Abstract
Our previous study demonstrated that an extract of Cordyceps militaris (CM) derived from Samia Cynthia ricini inhibits testosterone-induced prostate hypertrophy in an animal model. However, the bioactive components in the CM extract that modulate the function of prostate cells have not been identified and characterized. To do this, the present study explored the effective component(s) from CM that influences the viability of androgen-responsive LNCaP prostate cancer cells and androgen-independent PC3 prostate cancer cells. Several fractions of CM, separated by gel filtration chromatography, decreased the viability and proliferation of both types of prostate cancer cells in either the presence or absence of testosterone propionate (TP). The fractions reduced TP-induced expression of the androgen-regulated genes FKBP5, TMPRSS2, and KLK3. NMR identified cordycepin, adenosine, and uridine as the active components in the CM fractions. The proliferation of LNCaP cells was inhibited by cordycepin, but not by either adenosine or uridine. Cordycepin, adenosine, and uridine reduced TP-mediated increases in FKBP5 and TMPRSS2 expression, and TP-mediated phosphorylation of the androgen receptor. Specific adenosine A1, A2, and A3 receptor antagonists did not block the inhibitory effects of cordycepin on cell proliferation, whereas the A1 receptor antagonist CPDPX (8-cyclopentyl-1,3-dipropylxanthine) reversed cordycepin-mediated inhibition of FKBP5 and TMPRSS2 expression. Thus, cordycepin exhibited both adenosine receptor–independent and adenosine A1 receptor–dependent effects. These findings suggest that cordycepin inhibits the proliferation of prostate cells independently of adenosine receptors, and that its modulatory effect on androgen-regulated gene expression is mediated through the adenosine A1 receptor.
Introduction
The fungus Cordyceps militaris (CM) grows as a parasite on the larvae of moths of the order Lepidoptera, and then forms fruiting bodies. 1 In our previous study, we showed that an extract of CM derived from the moth Samia cynthia ricini inhibits androgen metabolism in an animal model of late-onset hypogonadism and suppresses testosterone-induced prostate hypertrophy in an animal model of benign prostatic hyperplasia (BPH). 2 Moreover, CM decreases the viability of prostate cancer cells in vitro. In general, the dried fruiting bodies of Cordyceps have antifatigue activities without side effects3,4 and are used in traditional Asian medicine as a tonic.5,6 CM has unique bioactivities that improve sexual function, sperm quality, 7 and testicular damage. 8
BPH, which is characterized primarily by stromal hyperplasia, occurs frequently in aging males and impairs quality of life. Androgens, including testosterone and dihydrotestosterone, act on androgen receptors (AR), regulate prostatic homeostasis, and are involved in male fertility and health. 9 Investigation of the possible mechanisms that lead to the onset and progression of prostate hypertrophy and prostate cancer is an area of active research,9-11 although the mechanism probably involves the excessive actions of androgens, the exact pathogenic mechanisms are not fully understood. 9 The present study was, therefore, undertaken to identify components of CM that could be used as therapeutic compounds to suppress the proliferation of prostate cells and to characterize the bioactive components of CM extract that affect the function of prostate cells.
Materials and Methods
Preparation of the Extract From CM Fruiting Bodies
A microbial strain of CM obtained from the National Institute of Technology and Evaluation (NBRC 100741) was cultured in a medium of Sabouraud dextrose broth with 1% wt/vol of yeast extract (SDY). The culture and extraction of CM fruiting bodies that parasitized Samia cynthia ricini (Ryoukyu-kaso in Japanese) were performed as previously described. 2 Briefly, the fruiting bodies of freeze-dried CM (45.2 g) were extracted with distilled water (900 mL) 3 times under reflux for 24 h. The solids were removed by centrifugation at 1610 g for 10 min, and the resulting aqueous solution was freeze-dried to provide an aqueous extract (33.2 g, 74% of the initial weight).
