival in the male’s cage and the male’s ejaculation. 17,22,23 However, we chose to calculate the RT as the time at which the female rabbits were put into the male rabbit’s cage until the male jumped on the female’s back to perform the mating act. In the FLZ group (group 2), RT was significantly higher than the control group and significantly higher than baseline at both 30 and 60 days (Tables 1 -3). Thus, FLZ reduces libido. The mechanism behind the libido decrease with FLZ is likely due to its antiandrogen effect, which inhibits TES synthesis in Leydig cells, leading to a decrease in the serum TES concentration. In our experiment, the RT of rabbits in the FLZ and TXCB-treated groups (groups 3 and 4) was not significantly different from baseline or from the control group and was significantly lower than the FLZ-only group (group 2). Therefore, TXCB appears to rescue the FLZ-induced decrease in libido. This is likely due to TXCB’s effect of increasing serum TES concentration.
We also investigated the effect of TXCB on various measures of sperm quantity and quality, including semen volume, sperm density, and total sperm count in a single ejaculation. In FLZ-treated animals (group 2), semen volume at 30 and 60 days was significantly lower than baseline and significantly lower than the semen volume in the control group at the same timepoints. The same pattern was seen for sperm density and total number of spermatozoa in one ejaculation. Thus, consistent with previous literature, 19 FLZ decreased rabbits’ semen volume, sperm density, and total sperm count in 1 ejaculation. In groups 3 and 4, which received both FLZ and TXCB, semen volume increased significantly compared with the control group. At 30 days, sperm density in both groups decreased significantly compared with baseline, but not relative to the control group at 30 days. However, by 60 days, sperm density tended to increase (Table 3). In the low-dose (180 mg/kg) TXCB group (group 3), the total number of sperm in 1 ejaculate decreased significantly from baseline at 30 days (P = 0.006) and was significantly lower than the control group at 30 days (P = 0.013). By 60 days, the total number of sperm in 1 ejaculation was significantly higher than at 30 days (P = 0.001) and higher than the baseline group at 60 days (P = 0.001). Thus, TXCB negated the FLZ’s detrimental effects on semen volume, sperm density, and total sperm count in 1 ejaculation, and this effect appeared to be dose dependent.
TXCB additionally increases TES levels, which may also be beneficial for sperm production, thereby increasing sperm count. TES is considered to be a prerequisite for sperm production and maturation as well as the development and function of secondary sexual characteristics. TES activates androgen receptors (AR) in Sertoli cells to initiate functional responses required for spermatogenesis. 22,23 TES also works with FSH to stimulate the proliferation of Sertoli cells and generate signaling molecules and nutrients to support sperm maturation. 24 Therefore, it is possible that TXCB increases sperm production by both of these mechanisms. Some literature on the medicinal herbs in TXCB support this hypothesis. Ethanol extract of ginseng increases spermatogenesis through both mechanisms above and increases the size of the spermatogenesis tube, number of secondary sperm cells, and sperm cells. 25 In other studies, animals treated with areca extract showed an increase in testicular weight and histological changes of the testicles that favor spermatogenesis. 26 Histological imaging showed many different cells at different stages of spermatogenesis. A substantial sperm count was visible in each tube. Sertoli cells were larger in size and richer in nutrients, as evidenced by the high amount of cytoplasm. Almost all Leydig cells showed hypertrophy with enlarged nuclei and dark-stained cytoplasm. Increases in cell volume and nucleus are strongly suggestive of steroid synthesis as a direct or indirect effect of drugs. Almost all vas deferens contain a substantial number of sperm. In some tubes, crystals were found scattered among the spermatozoa (Figures 2 and 3).
As for sperm motility parameters, our results indicate that FLZ reduced the rate of sperm motility, thus increasing the rate of sperm immobilization (Tables 2 and 3). The presence of TXCB increased the rate of sperm motility. We did not evaluate mitochondrial function in this study, so it was impossible to determine with certainty the molecular mechanism of TXCB on the increase in sperm motility. However, we expect that this effect can be explained through the antioxidant properties of TXCB. Reactive oxygen species (ROS) are usually produced during mitochondrial oxidative phosphorylation, but high concentrations of these compounds can impair mitochondrial activity and thus impair sperm motility. MDA, a product of free oxygen radicals, is one of the end products in lipid peroxidation. Evaluating MDA concentration allows indirect assessment of the presence or activity of ROS. When the concentration of MDA is higher, the presence of ROS is greater and vice versa. Previous literature demonstrates that lipid peroxidation has an adverse effect on semen quality and that semen MDA concentration was significantly higher in people with weak sperm compared with those with normal sperm. 27,28 In this study, TXCB-treated animals showed a lower concentration of MDA in both semen and serum versus controls. This provides strong evidence that TXCB’s antioxidant activity could be the reason for TXCB’s positive effect on sperm motility.
Sperm survival and morphology are among the factors that influence male fertility. In our experiment, the FLZ-treated group showed increased rates of dead and morphologically AS over time, and these were significantly higher than in the control group. Thus, FLZ increased the sperm death rate and the rate of AS morphology in rabbits. This is consistent with previous literature. 29 The mechanism of this effect is not clear, but it may relate to an increase of MDA due to increased lipid peroxidation. Animals administered TXCB had a significantly lower sperm death rate and rate of AS morphology than the FLZ group at each time point (Figure 2). Therefore, the oral administration of TXCB may prevent FLZ from increasing sperm death and altering sperm morphology.
Materials and Methods
Chemical Reagents
Fluconazole USP 40 standard was purchased from Synergene, India, and Papanicolaou’s solution 2a Orange G solution (OG6), Papanicolaou’s solution 3b polychromatic solution (EA 50), and hematoxylin solution modified (acc. to Gill II) from Merck KGaA, Germany.