The aqueous extract (3.0 g) was eluted on a Sephadex LH-20 column with 50% MeOH in H2O to obtain 3 pooled fractions (Frs1-3), based on a thin-layer chromatographic analysis. Fr2 (190.4 mg) was subjected to silica gel column chromatography using chloroform–MeOH gradients (vol/vol, 10:1-0:1) for elution, to yield 3 subfractions (Frs2.1-2.3). Fr2.2 (10.2 mg) was purified using silica gel column chromatography using chloroform–MeOH gradients (vol/vol, 10:1-4:1) to obtain cordycepin (8.0 mg). Fr2.3 (23.1 mg) was purified using silica gel column chromatography with ethyl acetate–MeOH (vol/vol, 2:1) to obtain uridine (1.0 mg) and adenosine (12.9 mg). The 3 known nucleosides (cordycepin, uridine, and adenosine) were identified by comparing their NMR and MS data with those previously reported. 2 1H and 13C NMR spectra were recorded on a Bruker Biospin AVANCE III HD 400 spectrometer (400 MHz for 1H, 100 MHz for 13C). High-resolution electrospray ionization mass spectra were obtained using a Micromass LCT spectrometer with a time-of-flight analyzer. Plates precoated with silica gel containing a fluorescent indicator (Merck TLC silica gel 60 F254) were used for analytical and preparative thin-layer chromatography. Silica gel (Chromatorex PSQ 60B, Fuji Silysia Chemical) and Sephadex LH-20 gel (GE Healthcare) columns were used for column chromatography. All solvents used for column chromatography were of analytical grade (Kanto Chemical Co.).
Cell Culture
The human prostate cancer cell lines LNCaP and PC3 (Japanese Collection of Research Bioresources Cell Bank, National Institutes of Biomedical Innovation, Health and Nutrition) were cultured in RPMI 1640 medium (Fujifilm Wako Pure Chemical) including 10% fetal bovine serum, antibiotics, and antimycotics at 37 °C under 5% CO2 in humidified air. 2 Prostate cancer cells were pretreated with CM (10, 50, or 100 µg/mL), CM fractions (Fr1, Fr2, Fr3, Fr2.2, or Fr2.3; 10, 50, or 100 µg/mL), cordycepin (0.5, 2.5, or 5 µg/mL; Fujifilm Wako Pure Chemical), adenosine (1, 5, or 10 µg/mL; Sigma-Aldrich, St. Louis, MO, USA), uridine (0.1, 0.5, or 1 µg/mL; Sigma-Aldrich), 8-cyclopentyl-1,3-dipropylxanthine (CPDPX, an adenosine A1 receptor antagonist; 10 µM; Sigma-Aldrich), AB928 (an adenosine A2a/b receptor antagonist; 1 µM; Selleck Chemicals, Houston, TX, USA), or MRS1191 (an adenosine A3 receptor inhibitor; 20 µM, Sigma-Aldrich) for 1 h, and then treated with or without testosterone propionate (TP; 100 nM; Fujifilm Wako Pure Chemical) for 6 or 48 h.
Cell Viability and Proliferation Assays
LNCaP or PC3 cells were seeded in 96-well culture plates and then treated with TP (100 ng/mL; Fujifilm Wako Pure Chemical) in either the presence or absence of CM (10, 50, or 100 µg/mL), CM fractions (Fr1, Fr2, Fr3, Fr2.2, or Fr2.3; 10, 50, or 100 µg/mL), cordycepin (0.5, 2.5, or 5 µg/mL; Fujifilm Wako Pure Chemical), adenosine (1, 5, or 10 µg/mL; Sigma-Aldrich), or uridine (0.1, 0.5, or 1 µg/mL; Sigma-Aldrich) for 48 h. Cell viability and proliferation were assessed using a colorimetric WST-8 cell viability assay (Cell Counting Kit-8; Dojindo) according to the manufacturer's protocol. 2
RNA Extraction and Quantitative RT-PCR
Total RNA was extracted from cultured prostate cells using ISOGEN reagent (Nippon Gene) according to the manufacturer's protocol. Isolated RNA was reverse transcribed with a LunaScript RT SuperMix kit (New England Biolabs), and the generated cDNA was subjected to qPCR amplification using PowerUP SYBR Green master mix (Thermo Fisher Scientific). The primers are listed in Table 1. Calibration curves were used to confirm that the amplification efficiency of each target gene, and that of the reference gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were comparable. The average threshold (Ct) values for each target gene were determined using software for the Sequence Detection System (v2.3; Thermo Fisher Scientific). 12
Primers for Real-Time PCR Analyses.

Abbreviations: F, forward; R, reverse.
Western Blot Analysis
LNCaP cells were lysed in RIPA buffer (Cell Signaling Technology). The constituent proteins were separated by SDS-PAGE and transferred onto polyvinylidene difluoride membranes (Merck Millipore). The membranes were incubated with a mouse polyclonal antibody against phosphorylated AR (p-AR) (1:2000, Abcam) or a mouse monoclonal anti-GAPDH antibody (1:5000, Fujifilm Wako Pure Chemical). Immunoreactive bands were detected using enhanced chemiluminescence (Merck Millipore) after incubation with horseradish peroxidase-labeled goat antimouse IgG (1:5000; Vector Laboratories). 12
Statistical Analysis
All experimental data from the WST-8 assay and qPCR analysis represent the results of 3 or more independent experiments. Data are expressed as the mean ± SEM and were compared using Dunnett's test in R software (v4.0.5). P < .05 was considered statistically significant.
Results
The Inhibitory Effects of CM and its Fractions on the Viability and Proliferation of Prostate Cells
To identify the components of CM that affect cell viability, CM was fractionated by gel filtration chromatography, and the resulting 3 fractions (Fr1-3) were tested in a cell viability assay using androgen-responsive LNCaP cells and androgen-independent PC3 cells. CM treatment (10, 50, and 100 µg/mL) decreased the viability of LNCaP cells (Figure 1A and B). Treatment with Fr2 or Fr3 also decreased the viability of both prostate cancer cell lines in a concentration-dependent manner, but Fr1 had no effect (Figure 1A and B). Then, the effects of CM fractions were examined in the presence of TP. The TP-induced proliferation of LNCaP cells was inhibited by Fr2, Fr3, and CM (Figure 1C). The proliferation of PC3 cells in the presence of TP was inhibited by Fr2 and Fr3 (Figure 1D).

The inhibitory effects of Cordyceps militaris (CM) and its fractions on the viability and proliferation of prostate cells. LNCaP (A) and PC3 cells (B) were treated with either CM or fraction (Fr)1, Fr2, or Fr3 (10, 50, or 100 µg/mL), for 48 h, followed by WST-8 assay. *P < .05, **P < .01 versus control (Ctrl). LNCaP (C) and PC3 cells (D) were treated with CM, Fr1, Fr2, or Fr3 (10, 50, or 100 µg/mL) for 1 h and then stimulated with testosterone propionate (TP; 100 ng/mL). WST-8 assays were performed after 48 h. *P < .05 versus Ctrl. #P < .05, ##P < .01 versus TP.
The Effect of CM Fractions on Expression of Androgen-Regulated Signaling Factors in LNCaP Cells
To determine whether the CM fractions affect the androgen signaling pathway, the expression was examined of the androgen-regulated genes FKBP5, TMPRSS2, and KLK3 (prostate-specific antigen). TP-induced expression of these genes was decreased by Fr2, Fr2, and CM (Figure 2).