General Experimental Procedures
Sperm quality analyzers QwikCheckTM Gold (Israel); inverted microscope Nikon Eclipse Ti2 (Japan); automated slide stainer Leica Autostainer XL- ST5010, Germany; microplate reader ELISA Chromate 4300, Awareness Technology, USA. Microplate Washer Stat Fax- 2600, Awareness Technology, USA. Microwell plate mixer/incubator Stat Fax- 2200, Awareness Technology, USA. Deep refrigerator, Panasonic MDF-U33V-PB, Japan. ELISA Rabbit MDA ELISA Kit (KTE90101) and ELISA EliKine Testosterone ELISA Kit (KET0001), Abbkine, China.
Animals
Mature and healthy New Zealand male rabbits (body weight 2.5 ± 0.2 kg) were kept separately in cages (room temperature 20-30 °C) with access to food pellets and water. The rabbits were kept stable before the study and trained (for semen) with a fake vagina for 2 weeks at a frequency of twice per week. A total of 32 male rabbits were randomly divided into 4 groups: group 1 (control group) received oral administration of NaCMC 2.0% for 30 days and drank physiological saline for 60 days; group 2 (pathological group) was orally administered fluconazole (in NaCMC 2.0%, 50 mg/kg) for 30 days and drank physiological saline for 60 days; group 3 (low-dose group) received fluconazole (in NaCMC 2.0, 50 mg/kg) for 30 days and TXCB (diluted in physiological saline) at a dose of 180 mg/kg for 60 days. Group 4 (high-dose group) received fluconazole (in NaCMC 2.0%, 50 mg/kg) for 30 days and TXCB (360 mg/kg) for 60 days.
Evaluation Criteria
Semen samples were collected at 3 timepoints: before drug administration (baseline), after 30 days, and after 60 days (please see raw data). In all cases, samples were collected in the morning. A female rabbit and a fake vagina were used to induce ejaculation. RT was counted as the time between the release of a female rabbit into the male rabbit’s pen and the male rabbit jumping onto the female’s back to initiate mating.
The volume of seminal fluid (EV) was measured via the sperm collection tube. pH was determined by paper pH-indicator strips (pH 6.5-10.0, Special Indicator, Merck-Germany). Parameters including SC, TSM, PSM, NPSM, and SIM (ie, percentage of nonmobile sperm) were analyzed using a QwikCheck Gold-Israel automatic sperm analyzer with an E-capillary. TSO (ie, sperm density × seminal volume) and DS (ie, percentage of dead sperm) were assessed using a fluorescence microscope (Nikon Eclipse Ti2, Japan) after Eosin-nigrosin staining. AS (ie, percentage of spermatozoa with abnormal morphology) was assessed using Papanicolaou dye (Papanicolaou solution and hematoxylin solution, Merck, Germany) with a Leica Autostainer XL-ST5010 automatic dyeing machine (Germany) on a fluorescence microscope (Nikon Eclipse Ti2, Japan). The semen samples were then separated from the seminal fluid (via centrifugation at 3000 rpm for 15 minutes) and stored at −80 °C (Panasonic MDF-U33V-PB deep cabinet, Japan) for SPMDA analysis.
After sperm collection, rabbit ear vein blood was taken into a serum separator tube. It was allowed to coagulate for 30 minutes at room temperature and then centrifuged at 3000 rpm for 15 minutes in order to remove the serum. The sample was stored at −80 °C until analysis. ELISA kits were used to determine TS (EliKine Testosterone ELISA Kit, Abbkine), SMDA, and SPMDA (Rabbit MDA ELISA Kit, Abbkine) according to the manufacturer’s instructions, using a Chromate 4300 reader (Awareness Technology, USA). Evaluation of testicular microscopic images occurred at the end of the experiment (Supplemental Material).
Statistical Analysis
In the figures, data are presented as mean ± SD. Differences in measures of sperm quality between the groups were compared using one-way analysis of variance with the post hoc least-significant differences test. P values ≤0.05 were considered statistically significant. All statistical results were calculated using the IBM SPSS Statistics 20.0 software.
Conclusions
To the best of our knowledge, this study demonstrates for the first time that oral administration of TXCB is safe and shows promise for improving male fertility in a rabbit model. In male rabbits whose fertility was impaired by FLZ, TXCB capsules at doses of 180 and 360 mg/kg decreased RT and increased sperm quality and quantity in terms of semen volume, sperm count, total sperm count in 1 ejaculation, sperm motility, and progression rate. Furthermore, TXCB decreased the rate of sperm death and reduced the rate of morphologically AS. Therefore, TXCB appears to have an impact on many stages of spermatogenesis and thus should be further investigated to improve sexual function and male reproduction.
Supplemental Material
Supplementary Material 1 - Supplemental material for In Vivo Efficacy of TXCB, a Vietnamese Herbal Medicine Prescription, on Seminal Quality, Serum Testosterone, and Malondialdehyde Concentration in Rabbits
Supplemental material, Supplementary Material 1, for In Vivo Efficacy of TXCB, a Vietnamese Herbal Medicine Prescription, on Seminal Quality, Serum Testosterone, and Malondialdehyde Concentration in Rabbits by Vu Ngoc Thang, Nguyen Thanh Ha Tuan, Pham Quoc Binh, Nguyen Minh Phuong, Thai Danh Tuyen, Doan Minh Thuy, Manh Hung Tran and Nguyen Hoang Ngan in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Cao Bang Province, Vietnam (NHG-001).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