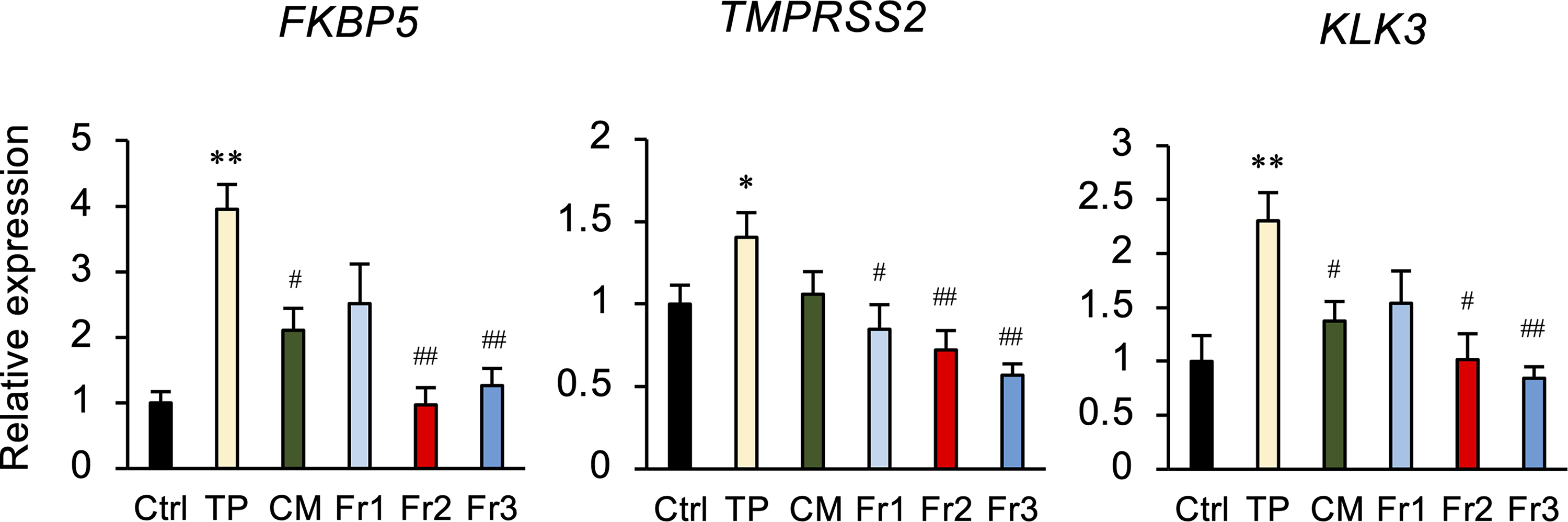
The effect of Cordyceps militaris (CM) fractions on the expression of androgen-regulated signaling factors in LNCaP cells. Cells were treated with either 50 µg/mL CM or fraction (Fr)1, Fr2, or Fr3, for 6 h, and RNAs extracted from each treatment group were subjected to qPCR to evaluate the expression of FKBP5, TMPRSS2, and KLK3 mRNA. GAPDH was used as an internal control for RNA integrity. Data are from 3 independent in vitro culture experiments. *P < .05, **P < .01 versus Ctrl. #P < .05, ##P < .01, versus testosterone propionate (TP).
The Inhibitory Effect of Fr2 and its Fractions on LNCaP Cell Proliferation
Since Fr2 showed the most potent inhibitory effect on cell proliferation and androgen-regulated gene expression, Fr2 was fractionated into 3 further fractions, Fr2.1-2.3, by silica gel column chromatography to identify the active components. Treatment of LNCaP cells with Fr2.2, Fr2.3, CM, or Fr2 decreased cell viability (Figure 3A). Furthermore, Fr2.2 and Fr2.3 inhibited TP-mediated proliferation of LNCaP cells (Figure 3B).

The inhibitory effect of Fr2 on proliferation of LNCaP cells. (A) Cells were treated with Cordyceps militaris (CM; 50 or 100 µg/mL), fraction (Fr)2 (50 µg/mL), Fr2.2 (10, 50, or 100 µg/mL), or Fr2.3 (10, 50, or 100 µg/mL) for 48 h, followed by a WST-8 assay. *P < .05, **P < .01 versus control (Ctrl). (B) Cells were treated with CM (50 or 100 µg/mL), Fr2 (50 or 100 µg/mL), Fr2.2 (10, 50, or 100 µg/mL), or Fr2.3 (10, 50, or 100 µg/mL) for 1 h and then stimulated with testosterone propionate (TP; 100 ng/mL). WST-8 assays were performed after 48 h. **P < .01 versus Ctrl.
The Effects of Cordycepin, Adenosine, and Uridine on Androgen-Dependent Activity
Based upon the results showing that Fr2.2 and Fr2.3 inhibited the proliferation of LNCaP cells, the fractions were subjected to NMR analysis to identify the active substances. The analysis identified cordycepin, adenosine, and uridine in Fr2.2 and Fr2.3. The content of cordycepin, adenosine, and uridine per gram of CM was 1.73, 4.3, and 0.33 mg, respectively. The effect of these compounds on the viability of LNCaP cells was examined. Cordycepin decreased cell viability in a concentration-dependent manner but neither adenosine nor uridine had any effect (Figure 4A). Cordycepin, but not adenosine or uridine, inhibited TP-induced proliferation of LNCaP cells (Figure 4B). In addition, TP-induced expression of FKBP5 and TMPRSS2 was decreased by cordycepin, adenosine, and uridine (Figure 4C). TP-induced expression of KLK3 was decreased by cordycepin and adenosine. TP-induced phosphorylation of AR was inhibited by cordycepin, adenosine, and uridine (Figure 4D).

The inhibitory effects of cordycepin, adenosine, and uridine on androgen-dependent activity in LNCaP cells. (A) LNCaP cells were treated with fraction (Fr)2 (50 µg/mL), cordycepin (Cor; 0.5, 2.5, or 5.0 µg/mL), adenosine (Ade; 1.0, 5.0, or 10 µg/mL), or uridine (Uri; 0.1, 0.5, or 1 µg/mL) for 48 h, followed by WST-8 assay. *P < .05, **P < .01 versus control (Ctrl). (B) Cells were treated with Fr2 (50 µg/mL), Cor (0.5, 2.5, or 5.0 µg/mL), Ade (1.0, 5.0, or 10 µg/mL), or Uri (0.1, 0.5, or 1 µg/mL) for 1 h and then stimulated with testosterone propionate (TP; 100 ng/mL). WST-8 assays were performed after 48 h. *P < .05 versus Ctrl, #P < .05 versus TP. (C) Cells were treated with Fr2 (50 µg/mL), Cor (5.0 µg/mL), Ade (10 µg/mL), or Uri (1 µg/mL) for 1 h and then stimulated with TP (100 ng/mL) for 6 h. RNAs were subjected to qPCR to evaluate the expression of FKBP5, TMPRSS2, and KLK3 mRNA. GAPDH was used as an internal control for RNA integrity. Data are from 3 independent in vitro culture experiments. **P < .01 versus Ctrl. #P < .05, ##P < .01 versus TP. (D) Expression of phosphorylated (p)-androgen receptor (AR) protein. Cells were treated with Cor (5.0 µg/mL) for 1 h and then stimulated with TP (100 ng/mL) for 6 h. Cell lysates were subjected to immunoblotting. GAPDH served as a loading control.
Role of the Adenosine A1 Receptor in the Inhibitory Actions of Cordycepin
Cordycepin is a natural adenosine analog that binds to adenosine receptors. Therefore, we used specific antagonists of adenosine A1–A3 receptors to determine which receptors mediate the actions of cordycepin on cell proliferation and expression of androgen-regulated genes in LNCaP cells. None of the antagonists modulated cordycepin-mediated inhibition of cell proliferation (Figure 5A). However, cordycepin-mediated decreases in FKBP5 and TMPRSS2 expression levels were produced by the A1 receptor antagonist CPDPX, but not by either the A2 receptor antagonist AB928 or the A3 receptor antagonist MRS1191 (Figure 5B). CPDPX tended to recover KLK3 expression levels, but the effect was not significant.
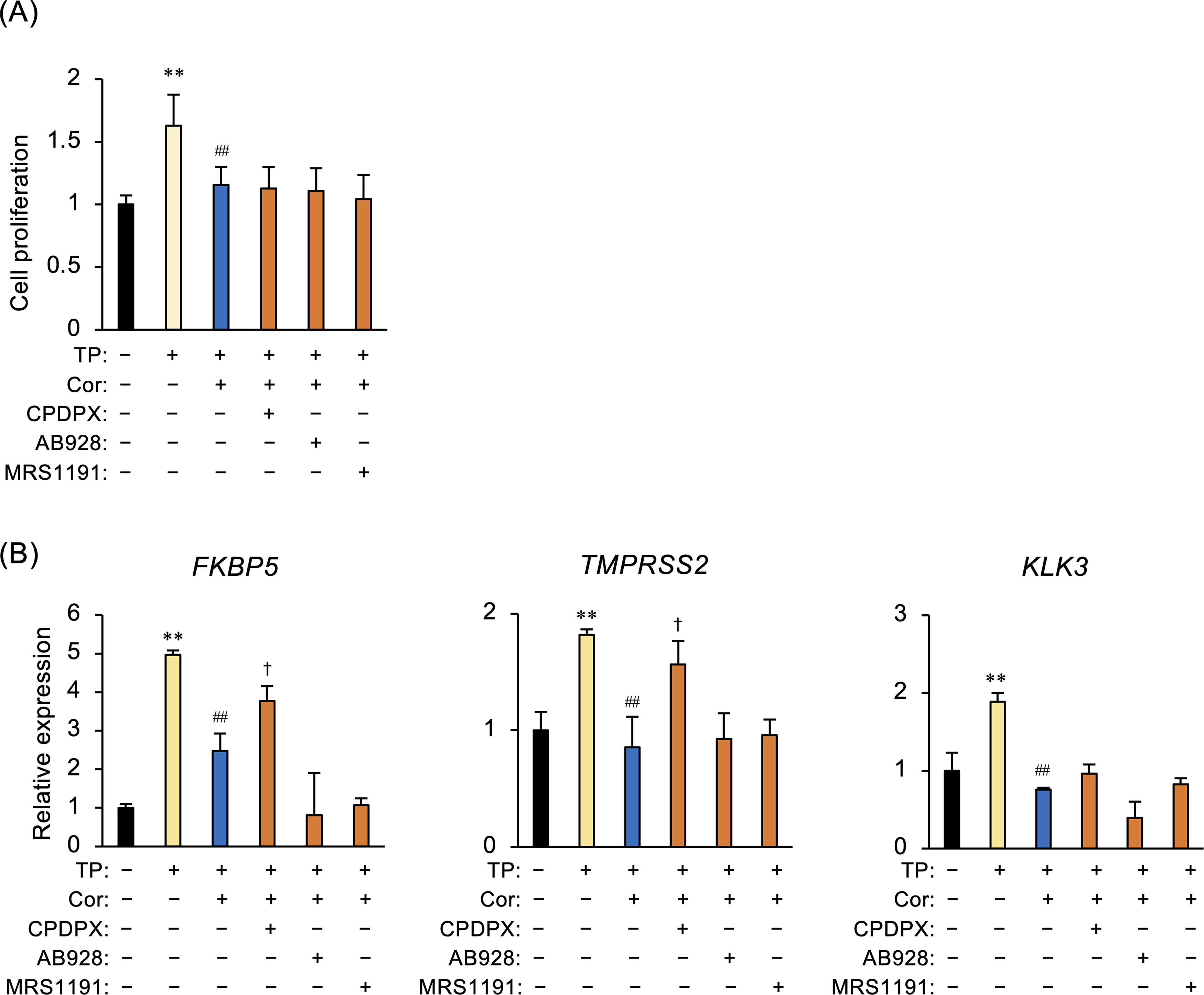
Role of the adenosine A1 receptor in the inhibitory actions of cordycepin in LNCaP cells. (A) LNCaP cells were pretreated with CPDPX (an adenosine A1 receptor antagonist, 10 µg/mL), AB928 (an adenosine A2 receptor antagonist, 1 µg/mL), or MRS1191 (an adenosine A3 receptor antagonist, 20 µg/mL) for 1 h and then treated with cordycepin (Cor; 5.0 µg/mL) for 1 h. Cells were cultured for 48 h in the presence of testosterone propionate (TP; 100 ng/mL), followed by WST-8 assays. **P < .01 versus Ctrl. ##P < .01 versus TP. (B) Cells pretreated with CPDPX, AB928, or MRS1191 were treated for 1 h with Cor (concentrations were as described above), followed by culture for 6 h in the presence of TP (100 ng/mL). Changes in expression of FKBP5, TMPRSS2, and KLK3 mRNA were evaluated by qPCR. GAPDH was used as an internal control for RNA integrity. Data are from 3 independent in vitro culture experiments. **P < .01 versus Ctrl. ##P < .01 versus TP. †P < .05 versus TP/Cor.
Discussion
In this study, we showed that CM-derived fractions decreased the viability of LNCaP and PC3 prostate cells and decreased TP-induced LNCaP cell proliferation in a concentration-dependent manner. We identified cordycepin, adenosine, and uridine as components of Fr2.2 and Fr2.3, which were derived from Fr2 of CM. Cordycepin, a natural adenosine analog was identified as the major active component of CM, and this compound reduced LNCaP cell viability and proliferation, while adenosine and uridine had no significant effect. Cordycepin downregulated the expression of androgen-regulated genes, which were blocked by an adenosine A1 receptor antagonist. Thus, our results show that the inhibitory effects of CM extract on prostate cell functions are, at least partially, mediated through cordycepin-mediated activation of the adenosine A1 receptor.
Androgen signaling via AR is associated with prostate homeostasis and tumorigenesis. AR signaling not only promotes the expression of fusion genes but also acts as part of a feedback loop to activate the AR and control its expression levels; these actions are involved in the pathogenesis of prostate cancer. 9 FKBP5 expression is regulated by AR signaling and encodes an immunophilin that regulates prostate physiology. 13 TMPRSS2 is an androgen-regulated cell-surface serine protease that is expressed primarily in the prostate epithelium. This enzyme is highly expressed in most human prostate cancers and metastatic prostate cancers. 14 Moreover, fusion of TMPRSS2 with the ETS (E twenty-six) transcription factor ERG is implicated in cancer development and progression.15,16 Cordycepin exhibits several bioactivities, including immunomodulatory effects, inhibition of tumor growth, and increasing the secretion of adrenocortical hormones.1,17 Here, we found that CM and cordycepin suppressed TP-induced phosphorylation of AR, and TP-induced increases in FKBP5, TMPRSS2, and KLK3 expression. Furthermore, the expression of FKBP5 and TMPRSS2 was restored by an adenosine A1 receptor antagonist. Cordyceps sinensis regulates the growth of prostate cancer cells via an androgen receptor–dependent pathway 18 but inhibits the proliferation of breast, ovarian, non-small cell lung, and colon cancer cells via androgen-independent signaling pathways.19-22 Adenosine is an endogenous mediator whose effects are exerted through 4 G protein-coupled receptors: A1, A2A, A2B, and A3.23,24 Stimulation of the A1 receptor decreases adenylyl cyclase activity and cAMP production and increases phospholipase C (PLC)-β activity and Ca2+ levels. Activation of the adenosine A1 receptor also induces the phosphorylation activity of mitogen-activated protein kinase p38, ERK1/2, and JNK1/2 in several types of cells. The antiproliferative effects of A1 receptor activation have been reported in colon cancer, breast cancer, and glioblastoma. 25 The A1 receptor reduces glioblastoma proliferation and increases sensitivity to chemotherapy by stimulating cell apoptosis. 26 Our findings suggest that CM and cordycepin may alter prostate cell functions by acting on adenosine A1 receptors to inhibit intracellular signaling pathways associated with androgen signaling. However, adenosine receptor antagonists did not modulate the effect of cordycepin on androgen-sensitive LNCaP cell proliferation. Previously, we showed that the CM-mediated reduction in prostate hypertrophy was independent of serum testosterone levels in a mouse BPH model. 2 Further, CM and cordycepin inhibited the viability of androgen-insensitive PC3 cells. Interestingly, cordycepin induces autophagy and cellular senescence 27 and inhibits cell proliferation by inhibiting apoptosis and the PI3K/AKT signaling pathway in cancer cells. 28 Cordyceps sinensis comprises a variety of bioactive components, including adenosine, β-glucan, ergosterol, and mannitol, as well as cordycepin. 6 In this study, we found that adenosine and uridine did not affect the proliferation of prostate cells. These findings suggest that the majority of the bioactivity of CM is mediated by cordycepin but not all the effects of cordycepin occur via the activity of the adenosine A1 receptor. CM might inhibit abnormal cell proliferation, such as that seen in prostate cancer cells and BPH, via autophagy and apoptosis; CM has little effect on the proliferation of non-prostate human endometrial epithelial cells. 2 Our data suggest that the adenosine A1 receptor modulates the function of cells that show abnormal proliferation, such as prostate cancer cells and those in BPH, through autophagy and apoptosis, independently of androgen signaling. Further studies are needed to elucidate the detailed pharmacology of cordycepin on prostate pathophysiology.
In conclusion, we identified cordycepin as the main active ingredient of CM that inhibits the proliferation of cultured prostate cells, and presumably the cells involved in BPH. The molecular mechanisms by which cordycepin in CM blocks prostate cell proliferation in androgen signaling- and adenosine signaling-dependent and -independent pathways may help identify novel therapies for BPH and castration-resistant prostate cancer.
Footnotes
Acknowledgments
The authors are grateful to Toyokazu Nakasone, CEO (Okinawa UKAMI Sericulture Co. Ltd) for providing Cordyceps militaris fruiting body.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Economy, Trade and Industry, (grant number the Strategic Foundational Technology Improvement).
